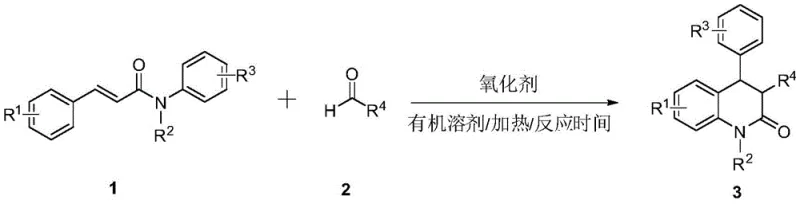
Advanced Metal-Free Cyclization for Scalable Production of Dihydroquinolinone Intermediates
The pharmaceutical industry continuously seeks robust and cost-effective synthetic routes for privileged scaffolds, and the recent disclosure in patent CN109836383B presents a significant advancement in the preparation of 3,4-dihydroquinolin-2(1H)-one compounds. This specific structural motif is ubiquitous in bioactive natural products and serves as a critical building block for numerous drug candidates, necessitating a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials. The patented methodology introduces a novel metal-free approach that utilizes cinnamamide compounds and aliphatic aldehydes as primary feedstocks, undergoing a decarbonylation addition cyclization reaction mediated by an organic peroxide oxidant. By shifting away from traditional transition metal catalysis, this innovation addresses long-standing concerns regarding metal residue limits in active pharmaceutical ingredients (APIs) while simultaneously streamlining the production workflow. The technical breakthrough lies in the ability to construct the quinoline core efficiently under relatively mild thermal conditions, offering a compelling alternative for manufacturers focused on cost reduction in API manufacturing and process safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dihydroquinolin-2(1H)-one derivatives has relied heavily on methodologies that introduce significant operational complexity and cost burdens to the supply chain. Traditional routes often necessitate the use of precious metal catalysts, such as silver salts, which not only inflate the raw material expenses but also introduce stringent purification requirements to meet regulatory standards for heavy metals. Furthermore, many existing protocols require specialized substrates that are not commercially available off-the-shelf, forcing manufacturers to engage in multi-step precursor synthesis that lowers overall atom economy. The reliance on harsh reaction conditions, including strong acids or bases and extreme temperatures, frequently leads to the formation of complex impurity profiles that are difficult to separate, thereby reducing the final isolated yield. Additionally, some prior art methods demand inert atmosphere protection and the use of stoichiometric additives like carbodiimides, which generate substantial chemical waste and complicate the post-reaction workup, making these processes less attractive for green chemistry initiatives and large-scale industrial application.
The Novel Approach
In stark contrast to these legacy techniques, the method described in CN109836383B leverages a radical-mediated cyclization strategy that completely eliminates the need for transition metal catalysts. By employing di-tert-butyl peroxide (DTBP) as a radical initiator in fluorobenzene solvent, the reaction proceeds through a clean decarbonylation pathway that directly couples cinnamamides with aliphatic aldehydes. This metal-free paradigm shift ensures that the resulting high-purity dihydroquinolinone derivatives are free from toxic metal contaminants, drastically simplifying the downstream purification process and reducing the environmental footprint of the manufacturing operation. The reaction conditions are remarkably straightforward, utilizing standard heating equipment at 130°C without the necessity for inert gas blanketing, which enhances operational safety and reduces capital expenditure on specialized reactor infrastructure. Moreover, the broad substrate scope demonstrated in the patent allows for the introduction of diverse functional groups at the 3-position of the quinoline ring, providing medicinal chemists with a versatile platform for rapid structure-activity relationship (SAR) studies and lead optimization campaigns.
Mechanistic Insights into DTBP-Mediated Radical Cyclization
The core of this innovative synthesis lies in a sophisticated radical cascade mechanism initiated by the thermal decomposition of di-tert-butyl peroxide. Upon heating to 130°C, the peroxide undergoes homolytic cleavage to generate tert-butoxy radicals, which subsequently abstract a hydrogen atom from the aliphatic aldehyde substrate to form an acyl radical species. This highly reactive intermediate then undergoes a rapid decarbonylation step, releasing carbon monoxide and generating a secondary alkyl radical that is poised for intramolecular attack. The alkyl radical adds selectively to the beta-position of the cinnamamide double bond, triggering a cascade of electron rearrangements that ultimately result in the formation of the new carbon-carbon bond and the closure of the dihydroquinoline ring system. This elegant sequence avoids the formation of stable organometallic intermediates, thereby bypassing the kinetic barriers and side reactions often associated with metal-catalyzed cross-coupling reactions, ensuring a more direct and efficient path to the target heterocyclic core.
From an impurity control perspective, the absence of metal catalysts fundamentally alters the impurity profile of the reaction mixture, favoring organic byproducts that are generally easier to separate via standard chromatographic techniques. In metal-catalyzed processes, trace amounts of catalyst can coordinate with the product or intermediates, leading to persistent impurities that require specialized scavenging resins or recrystallization steps to remove. In this radical protocol, the primary byproducts stem from the oxidant decomposition and unreacted starting materials, which can be effectively managed through optimized solvent selection and workup procedures. The use of fluorobenzene as the solvent further enhances the solubility of the radical intermediates and stabilizes the transition states, contributing to higher selectivity and reduced formation of polymeric side products. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that the critical quality attributes of the final API intermediate can be consistently maintained through precise control of reaction temperature and oxidant loading.
How to Synthesize 3,4-Dihydroquinolin-2(1H)-one Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the radical initiator and the thermal profile of the reaction to maximize yield and safety. The patent outlines a standardized procedure where cinnamamide and aliphatic aldehyde are combined with DTBP in fluorobenzene within a pressure-resistant vessel, ensuring that the volatile components remain contained during the elevated temperature phase. Operators must monitor the reaction progress via thin-layer chromatography (TLC) to determine the optimal endpoint, typically around 11 hours, before proceeding to the isolation stage. The detailed standardized synthesis steps see the guide below for specific molar ratios and purification parameters tailored to different substrate variations.
- Charge a pressure-resistant tube with cinnamamide compound, aliphatic aldehyde, di-tert-butyl peroxide (oxidant), and fluorobenzene solvent.
- Seal the tube and heat the mixture in an oil bath at 130°C with magnetic stirring for approximately 11 hours to facilitate radical cyclization.
- Upon completion, cool the reaction to room temperature and purify the crude product via flash column chromatography to isolate the target dihydroquinolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of expensive noble metal catalysts represents a direct reduction in the bill of materials, allowing for more competitive pricing structures in long-term supply agreements. Furthermore, the reliance on commodity chemicals like cinnamamides and simple aldehydes ensures a robust and diversified supply base, mitigating the risks associated with single-source dependencies on specialized reagents. The simplified workup procedure, which avoids complex metal scavenging steps, significantly shortens the production cycle time, enabling faster turnaround for custom synthesis orders and improving overall asset utilization rates within the manufacturing facility.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts such as silver salts eliminates a major cost driver traditionally associated with heterocycle synthesis. Without the need for expensive catalysts or the subsequent metal removal processes, the overall cost of goods sold (COGS) is substantially lowered. Additionally, the use of fluorobenzene as a recyclable solvent and the high atom economy of the decarbonylation pathway contribute to further economic efficiencies, making this route highly attractive for cost-sensitive generic drug production.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including various substituted cinnamamides and aliphatic aldehydes, are widely available from multiple global chemical suppliers. This abundance ensures that production schedules are not disrupted by raw material shortages, a common issue with proprietary or highly specialized reagents. The stability of the reaction conditions also means that the process can be easily transferred between different manufacturing sites without significant re-validation efforts, enhancing supply chain resilience and continuity.
- Scalability and Environmental Compliance: The process operates under relatively mild thermal conditions without the need for cryogenic cooling or high-pressure equipment, facilitating easy scale-up from laboratory to pilot and commercial production scales. The absence of heavy metals aligns perfectly with increasingly stringent environmental regulations and green chemistry principles, reducing the burden of hazardous waste disposal. This eco-friendly profile not only lowers compliance costs but also enhances the corporate sustainability metrics of the final drug product, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, aiming to clarify the operational parameters and potential applications for industry partners seeking to integrate this technology into their pipelines.
Q: What are the key advantages of this metal-free synthesis method over traditional routes?
A: Unlike traditional methods that often require expensive transition metal catalysts like silver salts or harsh reaction conditions, this patented process utilizes a metal-free radical mechanism. This eliminates the risk of heavy metal contamination in the final API, significantly simplifies the purification workflow by removing the need for metal scavengers, and reduces overall raw material costs.
Q: What represents the optimal reaction conditions for this cyclization?
A: The patent specifies that the optimal conditions involve using di-tert-butyl peroxide (DTBP) as the oxidant and fluorobenzene as the solvent. The reaction is typically conducted at a temperature of 130°C for a duration of 11 hours. These conditions ensure efficient decarbonylation and subsequent radical addition cyclization to form the 3,4-dihydroquinolin-2(1H)-one core structure with good yields.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly amenable to scale-up. It employs cheap and readily available raw materials such as cinnamides and aliphatic aldehydes. The operation is straightforward, requiring standard heating equipment without the need for inert gas protection or complex additive systems, making it ideal for producing high-purity pharmaceutical intermediates on a commercial scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydroquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free cyclization technology for the next generation of pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of peroxide-mediated reactions, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical grade materials.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your specific drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free route for your specific molecule. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in complex heterocycle synthesis can accelerate your project timelines and optimize your manufacturing costs.
