Advanced Metal-Free Synthesis of Dibenzo Oxazepine Derivatives for Commercial Pharma Applications
Advanced Metal-Free Synthesis of Dibenzo Oxazepine Derivatives for Commercial Pharma Applications
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign pathways for constructing complex heterocyclic scaffolds. Patent CN113072518A introduces a groundbreaking methodology for the synthesis of 8-(9-aryl-9-fluorenyl)-dibenzo[b,f][1,4]oxazepine derivatives, a class of compounds with significant potential in medicinal chemistry due to their diverse biological activities including antidepressant and antiviral properties. This innovation addresses critical pain points in traditional organic synthesis by eliminating the reliance on precious transition metal catalysts and harsh oxidants. The core structural motif, as depicted in the general formula below, features a rigid fluorenyl backbone attached to the dibenzoxazepine core, offering unique conformational constraints that are highly valued in structure-activity relationship (SAR) studies.
![General chemical structure of 8-(9-aryl-9-fluorenyl)-dibenzo[b,f][1,4]oxazepine derivatives showing variable substituents R1-R5](/insights/img/dibenzo-oxazepine-synthesis-pharma-supplier-20260308043706-010.webp)
For R&D directors and process chemists, the significance of this patent lies in its departure from conventional catalytic systems. Historically, functionalizing the benzene ring of dibenzoxazepines required sophisticated and costly metal complexes. The disclosed method utilizes trifluoromethanesulfonic acid (TfOH) as a potent Brønsted acid catalyst in acetonitrile, enabling the direct coupling of 9-aryl-9-fluorenols with dibenzoxazepine precursors. This shift not only aligns with the principles of green chemistry by reducing toxic waste but also streamlines the purification process, a key factor for procurement teams evaluating the total cost of ownership for critical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the functionalization of dibenzoxazepine derivatives has been heavily reliant on transition metal catalysis, which presents substantial logistical and economic challenges for large-scale manufacturing. For instance, earlier reports by Zhang et al. utilized iridium complexes such as [Ir(COD)Cl]2 coupled with chiral phosphine ligands to effect hydrogenation or functionalization. As illustrated in the reaction scheme below, these methods necessitate stringent exclusion of air and moisture, expensive ligand systems, and rigorous post-reaction metal scavenging steps to ensure the final product meets ICH Q3D guidelines for elemental impurities.
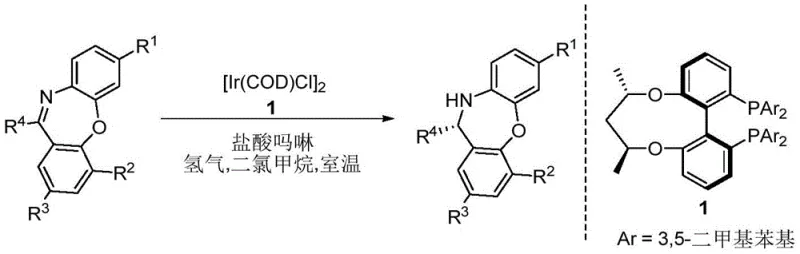
Furthermore, alternative approaches involving organozinc reagents, such as the Aza-Reformatsky reactions reported by Pedro and Vila, introduce additional safety hazards associated with pyrophoric materials and moisture sensitivity. These conventional routes often suffer from limited substrate scope, where electron-withdrawing or bulky substituents can drastically reduce yields or enantioselectivity. The requirement for specialized catalysts like diarylprolinol or cyclic amino acids further inflates the raw material costs and complicates the supply chain, making these methods less attractive for the production of commodity-grade pharmaceutical intermediates where cost efficiency is paramount.
The Novel Approach
In stark contrast, the methodology disclosed in CN113072518A offers a streamlined, metal-free alternative that leverages the electrophilic activation of the hydroxyl group in 9-aryl-9-fluorenols. The reaction proceeds via a direct nucleophilic substitution mechanism catalyzed by trifluoromethanesulfonic acid, as clearly demonstrated in the synthetic pathway shown below. This approach allows for the efficient coupling of variously substituted fluorenols with the dibenzoxazepine core under relatively mild thermal conditions (120°C) without the need for inert atmosphere techniques beyond standard anhydrous preparations.
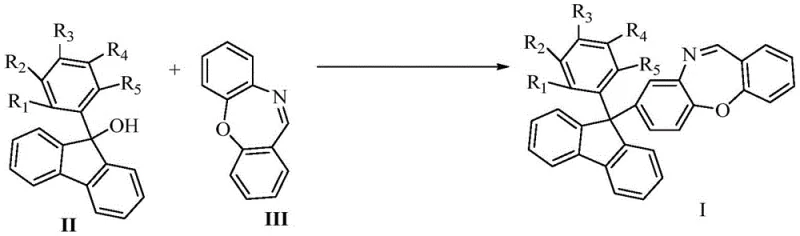
The versatility of this novel approach is evidenced by its tolerance to a wide range of functional groups, including halogens (F, Cl), alkyl groups (methyl, tert-butyl), and alkoxy groups (methoxy). By avoiding the use of transition metals, the process inherently reduces the risk of metal contamination, thereby simplifying the downstream purification workflow. The use of acetonitrile as a solvent further enhances the industrial viability of this route, as it is a common, recyclable solvent with well-established handling protocols in GMP facilities. This represents a significant technological leap forward, transforming a complex multi-step catalytic cycle into a straightforward, one-pot coupling reaction that is easier to control and optimize.
Mechanistic Insights into Acid-Catalyzed Nucleophilic Substitution
The mechanistic underpinning of this transformation involves the protonation of the hydroxyl group on the 9-aryl-9-fluorenol (Compound II) by the strong Brønsted acid catalyst, trifluoromethanesulfonic acid. This protonation converts the poor leaving group (-OH) into a highly reactive water molecule, facilitating the formation of a stabilized carbocation intermediate at the C9 position of the fluorene ring. The stability of this carbocation is enhanced by the resonance delocalization across the two aromatic rings of the fluorene system, which drives the reaction forward even with moderately nucleophilic partners. The dibenzoxazepine (Compound III) then acts as a nucleophile, attacking the electrophilic carbocation center to form the new C-C bond, resulting in the target 8-(9-aryl-9-fluorenyl) derivative.
From an impurity control perspective, this mechanism offers distinct advantages over radical or metal-mediated pathways. The absence of redox-active metal species minimizes the formation of oxidative byproducts or homocoupling impurities that are often difficult to separate. Furthermore, the reaction conditions are sufficiently mild to prevent the decomposition of the sensitive imine or ether linkages within the dibenzoxazepine scaffold. The selectivity is primarily governed by the stability of the carbocation intermediate and the nucleophilicity of the heterocycle, allowing for predictable outcomes across different substituents. This predictability is crucial for R&D teams aiming to rapidly generate libraries of analogs for biological screening without encountering unpredictable side reactions.
How to Synthesize 8-(9-aryl-9-fluorenyl)-dibenzo[b,f][1,4]oxazepine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these high-value intermediates with consistent quality. The process begins with the preparation of the key building blocks: the 9-aryl-9-fluorenol and the dibenzoxazepine core. The fluorenol is synthesized via a Grignard reaction between substituted bromobenzene and 9-fluorenone, a robust and well-understood transformation in organic synthesis. The dibenzoxazepine core is prepared through a condensation reaction between 2-aminophenol and 2-fluorobenzaldehyde in polyethylene glycol, utilizing potassium carbonate as a base. These precursors are then combined in the final coupling step.
- Prepare 9-aryl-9-fluorenol (Compound II) by reacting substituted bromobenzene with magnesium to form a Grignard reagent, followed by addition to 9-fluorenone in anhydrous THF.
- Synthesize dibenzo[b,f][1,4]oxazepine (Compound III) via condensation of 2-aminophenol and 2-fluorobenzaldehyde in polyethylene glycol with potassium carbonate at 100°C.
- Couple Compound II and Compound III in acetonitrile using 10 mol% trifluoromethanesulfonic acid catalyst at 120°C for 24 hours to yield the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route translates into tangible operational efficiencies and risk mitigation. The most immediate impact is the elimination of precious metal catalysts such as iridium or palladium, which are subject to volatile market pricing and geopolitical supply constraints. By replacing these expensive inputs with commodity chemicals like trifluoromethanesulfonic acid and acetonitrile, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing. This shift not only lowers the direct material costs but also reduces the working capital tied up in expensive catalyst inventory.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts eliminates the need for specialized metal scavenging resins and extensive purification steps required to meet strict residual metal specifications. This simplification of the downstream processing significantly reduces solvent consumption and waste disposal costs. Furthermore, the high atom economy of the coupling reaction ensures that raw materials are efficiently converted into the desired product, minimizing waste generation and maximizing overall process yield. The use of readily available starting materials like bromobenzene derivatives further stabilizes the cost structure against supply chain shocks.
- Enhanced Supply Chain Reliability: The reliance on commodity reagents rather than bespoke chiral ligands or air-sensitive organometallics greatly enhances supply chain resilience. Suppliers can source trifluoromethanesulfonic acid and acetonitrile from multiple global vendors, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions, which do not require ultra-low temperatures or extreme pressures, allows for production in a wider range of manufacturing facilities, including those with standard glass-lined reactor capabilities. This flexibility ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process aligns perfectly with modern environmental, health, and safety (EHS) standards by avoiding toxic heavy metals and pyrophoric reagents. This facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. The reaction operates at moderate temperatures (120°C) and atmospheric pressure, making it inherently safer to scale up from kilogram to multi-ton quantities. The simplified waste stream, devoid of heavy metal contaminants, lowers the cost and complexity of wastewater treatment, supporting sustainable manufacturing practices and long-term operational license.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making.
Q: Why is the transition-metal-free approach significant for pharmaceutical intermediates?
A: Traditional methods often rely on expensive iridium or zinc catalysts which require complex removal processes to meet strict residual metal limits in APIs. This novel acid-catalyzed route eliminates heavy metals entirely, simplifying purification and reducing regulatory burden.
Q: What are the typical yields and purity profiles for this synthesis?
A: The patent reports isolated yields ranging from 59% to 76% across various substituents (e.g., methyl, methoxy, tert-butyl, halogen). The mild reaction conditions minimize side reactions, facilitating the production of high-purity intermediates suitable for downstream drug synthesis.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process utilizes commercially available starting materials like bromobenzene and 9-fluorenone, and avoids sensitive reagents like organozinc compounds. The use of standard solvents like acetonitrile and robust acid catalysis supports safe scale-up from laboratory to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-(9-aryl-9-fluorenyl)-dibenzo[b,f][1,4]oxazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative, green synthesis routes like the one described in CN113072518A for the production of complex heterocyclic intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the highest standards required for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this metal-free technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical solution available in the market.
