Advanced Organometallic Reduction Strategy for High-Purity Cardiovascular Drug Intermediates
Advanced Organometallic Reduction Strategy for High-Purity Cardiovascular Drug Intermediates
The global pharmaceutical landscape is increasingly demanding robust, scalable, and highly selective synthetic routes for complex drug intermediates, particularly in the cardiovascular sector where safety and efficacy are paramount. Patent CN108314670B introduces a transformative preparation method for (S)-2-chloro-1-(6-fluoro-1-chroman-2-yl)-ethanol, a critical chiral building block for the synthesis of Nebivolol hydrochloride, a third-generation beta-blocker. This innovation addresses the longstanding industry challenges associated with low chiral purity and difficult separation processes inherent in traditional reduction methodologies. By leveraging a sophisticated organoaluminum catalytic system within a specifically engineered mixed solvent environment, this technology enables the production of high-purity intermediates with exceptional stereochemical control. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the supply chain for antihypertensive medications, ensuring consistent quality while mitigating the risks associated with volatile exothermic reactions found in older protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-1-(6-fluoro-1-chroman-2-yl)-ethanol has relied heavily on catalytic reduction using borohydride reagents, such as potassium borohydride (KBH4), often in ethanol solvents. As illustrated in the reaction scheme below, this conventional approach is fraught with operational hazards and inefficiencies that complicate large-scale manufacturing. The reduction process is characterized by substantial heat release, necessitating rigorous temperature control typically around 0°C and the careful batch-wise addition of the reducing agent to prevent runaway reactions. Furthermore, these borohydride-mediated reductions fundamentally lack intrinsic chiral selectivity, frequently resulting in racemic mixtures or products with low optical purity. This deficiency forces manufacturers to engage in complex, costly, and yield-diminishing separation and purification steps to isolate the desired enantiomer, thereby inflating production costs and extending lead times.
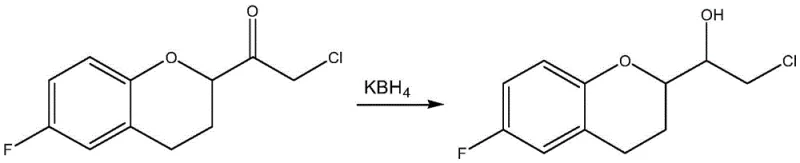
The Novel Approach
In stark contrast to the hazardous and non-selective borohydride methods, the novel approach detailed in the patent utilizes an organic aluminum salt catalyst within a dual-solvent system comprising isopropanol and an aprotic weak polar solvent. This strategic shift moves the reaction kinetics from a kinetically controlled, violent reduction to a thermodynamically controlled, mild catalytic cycle. The use of organic aluminum species allows for the formation of a specific cyclic transition state that dictates the stereochemical outcome of the hydride transfer, inherently favoring the formation of the desired (S)-enantiomer. This method eliminates the need for cryogenic conditions, operating effectively at moderate temperatures between 30°C and 80°C, which drastically simplifies reactor requirements and energy consumption. For a reliable pharmaceutical intermediate supplier, adopting this methodology translates to a streamlined process with fewer unit operations, reduced safety risks, and a final product that meets stringent chiral purity specifications without the need for extensive downstream resolution.
Mechanistic Insights into Organoaluminum-Catalyzed Asymmetric Reduction
The core of this technological breakthrough lies in the unique coordination chemistry facilitated by the organic aluminum catalyst and the specific solvent matrix. Under the influence of the isopropanol and aprotic weak polar solvent system, the organic aluminum species coordinates with the carbonyl oxygen of the substrate to form a four-coordinated aluminum complex. This coordination is pivotal as it organizes the molecular geometry into a rigid cyclic transition state intermediate, as depicted in the catalytic cycle mechanism. Within this structured environment, the hydride ion is transferred from the alkoxy ligand of the aluminum catalyst directly to the carbonyl carbon through a concerted cyclic mechanism. This intramolecular-like transfer ensures that the hydride attacks from a specific spatial direction, thereby enforcing high chiral selectivity and generating the (S)-configured alcohol with high fidelity. The avoidance of solvent effects, which would otherwise disrupt this delicate coordination sphere, is crucial; protic or strongly polar solvents would compete for coordination sites on the aluminum center, preventing the formation of the active transition state and stalling the reaction.
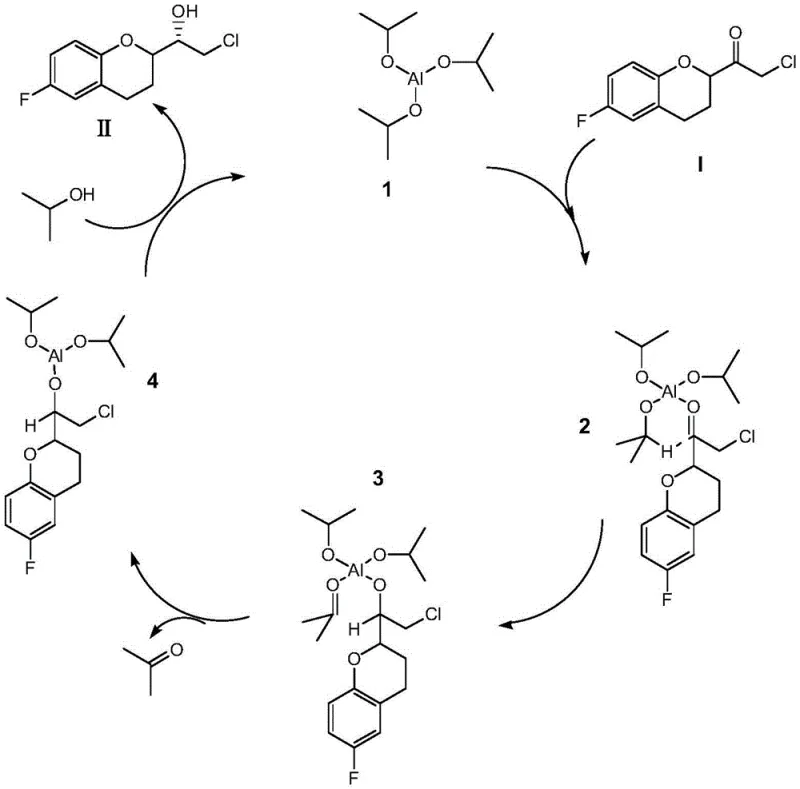
Furthermore, the mechanistic pathway ensures the regeneration of the active catalytic species, sustaining the cycle efficiently. Following the hydride transfer, an effective leaving group (a ketone byproduct) is generated, creating a three-coordination aluminum intermediate state. This intermediate is rapidly replaced by fresh isopropanol from the mixed solvent system, regenerating the corresponding catalytic organic aluminum species and allowing the conversion of the substrate to proceed continuously. This catalytic turnover is highly efficient, requiring only sub-stoichiometric amounts of the aluminum salt (molar ratios of substrate to catalyst ranging from 1:0.2 to 1:0.4). The preservation of the original chirality at the pyran ring chiral center is another critical aspect of this mechanism; the mild conditions and specific coordination mode ensure that no epimerization occurs at adjacent stereocenters. This level of mechanistic control provides R&D teams with the confidence that the process is robust, reproducible, and capable of delivering the high-purity intermediates required for modern GMP manufacturing of cardiovascular therapeutics.
How to Synthesize (S)-2-chloro-1-(6-fluoro-1-chroman-2-yl)-ethanol Efficiently
The implementation of this advanced reduction protocol requires precise adherence to solvent ratios and temperature profiles to maximize the formation of the critical cyclic transition state. The process begins by dissolving the ketone substrate in the optimized mixed solvent system, followed by the introduction of the organoaluminum catalyst under inert conditions. The reaction is then gently heated to initiate the catalytic cycle, maintaining a steady temperature to ensure complete conversion while preserving stereochemical integrity. Detailed standard operating procedures regarding specific molar ratios, solvent choices (such as dichloromethane or toluene), and workup protocols are essential for successful technology transfer and scale-up.
- Dissolve the ketone substrate in a mixed solvent system comprising isopropanol and an aprotic weak polar solvent such as dichloromethane or toluene.
- Add the organic aluminum salt catalyst, such as aluminum isopropoxide, to the reaction mixture under stirring at room temperature.
- Heat the reaction mixture to a mild temperature range of 30-80°C to facilitate the catalytic reduction and hydride transfer.
- Upon completion, perform an acidic aqueous workup to quench the reaction, separate the organic phase, and purify via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional borohydride reduction to this organoaluminum-catalyzed method offers profound strategic benefits that extend beyond mere chemical yield. The elimination of highly exothermic reaction steps significantly reduces the safety burden on manufacturing facilities, lowering insurance costs and minimizing the risk of production stoppages due to thermal incidents. Moreover, the simplified workup procedure, which avoids complex pH adjustments and extensive purification steps associated with racemic mixtures, leads to a drastic reduction in processing time and solvent consumption. This efficiency gain directly impacts the cost of goods sold (COGS), making the supply of this critical intermediate more economically viable and resilient against market fluctuations. By adopting this technology, companies can secure a more stable supply of high-quality raw materials, essential for maintaining uninterrupted production of finished cardiovascular drugs.
- Cost Reduction in Manufacturing: The adoption of organic aluminum salts as mild reducing agents eliminates the need for expensive and hazardous handling protocols associated with strong hydride donors. Since the reaction proceeds with high chiral selectivity, the costly and yield-loss-prone steps of chiral resolution or recrystallization are largely obviated. This streamlining of the synthetic route removes several unit operations, significantly reducing labor, energy, and waste disposal costs. The ability to use sub-stoichiometric amounts of catalyst further enhances the economic profile, ensuring that the overall manufacturing process is lean and cost-effective without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: The mild reaction conditions (30-80°C) and the stability of the organic aluminum catalysts contribute to a highly robust manufacturing process that is less susceptible to batch-to-batch variability. Unlike processes requiring cryogenic cooling or strict anhydrous conditions that are difficult to maintain on a multi-ton scale, this method is forgiving and easily adaptable to standard industrial reactors. This operational simplicity ensures consistent delivery schedules and reduces the likelihood of supply disruptions caused by technical failures. For global supply chains, this reliability is paramount, ensuring that downstream API manufacturers receive their key intermediates on time, every time, supporting the continuous availability of life-saving hypertension medications.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this green chemistry approach minimizes the generation of hazardous waste streams typically associated with boron byproducts. The use of recyclable solvents like toluene and isopropanol, combined with the high atom economy of the catalytic cycle, aligns with modern sustainability goals and strict environmental regulations. The process is inherently scalable, having been demonstrated to work efficiently with various substrate loads, facilitating a smooth transition from pilot plant to commercial production. This scalability ensures that as demand for Nebivolol grows, the supply of its precursor can be ramped up rapidly without the need for specialized, capital-intensive infrastructure, providing a future-proof solution for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this organoaluminum-catalyzed reduction process. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing clarity on solvent selection, catalyst loading, and stereochemical outcomes. Understanding these nuances is critical for process chemists aiming to integrate this methodology into their existing manufacturing workflows.
Q: Why is the mixed solvent system critical for chiral selectivity in this reduction?
A: The combination of isopropanol and an aprotic weak polar solvent (like dichloromethane or toluene) is essential to avoid solvent effects that disrupt the formation of the four-coordinated aluminum cyclic transition state. Protic or strongly polar solvents prevent this specific intermediate from forming, leading to poor reaction progress and low stereoselectivity.
Q: What advantages does organoaluminum catalysis offer over traditional borohydride reduction?
A: Unlike potassium borohydride reductions which are highly exothermic, require batch additions, and often yield racemic mixtures with difficult separations, the organoaluminum method operates under mild thermodynamic control. This results in significantly higher chiral purity (over 95% ee), simplified operation without strict temperature controls near 0°C, and easier downstream purification.
Q: Which organic aluminum salts are suitable for this transformation?
A: The process demonstrates versatility with various aluminum alkoxides and hydrides, including aluminum ethoxide, aluminum isopropoxide, aluminum tert-butoxide, and lithium aluminum tri-tert-butoxide hydride. These catalysts effectively coordinate with the carbonyl oxygen to drive the asymmetric hydride transfer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-chloro-1-(6-fluoro-1-chroman-2-yl)-ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development and commercialization of next-generation cardiovascular therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of (S)-2-chloro-1-(6-fluoro-1-chroman-2-yl)-ethanol adheres to the highest international standards. Our facility is equipped to handle the specific solvent systems and catalytic requirements of this advanced reduction process, providing a secure and compliant source for your supply chain.
We invite pharmaceutical partners to collaborate with us to leverage this cutting-edge synthetic technology for their Nebivolol programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the superior performance of our organoaluminum-catalyzed intermediates against your current supply benchmarks. Together, we can drive down costs and enhance the reliability of the global supply for essential hypertension treatments.
