Advanced Catalytic Synthesis of Quinoline-2 Formate Derivatives for Commercial Pharmaceutical Applications
Advanced Catalytic Synthesis of Quinoline-2 Formate Derivatives for Commercial Pharmaceutical Applications
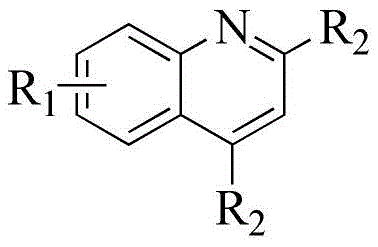
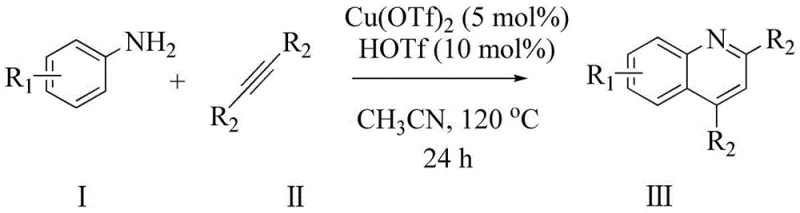
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for efficient, scalable, and environmentally benign synthetic routes for high-value intermediates. A pivotal advancement in this domain is documented in patent CN106380446B, which discloses a robust methodology for the synthesis of quinoline-2 formate derivatives. This technology represents a significant leap forward from classical approaches, utilizing a copper-catalyzed cyclization strategy that merges aromatic amines with electron-withdrawing alkynes. For global procurement leaders and R&D directors seeking a reliable quinoline-2 formate derivatives supplier, understanding the nuances of this catalytic system is crucial. The process leverages copper(II) triflate [Cu(OTf)₂] and triflic acid (HOTf) to facilitate the construction of the quinoline core under relatively mild thermal conditions, offering a compelling alternative to legacy methods that often suffer from harsh reaction environments and poor atom economy.
The strategic importance of quinoline scaffolds cannot be overstated, given their ubiquity in bioactive molecules, agrochemicals, and functional materials. The ability to access these structures with high regioselectivity and minimal byproduct formation is a key differentiator in modern process chemistry. This specific patent highlights a pathway that not only simplifies the operational workflow but also enhances the overall safety profile of the manufacturing process. By eliminating the need for stoichiometric amounts of strong mineral acids or toxic oxidants typically associated with older quinoline syntheses, this method aligns perfectly with the green chemistry principles that are increasingly mandated by regulatory bodies and corporate sustainability goals alike. Consequently, adopting this technology can lead to substantial cost reduction in pharmaceutical intermediates manufacturing by streamlining waste treatment and reducing raw material consumption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of quinoline derivatives has relied heavily on the Skraup synthesis and the Combes condensation, both of which present significant logistical and safety challenges for large-scale operations. The Skraup method, while classic, necessitates the use of concentrated sulfuric acid at elevated temperatures, often requiring nitrobenzene or arsenic acid as oxidants, which introduces severe toxicity hazards and complicates waste disposal protocols. Furthermore, the Combes synthesis, which involves the condensation of aromatic amines with beta-diketones, is frequently plagued by issues regarding substrate compatibility; specifically, the presence of electron-withdrawing groups on the aromatic amine ring drastically reduces the electron cloud density, thereby inhibiting the necessary electrophilic substitution steps. This limitation severely restricts the chemical space accessible via traditional routes, forcing chemists to resort to multi-step protection-deprotection sequences that inflate costs and extend lead times.
In addition to substrate limitations, conventional methods often suffer from poor selectivity, leading to complex mixtures of isomers that are difficult and expensive to separate. For instance, when unsymmetrical 1,3-dicarbonyl compounds are used in Combes-type reactions, the initial condensation can proceed in two directions, generating a mixture of beta-amino-ketenes that ultimately cyclize into isomeric quinoline products. Resolving these mixtures requires extensive chromatographic purification or recrystallization efforts, which significantly impacts the overall yield and throughput of the manufacturing process. Moreover, the harsh acidic conditions inherent in these legacy processes can degrade sensitive functional groups, limiting the diversity of the final molecular library and hindering the development of next-generation active pharmaceutical ingredients (APIs) that require delicate structural motifs.
The Novel Approach
In stark contrast to these archaic techniques, the method disclosed in patent CN106380446B utilizes a transition metal-catalyzed annulation strategy that operates under significantly milder and more controlled conditions. By employing a catalytic system composed of Cu(OTf)₂ and HOTf, the reaction effectively activates the electron-withdrawing alkyne towards nucleophilic attack by the aromatic amine, facilitating a smooth cyclization to form the quinoline ring. This approach bypasses the need for aggressive dehydrating agents or toxic oxidants, thereby reducing the environmental footprint and improving operator safety. The reaction tolerates a wide array of functional groups, including halogens and trifluoromethyl groups, which are critical for modulating the pharmacokinetic properties of drug candidates. This broad substrate scope allows for the direct synthesis of complex, functionalized quinolines without the need for cumbersome protecting group strategies.
Furthermore, the novel approach offers superior selectivity and ease of purification, as evidenced by the high purity levels reported in the patent examples. The reaction proceeds cleanly to afford the desired quinoline-2 formate derivatives with minimal byproduct formation, simplifying the downstream processing requirements. Instead of grappling with complex isomeric mixtures, manufacturers can rely on straightforward extraction and column chromatography to isolate the target compound in high yields. This efficiency translates directly into improved process economics, as it reduces the consumption of solvents and silica gel while maximizing the output per batch. For supply chain managers, this reliability ensures a consistent flow of high-purity quinoline intermediates, mitigating the risks associated with production bottlenecks and quality failures that are common with less robust synthetic methodologies.
Mechanistic Insights into Cu(OTf)₂-Catalyzed Cyclization
The efficacy of this synthetic route hinges on the synergistic interaction between the copper catalyst and the triflic acid additive, which together create a highly reactive environment for the cyclization event. The mechanism likely involves the coordination of the copper center to the electron-deficient alkyne, increasing its electrophilicity and rendering it susceptible to nucleophilic attack by the amino group of the aniline substrate. The presence of HOTf serves to protonate intermediate species and stabilize the transition states, effectively lowering the activation energy barrier for the ring-closing step. This dual-activation mode ensures that even less nucleophilic anilines, such as those bearing electron-withdrawing substituents, can participate efficiently in the reaction. The result is a streamlined catalytic cycle that minimizes catalyst loading while maintaining high turnover frequencies, a critical factor for scaling up the process to commercial quantities.
From an impurity control perspective, the specificity of this copper-catalyzed pathway is advantageous because it avoids the radical-mediated side reactions often seen in oxidative cyclizations. The mild thermal conditions (80°C to 120°C) prevent the thermal degradation of sensitive ester groups on the alkyne moiety, ensuring that the integrity of the quinoline-2 formate structure is preserved throughout the synthesis. Additionally, the use of acetonitrile or similar polar aprotic solvents facilitates the solubility of both organic substrates and the ionic catalyst species, promoting homogeneous reaction kinetics. This homogeneity is essential for achieving consistent batch-to-batch reproducibility, a key metric for commercial scale-up of complex pharmaceutical intermediates. By understanding these mechanistic underpinnings, process chemists can fine-tune reaction parameters to optimize yield and purity further, ensuring that the final product meets the stringent specifications required for clinical and commercial applications.
How to Synthesize Quinoline-2 Formate Derivatives Efficiently
The execution of this synthesis requires precise adherence to the molar ratios and thermal profiles outlined in the patent to ensure optimal conversion and selectivity. The process begins with the sequential addition of the aromatic amine and the electron-withdrawing alkyne into a reaction vessel, followed by the introduction of the solvent and the catalytic system. Maintaining the correct stoichiometry is vital, as an excess of the alkyne helps drive the equilibrium towards the product while compensating for any potential volatility or side reactions. The reaction mixture is then subjected to controlled heating in an oil bath, allowing the cyclization to proceed over a defined period before the workup phase commences. For a detailed breakdown of the specific operational parameters and safety precautions, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with aromatic amine and electron-withdrawing alkyne in a molar ratio of 1: 2 to 4, along with the appropriate solvent.
- Add the catalyst system comprising Cu(OTf)2 (0.8-5 mol%) and HOTf additive (1.8-10 mol%) to the reaction mixture.
- Heat the mixture in an oil bath at 80-120°C for 8-24 hours, then cool, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed synthesis offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant simplification of the supply chain for raw materials; anilines and activated alkynes are commodity chemicals that are readily available from multiple global sources, reducing the risk of supply disruptions. Unlike methods that rely on bespoke or hazardous reagents, this process utilizes stable and commercially viable inputs, which stabilizes pricing and ensures long-term availability. Furthermore, the elimination of toxic oxidants like arsenic acid removes a major regulatory burden, simplifying the permitting process for manufacturing facilities and reducing the costs associated with hazardous waste disposal and environmental compliance monitoring.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive catalysts and the high atom economy of the cyclization reaction. By avoiding the need for stoichiometric amounts of strong acids and oxidants, the process significantly lowers the cost of goods sold (COGS). Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the consumption of purification media such as silica gel and solvents. This efficiency translates into lower operational expenditures and a more competitive pricing structure for the final pharmaceutical intermediates, allowing buyers to achieve better margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more resilient supply chain. Because the process tolerates a wide range of functional groups and does not require extreme pressures or cryogenic temperatures, it is less susceptible to equipment failures or operational upsets. This reliability ensures consistent production schedules and shorter lead times for customers awaiting critical building blocks for their drug discovery programs. The ability to source high-purity quinoline derivatives from a stable and predictable manufacturing process is a strategic asset for companies looking to secure their API supply chains against market volatility and geopolitical uncertainties.
- Scalability and Environmental Compliance: From a sustainability perspective, this method aligns with modern green chemistry initiatives by reducing the generation of hazardous waste. The absence of heavy metal contaminants (other than the trace copper catalyst which is easily managed) and toxic byproducts simplifies the effluent treatment process, making it easier to meet strict environmental discharge limits. This environmental compatibility facilitates smoother regulatory approvals and enhances the corporate social responsibility (CSR) profile of the supply chain. Moreover, the simplicity of the workup procedure suggests that the process can be scaled from laboratory benchtop to multi-ton production with minimal re-engineering, supporting the commercial scale-up of complex heterocyclic compounds required for late-stage clinical and commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these quinoline derivatives. These answers are derived directly from the experimental data and technical disclosures found within the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing workflows and assess the potential impact on their product development timelines.
Q: What are the primary advantages of this method over traditional Skraup synthesis?
A: Unlike the traditional Skraup synthesis which requires harsh concentrated sulfuric acid, high temperatures, and toxic oxidants like nitrobenzene or arsenic acid, this novel copper-catalyzed method operates under significantly milder conditions (80-120°C) with safer reagents, resulting in less environmental pollution and easier handling.
Q: Does this synthesis tolerate electron-withdrawing groups on the aniline ring?
A: Yes, the method demonstrates excellent substrate scope and is specifically effective for anilines substituted with electron-withdrawing groups such as fluoro, chloro, bromo, and trifluoromethyl groups, which often pose challenges in electrophilic substitution reactions like the Combes synthesis.
Q: What purity levels can be achieved with this synthetic route?
A: The patent data indicates that the synthesized quinoline derivatives can achieve exceptional purity levels, with specific examples reporting purity as high as 99.9% following standard column chromatography purification, making them highly suitable for sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline-2 Formate Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the acceleration of drug discovery and development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the one described in CN106380446B, we can offer customized manufacturing solutions that optimize both cost and performance for our global partners.
We invite you to collaborate with us to explore how this efficient synthetic route can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this methodology can improve your bottom line. Please contact us today to request specific COA data for our quinoline portfolio or to discuss route feasibility assessments for your proprietary targets. Let us be your partner in navigating the complexities of fine chemical synthesis and securing a reliable supply of essential building blocks for the future.
