Advanced Metal-Free Synthesis of Fluoroalkyl Furyl Bisphosphine Oxides for Commercial Scale-Up
Advanced Metal-Free Synthesis of Fluoroalkyl Furyl Bisphosphine Oxides for Commercial Scale-Up
The rapid evolution of medicinal chemistry demands increasingly sophisticated building blocks, particularly those incorporating fluorine motifs which enhance metabolic stability and binding affinity. Patent CN111574564A introduces a groundbreaking methodology for the synthesis of fluoroalkyl-substituted furyl bisphosphine oxide compounds, addressing critical bottlenecks in the production of high-value pharmaceutical intermediates. This technology leverages a novel tandem cyclization strategy that constructs the furan core and installs phosphine oxide functionalities simultaneously, bypassing the need for pre-functionalized heterocyclic starting materials. By utilizing readily available polyfluoroalkyl substituted peroxy compounds and organic phosphine oxides, this approach offers a streamlined pathway to complex molecular architectures that are essential for modern drug discovery and material science applications.
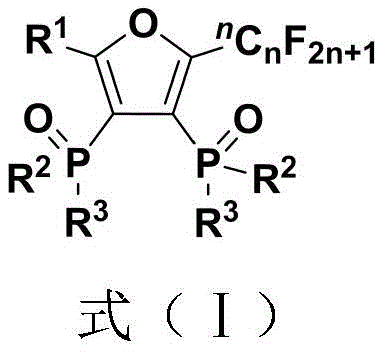
For R&D Directors seeking reliable pharmaceutical intermediate suppliers, the structural versatility offered by this patent is paramount. The general formula encompasses a vast array of substituents, allowing for the precise tuning of electronic and steric properties required for specific biological targets. The ability to introduce diverse R groups, ranging from simple alkyl chains to complex steroid-derived skeletons, positions this synthetic route as a powerful tool for generating focused libraries of potential drug candidates. Furthermore, the inherent stability of the phosphine oxide moiety provides a robust handle for subsequent derivatization or direct application as a ligand in catalytic systems, thereby expanding the utility of these compounds beyond mere intermediates into functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aromatic carbon-phosphorus bonds has relied heavily on transition metal-catalyzed cross-coupling reactions, such as those employing palladium or nickel complexes. While effective in certain contexts, these classical methods suffer from significant drawbacks that hinder their widespread adoption in cost-sensitive manufacturing environments. The requirement for expensive noble metal catalysts not only inflates raw material costs but also necessitates rigorous downstream processing to remove trace metal residues, which is a critical regulatory hurdle in pharmaceutical production. Additionally, traditional approaches often demand harsh reaction conditions, including high temperatures and inert atmospheres, which limit functional group tolerance and complicate process safety. The multi-step nature of conventional syntheses, typically requiring the pre-formation of the heterocyclic ring followed by separate phosphorylation steps, further reduces overall atom economy and operational efficiency.
The Novel Approach
In stark contrast, the methodology disclosed in CN111574564A represents a paradigm shift towards green and economical chemistry by eliminating the dependence on transition metals entirely. This innovative route utilizes a base-promoted tandem reaction that achieves ring closure and phosphorylation in a single operational step, dramatically simplifying the synthetic workflow. By operating under mild thermal conditions (50-90°C) and in the presence of air, the process removes the need for specialized equipment associated with anaerobic techniques, thereby lowering capital expenditure and operational complexity. The use of inexpensive alkali promoters and common polar aprotic solvents ensures that the cost of goods sold (COGS) is significantly optimized, making this an attractive option for cost reduction in pharmaceutical intermediate manufacturing. This one-pot strategy not only enhances step economy but also minimizes waste generation, aligning perfectly with modern sustainability goals in the chemical industry.
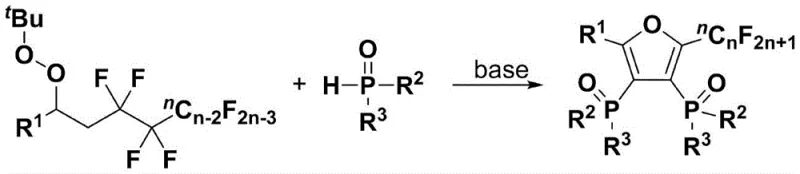
Mechanistic Insights into Base-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in its ability to orchestrate multiple bond-forming and bond-breaking events in a concerted fashion. The reaction initiates with the deprotonation of the organic phosphine oxide by the alkali promoter, generating a nucleophilic phosphorus species. This activated nucleophile then attacks the electron-deficient carbon centers of the polyfluoroalkyl substituted peroxy compound, triggering a cascade of intramolecular cyclizations. A key feature of this mechanism is the successive elimination of four molecules of hydrogen fluoride (HF), which drives the formation of the stable furan ring system while concurrently establishing the carbon-phosphorus bonds. This defluorinative cyclization is highly selective, ensuring that the fluorine atoms remaining on the alkyl chain are preserved, which is crucial for maintaining the desired lipophilicity and metabolic profile of the final product.
From an impurity control perspective, the metal-free nature of this catalytic cycle offers distinct advantages for ensuring high product purity. Without the introduction of transition metals, the risk of metal-catalyzed side reactions, such as homocoupling or over-reduction, is effectively mitigated. The reaction proceeds through well-defined ionic intermediates, which allows for predictable byproduct profiles that are easily separated during the workup phase. The use of column chromatography with standard silica gel stationary phases and ethyl acetate/petroleum ether mobile phases facilitates the isolation of the target bisphosphine oxides with purity levels exceeding 98%, as demonstrated in the patent examples. This high level of chemical fidelity is essential for high-purity pharmaceutical intermediates where even trace impurities can compromise the safety and efficacy of the final drug substance.
How to Synthesize Fluoroalkyl-Substituted Furyl Bisphosphine Oxide Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and selectivity. The process begins by combining the polyfluoroalkyl substituted peroxy compound and the organic phosphine oxide in a molar ratio that favors the formation of the bis-substituted product, typically ranging from 1:1 to 1:3. The choice of solvent plays a critical role, with dimethyl sulfoxide (DMSO) and N,N-dimethylformamide (DMF) proving to be optimal for solubilizing the reactants and stabilizing the ionic transition states. The addition of the alkali promoter, such as cesium carbonate or triethylenediamine, must be carefully managed to ensure complete deprotonation without inducing decomposition of the sensitive peroxy functionality. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Mix polyfluoroalkyl substituted peroxy compounds with organic phosphine oxide compounds in a reaction vessel.
- Add an alkali promoter such as cesium carbonate or triethylenediamine and a solvent like DMSO or DMF.
- Stir the mixture at 50-90°C under air atmosphere for 1-24 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free synthesis protocol offers substantial strategic benefits that extend beyond simple unit price reductions. The elimination of precious metal catalysts removes a major source of supply chain volatility, as the pricing of palladium and other noble metals is subject to significant geopolitical and market fluctuations. By relying on abundant and inexpensive base metals and organic reagents, manufacturers can secure a more stable and predictable cost structure for their raw materials. Furthermore, the simplified purification process reduces the consumption of specialized scavenging resins and solvents, leading to lower operational expenditures and a smaller environmental footprint. This robustness translates directly into enhanced supply chain reliability, ensuring consistent delivery schedules for critical fine chemical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of expensive transition metal catalysts from the bill of materials. Traditional cross-coupling methods often require catalyst loadings that, while low in molar percentage, represent a significant cost driver due to the high price of the metals themselves. By replacing these with cheap alkali bases like cesium carbonate or organic amines, the direct material cost is drastically lowered. Additionally, the one-pot nature of the reaction consolidates two synthetic steps into one, reducing labor costs, energy consumption for heating and cooling, and the overall time required for production. This consolidation also minimizes the loss of material between steps, improving the overall mass balance and yield of the process.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized organometallic reagents significantly de-risks the supply chain. Polyfluoroalkyl peroxy compounds and simple phosphine oxides are widely available from multiple global suppliers, preventing single-source bottlenecks. The reaction's tolerance to air and moisture further simplifies logistics, as it does not require the stringent handling protocols associated with air-sensitive catalysts. This ease of handling allows for more flexible manufacturing schedules and reduces the likelihood of production delays caused by equipment failures or operator errors related to inert atmosphere maintenance. Consequently, lead times for commercial scale-up of complex pharmaceutical intermediates can be significantly shortened.
- Scalability and Environmental Compliance: Scaling this process to multi-ton quantities is straightforward due to the mild reaction conditions and the absence of exothermic risks often associated with transition metal catalysis. The use of DMSO or DMF as solvents is well-established in industrial settings, with mature recovery and recycling protocols already in place. From an environmental standpoint, the avoidance of heavy metals simplifies waste treatment and disposal, helping facilities meet increasingly stringent regulatory standards for effluent discharge. The high atom economy of the tandem reaction means less chemical waste is generated per kilogram of product, supporting corporate sustainability initiatives and reducing the costs associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or new product development pipelines. The responses cover aspects ranging from reaction optimization to product application, ensuring a comprehensive understanding of the value proposition.
Q: What are the key advantages of this metal-free synthesis method?
A: The method eliminates the need for expensive transition metal catalysts like palladium, significantly reducing raw material costs and simplifying purification by avoiding heavy metal removal steps.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction uses mild temperatures (50-90°C), common solvents like DMSO, and operates under air atmosphere, making it highly suitable for large-scale commercial manufacturing.
Q: What is the substrate scope for this reaction?
A: The process demonstrates excellent functional group tolerance, accommodating various aryl, alkyl, and heteroaryl groups, including complex bio-active molecular skeletons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl-Substituted Furyl Bisphosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the next generation of pharmaceutical and material science applications. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and quality of every batch, guaranteeing that our high-purity pharmaceutical intermediates meet the exacting standards of the global market.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this efficient synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed sourcing decisions. Contact us today to discuss how we can support your project with reliable supply and technical excellence, ensuring your journey from discovery to commercialization is seamless and successful.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
