Advanced Manufacturing of Cyclopropyl Methyl Ketone for High-Purity Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries constantly demand higher purity intermediates to ensure the safety and efficacy of final active ingredients. Patent CN114573435A introduces a groundbreaking preparation method for cyclopropyl methyl ketone, a critical building block for broad-spectrum antibacterial agents like ciprofloxacin and various pyrethroid pesticides. This technical insight report analyzes the novel synthetic route which shifts away from traditional, costly raw materials towards a more economical alpha-acetyl-gamma-butyrolactone precursor. By integrating a unique magnetic impurity removal system, this methodology addresses longstanding challenges regarding byproduct formation, specifically DHMF, offering a robust pathway for cost reduction in pharmaceutical intermediates manufacturing. For R&D directors and procurement specialists, understanding this shift is vital for optimizing supply chains and achieving stringent purity specifications required for GMP-grade production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of cyclopropyl methyl ketone relies heavily on the direct cyclization of 5-chloro-2-pentanone. While chemically straightforward, this conventional approach suffers from significant economic and technical drawbacks that hinder large-scale efficiency. The primary issue lies in the high cost and limited availability of 5-chloro-2-pentanone itself, which acts as a bottleneck for commercial scale-up of complex pharmaceutical intermediates. Furthermore, the cyclization process often generates substantial amounts of low-boiling organic extractants and stubborn byproducts like 2-methyl-4,5-dihydrofuran (DHMF). These impurities are notoriously difficult to separate via standard rectification, often requiring energy-intensive distillation columns that degrade yield and increase the carbon footprint of the manufacturing process. Consequently, manufacturers face inconsistent purity profiles that can jeopardize downstream synthesis of chiral alcohols or perfume ketones.
The Novel Approach
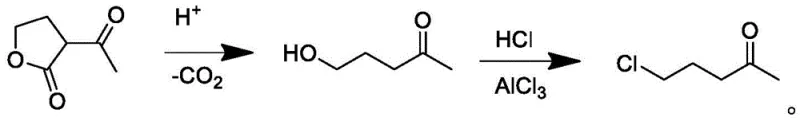
The innovative strategy outlined in the patent data fundamentally reengineers the upstream synthesis by utilizing alpha-acetyl-gamma-butyrolactone as the foundational raw material. This lactone derivative undergoes a ring-opening decarboxylation and nucleophilic chlorination in a hot hydrochloric acid medium to generate 5-chloro-2-pentanone in situ or as a dedicated intermediate step. This switch not only leverages a cheaper, more abundant feedstock but also integrates seamlessly with the subsequent cyclization phase. The process flow is streamlined to minimize unit operations, thereby enhancing the overall atom economy. By controlling the formation of the chloroketone precursor through specific catalytic conditions involving anhydrous aluminum trichloride, the method ensures a cleaner feed into the cyclization reactor, setting the stage for superior final product quality.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Magnetic Purification
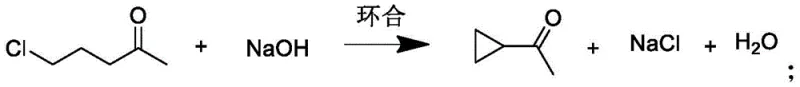
The core chemical transformation involves an intramolecular nucleophilic substitution where the terminal chloride displaces a proton to form the strained cyclopropane ring. As depicted in the reaction scheme, treating the chloroketone with a sodium hydroxide solution at 95-100 ℃ facilitates this ring closure efficiently. The molar ratio of 5-chloro-2-pentanone to sodium hydroxide is precisely maintained at 1:1.5 to ensure complete conversion while minimizing side reactions such as aldol condensation. This base-catalyzed cyclization is exothermic and requires careful thermal management to prevent the degradation of the sensitive cyclopropyl moiety. The reaction mixture naturally separates into layers upon completion, allowing for the immediate collection of the oil layer containing the crude ketone, which simplifies the initial workup procedure significantly compared to aqueous extraction methods.
Beyond the primary synthesis, the most distinct mechanistic advancement is the targeted removal of DHMF impurities using a reactive magnetic scavenger. The crude product typically contains DHMF, which co-distills with the target ketone. To eliminate this, the process employs a radical initiator, such as dibenzoyl peroxide or azobisisobutyronitrile, alongside a functionalized nano ferroferric oxide agent. Under heating at 60-80 ℃, free radicals initiate a polymerization reaction between the unsaturated double bonds on the surface of the magnetic particles and the DHMF molecules. This effectively traps the impurity onto the solid magnetic carrier. Post-reaction, a simple external magnet is used to adsorb and remove these particles, filtering out the impurities without the need for complex chromatography or repeated distillation. This mechanism ensures the final product achieves purity levels suitable for high-value API synthesis.
How to Synthesize Cyclopropyl Methyl Ketone Efficiently
Implementing this synthesis route requires precise adherence to the reaction parameters defined in the patent to maximize yield and purity. The process begins with the acid-mediated conversion of the lactone, followed by the alkaline cyclization, and concludes with the radical-mediated purification step. Operators must ensure strict temperature control during the chlorination phase to prevent over-chlorination or decomposition. Similarly, the purification step relies on the correct dosage of the initiator (0.1-0.5%) and the magnetic agent (4-6%) to ensure complete scavenging of DHMF. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for laboratory validation and pilot plant trials.
- Prepare 5-chloro-2-pentanone by reacting alpha-acetyl-gamma-butyrolactone with concentrated hydrochloric acid and anhydrous aluminum trichloride at 95-100°C.
- Perform intramolecular cyclization by adding the chloroketone to a 30-40% sodium hydroxide solution at 95-100°C for 3 hours to form the crude cyclopropyl methyl ketone.
- Purify the crude product by adding a radical initiator and magnetic reactive impurity removing agent at 60-80°C to polymerize and remove DHMF, followed by magnetic filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond simple chemical yield. The shift to alpha-acetyl-gamma-butyrolactone as a starting material directly addresses the volatility associated with sourcing specialized halogenated ketones. By utilizing a bulk chemical that is widely produced for other industrial applications, manufacturers can secure a more stable and resilient supply chain, mitigating the risks of raw material shortages that frequently plague the fine chemical sector. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive raw materials with cost-effective alternatives. Eliminating the need to purchase pre-synthesized 5-chloro-2-pentanone removes a significant markup from the bill of materials. Additionally, the magnetic impurity removal technique drastically simplifies the downstream purification process. By avoiding multiple high-vacuum distillation runs traditionally required to separate DHMF, the facility saves substantial energy costs and reduces solvent consumption. This leaner processing workflow translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Reliability in the chemical supply chain is often dictated by the complexity of the synthesis and the availability of precursors. This method simplifies the synthetic tree by reducing the number of discrete isolation steps and utilizing robust, commodity-grade reagents like hydrochloric acid and sodium hydroxide. The ease of sourcing these inputs means that production is less susceptible to geopolitical or logistical disruptions. Furthermore, the simplified workup procedure, which relies on phase separation and magnetic filtration rather than complex fractional distillation, reduces the mechanical load on production equipment, leading to higher uptime and more consistent output volumes for reliable agrochemical intermediate supplier networks.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process demonstrates excellent potential for green manufacturing. The use of magnetic separation minimizes the generation of liquid waste streams associated with traditional extraction and washing protocols. The ability to recover and potentially regenerate the magnetic carrier particles further aligns with sustainability goals. Scalability is enhanced by the mild reaction conditions; the cyclization occurs at atmospheric pressure and moderate temperatures (95-100 ℃), reducing the engineering requirements for high-pressure reactors. This makes the technology highly adaptable for both mid-scale pilot plants and multi-ton commercial facilities, ensuring that reducing lead time for high-purity intermediates is achievable without compromising on safety or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclopropyl methyl ketone synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of this supply source.
Q: Why is alpha-acetyl-gamma-butyrolactone preferred over 5-chloro-2-pentanone as a starting material?
A: Alpha-acetyl-gamma-butyrolactone is significantly more cost-effective and readily available than 5-chloro-2-pentanone. Furthermore, synthesizing the chloroketone in situ allows for better control over the subsequent cyclization process, reducing overall production costs.
Q: How does the new method address the issue of DHMF impurities?
A: The process utilizes a specialized reactive impurity removing agent containing nano ferroferric oxide. Under radical initiation, this agent polymerizes with DHMF (2-methyl-4,5-dihydrofuran), allowing for easy magnetic separation and resulting in purity levels exceeding 99%.
Q: What are the typical reaction conditions for the cyclization step?
A: The cyclization is conducted under mild alkaline conditions using a 30-40% sodium hydroxide solution at temperatures between 95°C and 100°C for approximately 3 hours, ensuring high conversion rates without harsh conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropyl Methyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications and advanced agrochemicals. Our team of expert chemists has extensively analyzed emerging technologies like the one described in CN114573435A to ensure our manufacturing capabilities remain at the forefront of the industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require kilogram quantities for clinical trials or metric tons for commercial launch, our facilities are equipped to deliver. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage these advanced synthetic routes for your specific project needs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how switching to our optimized supply chain can benefit your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, cost-effective, and high-purity supply of cyclopropyl methyl ketone for your next generation of products.
