Advanced Synthesis of Trandolapril Intermediates for Commercial Scale-Up and Cost Efficiency
The pharmaceutical industry continuously seeks robust synthetic pathways for cardiovascular therapeutics, and patent CN102887853A presents a significant breakthrough in the preparation of trandolapril intermediates. This specific technology focuses on the efficient synthesis of (2S,3aR,7aS)-octahydroindole-2-carboxylate benzyl ester, a critical chiral building block for angiotensin-converting enzyme inhibitors. The innovation lies in replacing traditional, costly enzymatic or heavy-metal catalyzed routes with a streamlined chemical process starting from commodity chemicals like cyclohexene and chloramine T. By leveraging a novel aziridine ring-opening strategy followed by precise oxidative functionalization, this method addresses long-standing issues regarding impurity profiles and operational complexity. For R&D directors and procurement specialists, understanding this pathway offers a strategic advantage in securing a stable supply of high-value pharmaceutical intermediates. 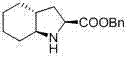
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of octahydroindole derivatives has been plagued by significant technical and economic barriers that hinder large-scale adoption. Prior art methods, such as those described in US6599318, often rely on enzymatic stereosynthesis which, while selective, incurs prohibitive costs and requires extremely harsh reaction conditions unsuitable for general industrial infrastructure. Other literature reports indicate the use of mercury nitrate, which poses severe environmental hazards and complicates waste disposal protocols, thereby increasing the overall carbon footprint and regulatory burden for manufacturers. Furthermore, routes starting from cyclohexanone often suffer from poor control over selective hydrogenation steps, leading to difficult-to-separate impurities that compromise the final optical purity of the API intermediate. These conventional approaches frequently result in low overall yields, sometimes as low as 32%, and involve cumbersome post-treatment processes like severe emulsification during extraction, which drastically reduces throughput efficiency.
The Novel Approach
In stark contrast, the methodology outlined in patent CN102887853A introduces a paradigm shift by utilizing a cyclohexene and chloramine T condensation to form a cyclohexane aziridine intermediate with yields reaching up to 70%. This foundational step is followed by a highly stereoselective reaction with allylmagnesium bromide, catalyzed by copper salts, which constructs two crucial chiral centers in a single operation with yields up to 95%. The subsequent oxidation steps utilize mild oxidizing agents like peracetic acid under controlled low-temperature conditions, ensuring minimal degradation of the sensitive indole core. This novel approach eliminates the need for expensive transition metal catalysts and toxic heavy metals, significantly simplifying the purification workflow through straightforward recrystallization and extraction techniques. 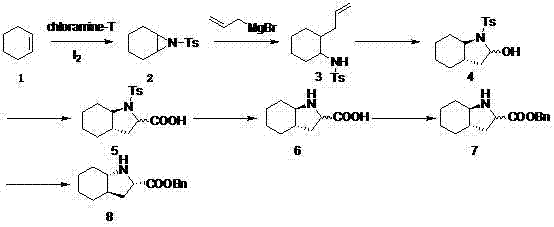
Mechanistic Insights into Cu-Catalyzed Aziridine Ring Opening
The core mechanistic advantage of this synthesis resides in the copper-catalyzed nucleophilic ring-opening of the cyclohexane aziridine by allylmagnesium bromide. This transformation is not merely a bond-forming event but a precise stereochemical control point where the trans-configuration is rigorously enforced. The copper catalyst, typically copper bromide, coordinates with the aziridine nitrogen, activating the ring towards nucleophilic attack while directing the allyl group to the specific carbon center required for the (2S,3aR,7aS) configuration. This step effectively bypasses the need for multiple protection-deprotection sequences common in older methodologies, thereby reducing the total number of unit operations and minimizing material loss. The reaction is conducted at low temperatures, specifically around -40°C, which kinetically favors the desired trans-isomer and suppresses the formation of cis-configured byproducts that are notoriously difficult to remove in downstream processing.
Following the construction of the carbon skeleton, the oxidation mechanism plays a pivotal role in establishing the carboxylic acid functionality essential for the final ester linkage. The process employs peracids or similar oxidants to convert the allyl side chain into the corresponding acid without affecting the sensitive secondary amine or the fused ring system. Impurity control is further enhanced during the deprotection and esterification stages, where specific acidic conditions are used to cleave the tosyl group cleanly. The final resolution step, utilizing tartaric acid derivatives or chiral preparative columns, ensures that any remaining racemic material is separated, delivering an optically pure product that meets the stringent specifications required for cardiovascular drug manufacturing. This multi-layered approach to impurity management ensures a consistent quality profile that is vital for regulatory approval.
How to Synthesize (2S,3aR,7aS)-Octahydroindole-2-Carboxylate Benzyl Ester Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control during the Grignard addition and the choice of solvents for crystallization. The process begins with the formation of the aziridine ring in a biphasic system using phase-transfer catalysts, which allows for easy separation of the organic product. Subsequent steps involve standard organic transformations such as oxidation, hydrolysis, and esterification, all of which are scalable using conventional reactor setups. The patent emphasizes the recyclability of solvents like ethyl acetate and toluene, which contributes to the overall economic viability of the process. Detailed standardized synthesis steps are provided below to guide process chemists in replicating these results accurately.
- React cyclohexene with chloramine T using iodine and a phase-transfer catalyst to form cyclohexane aziridine with high yield.
- Perform a stereoselective ring-opening reaction with allylmagnesium bromide and copper bromide at -40°C to establish chiral centers.
- Oxidize the intermediate using peracetic acid, followed by deprotection, esterification, and chiral resolution to obtain the final optically pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial strategic benefits by decoupling production from volatile supply chains associated with specialized enzymes or rare metal catalysts. The primary starting materials, cyclohexene and chloramine T, are commodity chemicals produced in massive volumes globally, ensuring a stable and predictable supply base that mitigates the risk of raw material shortages. This accessibility translates directly into cost stability, allowing procurement managers to negotiate long-term contracts with greater confidence. Furthermore, the elimination of toxic reagents like mercury nitrate reduces the costs associated with hazardous waste disposal and environmental compliance, which are often hidden expenses in traditional manufacturing budgets. The simplified workup procedures also mean reduced labor hours and faster batch turnover times, enhancing overall operational efficiency.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the substitution of high-cost reagents with inexpensive, bulk-available chemicals. By avoiding the use of expensive enzymatic biocatalysts or precious metal complexes, the direct material cost per kilogram of the intermediate is significantly lowered. Additionally, the high yields reported in key steps, such as the 95% yield in the Grignard reaction, minimize the amount of raw material wasted, further driving down the cost of goods sold. The ability to recycle solvents like tetrahydrofuran and dichloromethane adds another layer of cost efficiency, reducing the need for continuous fresh solvent purchases. These factors combine to create a manufacturing process that is inherently leaner and more cost-competitive than legacy methods.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust chemical transformations that are less sensitive to minor fluctuations in reaction conditions compared to biological processes. The reliance on standard organic synthesis techniques means that production can be easily transferred between different manufacturing sites without the need for specialized equipment or unique expertise. This flexibility ensures continuity of supply even if one production facility faces disruptions. Moreover, the stability of the intermediates allows for safer storage and transportation, reducing the logistical complexities often associated with unstable or temperature-sensitive biological reagents. This reliability is crucial for maintaining the production schedules of downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic runaways or gas-evolving steps that are difficult to manage in large reactors. The environmental profile is markedly improved by the exclusion of heavy metals and the implementation of solvent recovery systems, aligning with modern green chemistry principles. This compliance with environmental standards facilitates smoother regulatory approvals and reduces the risk of production halts due to environmental violations. The process generates less hazardous waste, simplifying the disposal process and lowering the environmental levy costs. Such sustainability features are increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on feasibility and performance. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their existing supply chains.
Q: What are the key advantages of this synthesis route over enzymatic methods?
A: Unlike enzymatic methods which require harsh conditions and high costs, this chemical route uses readily available starting materials like cyclohexene and chloramine T, offering simpler operation and better scalability for industrial production.
Q: How is stereochemical purity controlled in this process?
A: Stereochemical purity is achieved through a highly stereoselective Grignard reaction that constructs two chiral centers simultaneously, followed by a final resolution step using tartaric acid derivatives or chiral chromatography to ensure optical purity.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process avoids expensive transition metal catalysts and toxic reagents like mercury nitrate, utilizing standard unit operations such as crystallization and extraction that are easily adaptable to multi-ton commercial production scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3aR,7aS)-Octahydroindole-2-Carboxylate Benzyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of this intermediate in the global supply chain for cardiovascular medications. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (2S,3aR,7aS)-octahydroindole-2-carboxylate benzyl ester meets the highest industry standards. We are committed to leveraging advanced synthetic technologies like the one described in CN102887853A to deliver superior value to our partners.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our specialized expertise. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your production goals. Let us help you secure a reliable source for this essential pharmaceutical building block.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
