Revolutionizing 5-Bromoacetylsalicylate Production: A One-Step Commercial Scale-Up Strategy
Revolutionizing 5-Bromoacetylsalicylate Production: A One-Step Commercial Scale-Up Strategy
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for more efficient, safer, and cost-effective synthetic routes. A pivotal advancement in this domain is documented in Chinese Patent CN102557947A, which discloses a novel method for preparing 5-bromoacetylsalicylic acid methyl ester. This compound serves as a critical building block in the synthesis of various bioactive molecules, and its production has historically been plagued by low yields and hazardous processing conditions. The patented technology introduces a streamlined, one-step Friedel-Crafts acylation strategy that fundamentally alters the economic and safety profile of the supply chain. By directly reacting methyl salicylate with 2-bromoacetyl halides in the presence of a Lewis acid catalyst, this innovation bypasses the cumbersome multi-step sequences of the past. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize the commercial scale-up of complex pharmaceutical intermediates while adhering to stricter environmental and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this breakthrough, the industrial synthesis of 5-bromoacetylsalicylic acid methyl ester relied on inefficient and dangerous methodologies that posed substantial risks to both operational continuity and personnel safety. The traditional "Method 1" involved a two-step sequence starting with wintergreen oil (methyl salicylate), undergoing Friedel-Crafts acylation with acetyl chloride to form an intermediate, followed by a separate bromination step using liquid bromine. As illustrated in the reaction scheme below, this approach suffers from severe drawbacks, primarily due to the use of elemental liquid bromine, which is highly corrosive to equipment and poses serious environmental pollution risks.
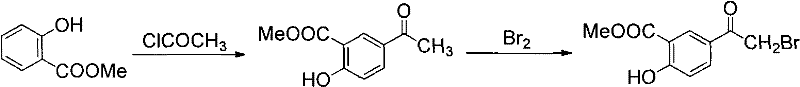
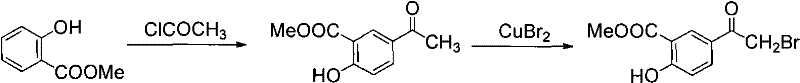
Furthermore, the cumulative yield of this two-step process is notoriously poor, often falling below 50%, which drastically inflates the cost of goods sold (COGS). An alternative "Method 2" attempted to mitigate the hazards of liquid bromine by utilizing cupric bromide for the bromination step. While this reduced some environmental toxicity, as shown in the subsequent diagram, it failed to resolve the fundamental issue of low overall efficiency, with total recovery rates still remaining under the 50% threshold. Additionally, the operational complexity of isolating intermediates and managing heavy metal waste streams made these conventional routes economically unviable for modern, high-volume production requirements.
Another literature-reported approach utilized salicylaldehyde as a starting material, reacting it with bromoacetyl chloride to generate the target aldehyde derivative. Although this method avoided liquid bromine, it yielded only approximately 66% and required specific aldehyde precursors that might not be as readily available or cost-effective as methyl salicylate. These historical limitations underscore the critical need for a process intensification strategy that consolidates steps and utilizes safer reagents, a gap that the new patent technology successfully fills.
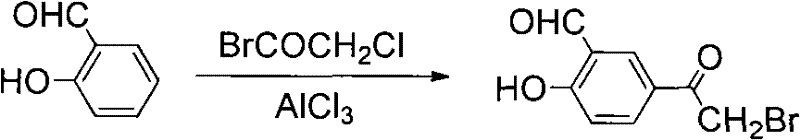
The Novel Approach
The innovative methodology presented in CN102557947A disrupts the status quo by collapsing the synthesis into a single, direct acylation step. Instead of building the carbon skeleton and then brominating it, or starting from a different aldehyde precursor, this route directly installs the bromoacetyl group onto the methyl salicylate ring. This is achieved through a Lewis acid-catalyzed Friedel-Crafts reaction between wintergreen oil and 2-bromoacetyl halides (specifically 2-bromoacetyl bromide or chloride). This strategic shift eliminates the need for intermediate isolation, thereby reducing solvent consumption, labor hours, and energy usage associated with heating and cooling cycles across multiple stages. The result is a robust process capable of achieving yields as high as 93.2%, nearly doubling the efficiency of the best prior art methods.
Mechanistic Insights into Lewis Acid-Catalyzed Friedel-Crafts Acylation
From a mechanistic perspective, the success of this reaction hinges on the precise activation of the acylating agent by the Lewis acid catalyst. When 2-bromoacetyl bromide interacts with a strong Lewis acid such as aluminum chloride (AlCl3), the carbonyl oxygen coordinates with the aluminum center, generating a highly electrophilic acylium ion species. This activated complex is then attacked by the electron-rich aromatic ring of the methyl salicylate. The regioselectivity of this substitution is governed by the directing effects of the existing substituents on the benzene ring: the hydroxyl group (-OH) is a strong ortho/para director, while the ester group (-COOMe) is a meta director. Fortunately, the position para to the hydroxyl group (position 5) is also meta to the ester group, creating a synergistic effect that directs the incoming electrophile exclusively to the desired 5-position. This inherent electronic bias ensures high purity of the crude product, minimizing the formation of regioisomers that would otherwise require difficult and yield-losing purification steps.
Impurity control is further enhanced by the mild reaction conditions employed, typically ranging from 25°C to 65°C. Unlike harsh bromination conditions that can lead to poly-bromination or ring degradation, this acylation pathway is chemically selective. The choice of solvent also plays a pivotal role in stabilizing the transition state and managing the exothermic nature of the reaction. Solvents such as methylene chloride, chloroform, or nitromethane are effective in solubilizing both the organic substrates and the Lewis acid complexes. By maintaining strict control over the molar ratios—optimally 1:1.3:3 for methyl salicylate, acyl halide, and Lewis acid respectively—the process minimizes side reactions such as hydrolysis of the ester or the acyl halide. This level of mechanistic control translates directly to a cleaner impurity profile, which is a critical parameter for high-purity pharmaceutical intermediates intended for downstream drug synthesis.
How to Synthesize 5-Bromoacetylsalicylic Acid Methyl Ester Efficiently
Implementing this synthesis requires careful attention to reagent quality and addition rates to manage the exotherm effectively. The general protocol involves suspending the Lewis acid in an anhydrous solvent, adding the acyl halide to form the active complex, and then slowly dripping in the methyl salicylate. The reaction is typically monitored until completion, usually within 12 to 16 hours, followed by a standard aqueous workup involving acidification and extraction. This straightforward procedure eliminates the need for specialized pressure vessels or cryogenic cooling, making it accessible for standard multipurpose reactors. For a detailed breakdown of the specific stoichiometric ratios, temperature profiles, and workup parameters validated in the patent examples, please refer to the standardized guide below.
- Prepare the reaction system by dissolving the Lewis acid catalyst (such as aluminum chloride) and the acylating agent (2-bromoacetyl bromide) in an anhydrous organic solvent like methylene chloride.
- Slowly add methyl salicylate (wintergreen oil) to the reaction mixture while maintaining a controlled temperature between 25°C and 65°C to ensure regioselectivity.
- Upon completion, quench the reaction with an ice-water mixture, adjust pH to acidic conditions, extract with organic solvent, and purify via recrystallization to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers transformative benefits that extend far beyond simple yield improvements. The shift from a multi-step, hazardous process to a single-step, mild reaction fundamentally de-risks the supply chain. By eliminating the requirement for liquid bromine, manufacturers can avoid the stringent regulatory compliance costs and specialized storage infrastructure associated with this hazardous material. Furthermore, the removal of the intermediate isolation step significantly reduces the turnaround time for production batches. This agility allows suppliers to respond more rapidly to fluctuating market demands, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring a more reliable flow of materials to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the drastic simplification of the operational workflow. By consolidating two distinct chemical transformations into a single reactor charge, the method effectively halves the labor intensity and utility consumption (steam, cooling water, electricity) per kilogram of output. Moreover, the substantial increase in yield—from less than 50% to over 90%—means that nearly double the amount of product can be generated from the same quantity of raw materials. This material efficiency, combined with the avoidance of expensive and hazardous reagents like liquid bromine or cupric bromide, results in a significantly lower cost base, enabling competitive pricing strategies without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. This new route relies on methyl salicylate (wintergreen oil) and 2-bromoacetyl bromide, both of which are commodity chemicals available from a broad global supplier base. In contrast, older methods relying on specific aldehyde precursors or hazardous elemental bromine were susceptible to supply disruptions caused by environmental regulations or production incidents at bromine plants. By utilizing robust, widely available starting materials and standard Lewis acid catalysts, this method ensures a stable and continuous supply capability, mitigating the risk of production stoppages that could jeopardize customer commitments.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen engineering challenges, particularly regarding heat transfer and waste management. This synthesis is inherently scalable because it operates at near-ambient temperatures (25°C to 65°C) and atmospheric pressure, removing the need for complex high-pressure or cryogenic equipment. From an environmental standpoint, the process generates significantly less waste; the absence of heavy metal bromination byproducts (like copper salts) simplifies wastewater treatment and reduces the burden on EHS (Environment, Health, and Safety) departments. This alignment with green chemistry principles facilitates easier permitting and long-term operational sustainability, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing on a multi-ton scale.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this intermediate into their supply chains, we have compiled answers to common questions based on the specific technical data provided in the patent documentation. These insights address the practical aspects of catalyst selection, reaction optimization, and the comparative advantages over legacy methods. Understanding these nuances is essential for R&D directors who are tasked with validating new vendors and ensuring that the quality of the intermediate meets the rigorous specifications required for GMP production environments.
Q: What are the primary advantages of this new synthesis method over traditional routes?
A: The new method eliminates the need for hazardous liquid bromine and reduces the process from two or more steps to a single direct acylation, significantly improving overall yield from below 50% to over 90%.
Q: Which Lewis acid catalysts are most effective for this transformation?
A: While various Lewis acids such as iron trichloride, zinc chloride, and boron trifluoride can be used, aluminum chloride has demonstrated superior performance in terms of yield and reaction rate in the patented examples.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes common industrial solvents like methylene chloride and avoids highly corrosive reagents like liquid bromine, making it highly scalable and safer for equipment maintenance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromoacetylsalicylic Acid Methyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route is only valuable if it can be executed with precision and consistency at scale. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our facilities are equipped with state-of-the-art corrosion-resistant reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-bromoacetylsalicylic acid methyl ester meets the exacting standards of the global pharmaceutical industry.
We invite you to explore how this optimized synthesis can enhance your project's economics and timeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
