Advanced Clean Synthesis of UV-531 for High-Performance Polymer Additive Manufacturing
Advanced Clean Synthesis of UV-531 for High-Performance Polymer Additive Manufacturing
The global demand for high-performance ultraviolet stabilizers continues to surge as the polymer industry seeks materials with enhanced durability and weatherability. At the forefront of this chemical evolution is the synthesis of UV-531, also known chemically as 2-hydroxy-4-n-octyloxybenzophenone or Octabenzone, a critical additive widely utilized in polyethylene, polypropylene, polystyrene, and ABS resin formulations. A pivotal advancement in this domain is documented in patent CN103508870A, which discloses a novel, clean synthetic method that fundamentally restructures the production workflow to maximize yield while minimizing environmental footprint. This technology represents a significant leap forward for any entity seeking a reliable UV-531 supplier, as it addresses long-standing inefficiencies associated with traditional manufacturing routes. By integrating a two-step approach that leverages in-situ chlorination and solvent-free phase transfer catalysis, the process achieves exceptional purity levels suitable for the most stringent commercial scale-up of complex polymer additives. The following analysis dissects the technical nuances of this patent, providing R&D directors and procurement specialists with a comprehensive understanding of its operational viability and economic potential.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of UV-531 has been plagued by significant technical and economic bottlenecks that hinder optimal cost reduction in polymer additive manufacturing. Traditional methodologies generally fall into two categories: single-stage reactions and two-stage processes reliant on organic solvents. The single-stage method, while operationally simple, suffers from notoriously low productivity and poor product quality due to the difficulty in controlling side reactions when UV-0 and alkylating agents react simultaneously in one vessel. Furthermore, the two-stage approaches that have been previously adopted often necessitate the use of expensive alkylating agents like n-octyl bromide or require substantial quantities of organic solvents such as higher ketones to facilitate the reaction between water-insoluble reactants. These solvent-based systems introduce severe downstream complications, including high energy consumption for solvent recovery, complex post-treatment procedures, and significant environmental hazards associated with volatile organic compound (VOC) emissions. Additionally, older chlorination techniques frequently relied on the direct feeding of gaseous hydrochloric acid, posing serious safety risks regarding acid mist exposure and requiring specialized corrosion-resistant equipment that drives up capital expenditure.
The Novel Approach
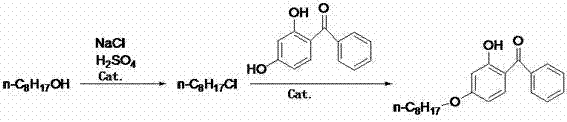
In stark contrast to these legacy systems, the methodology outlined in CN103508870A introduces a streamlined, eco-friendly pathway that effectively circumvents the aforementioned deficiencies through intelligent process engineering. The core innovation lies in the replacement of volatile hydrochloric acid with a safer combination of high-boiling sulfuric acid and sodium chloride for the preparation of the intermediate 1-chlorooctane. This modification not only enhances operator safety by eliminating acid mist but also allows for the recycling of salt byproducts from the subsequent washing stages, creating a closed-loop system that drastically improves raw material availability. Moreover, the condensation step is revolutionized by the application of phase transfer catalysts (PTC), which enable the reaction between 1-chlorooctane and 2,4-dihydroxybenzophenone to proceed smoothly without the need for any organic solvent. This solvent-free condition simplifies the workup procedure to a mere washing and separation step, thereby reducing energy consumption and eliminating the need for complex distillation units. The result is a robust, scalable process that delivers yields exceeding 90% with superior product quality, making it an ideal candidate for reducing lead time for high-purity UV-531 batches in a commercial setting.
Mechanistic Insights into Phase Transfer Catalyzed Etherification
The chemical elegance of this synthesis is rooted in the precise orchestration of nucleophilic substitution reactions facilitated by specific catalytic systems. In the first stage, the conversion of n-octanol to 1-chlorooctane is driven by the generation of hydrogen chloride in situ from the reaction between concentrated sulfuric acid (98%) and sodium chloride. The presence of zinc chloride acts as a potent Lewis acid catalyst, coordinating with the hydroxyl group of the alcohol to enhance its leaving group ability, thereby promoting the nucleophilic attack by the chloride ion at temperatures ranging from 110°C to 160°C. This ensures a high conversion rate of the alcohol while minimizing the formation of ether byproducts that typically plague acid-catalyzed dehydrations. The second stage involves the etherification of 2,4-dihydroxybenzophenone, where the phenolic hydroxyl group acts as the nucleophile. Under traditional conditions, the disparity in polarity between the organic halide and the inorganic base would prevent efficient reaction; however, the introduction of quaternary ammonium salts or polyethylene glycol-type ionic liquids bridges this gap. These phase transfer catalysts transport the phenoxide anion into the organic phase or bring the organic halide into the aqueous interface, significantly lowering the activation energy and allowing the condensation to proceed rapidly at 160°C to 180°C without external solvents.
From an impurity control perspective, this mechanism offers distinct advantages that are critical for R&D teams focused on specification compliance. The absence of organic solvents eliminates the risk of solvent-derived impurities or azeotropic complications during purification. Furthermore, the specific molar ratios employed—such as maintaining a slight excess of 1-chlorooctane (1.05 to 1.3 equivalents relative to UV-0)—ensure that the more valuable 2,4-dihydroxybenzophenone is fully consumed, preventing the presence of unreacted starting material in the final crystal lattice. The washing steps are designed to remove water-soluble byproducts like sodium chloride and sodium sulfate, while the final recrystallization from ethanol effectively removes trace colored impurities and residual catalysts. This rigorous control over the reaction environment ensures that the final product meets the stringent purity specifications required for food-contact plastics and high-end automotive coatings, validating the process as a source of high-purity UV-531 for discerning applications.
How to Synthesize UV-531 Efficiently
Implementing this synthesis route requires careful attention to thermal profiles and stoichiometric precision to replicate the high yields reported in the patent literature. The process is divided into three distinct operational phases: the preparation of the alkyl halide intermediate, the solvent-free condensation reaction, and the final purification via recrystallization. Operators must ensure that the chlorination step reaches complete conversion of n-octanol before proceeding, as residual alcohol can interfere with the subsequent etherification. Similarly, the selection of the phase transfer catalyst is crucial, with options ranging from cetyl trimethylammonium bromide to specialized ionic liquids, each offering varying degrees of efficiency depending on the specific batch scale. The following guide outlines the standardized protocol derived from the patent examples, serving as a foundational reference for process engineers looking to adopt this technology.
- Prepare 1-chlorooctane by reacting n-octanol with sodium chloride and concentrated sulfuric acid in the presence of a zinc chloride catalyst at 110-160°C.
- Perform condensation by mixing 1-chlorooctane with 2,4-dihydroxybenzophenone, an alkaline catalyst, and a phase transfer catalyst, heating to 160-180°C without organic solvents.
- Purify the crude product through washing, activated carbon decolorization, and recrystallization using ethanol to obtain high-purity UV-531 crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling strategic benefits that extend far beyond simple chemical yield improvements. The elimination of organic solvents in the condensation step represents a massive reduction in raw material procurement costs, as there is no longer a need to purchase, store, and recover large volumes of volatile ketones or other organic media. This simplification of the bill of materials directly translates to substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing structures in the global market. Furthermore, the ability to recycle salt byproducts from the wastewater back into the initial chlorination step creates a circular economy within the plant, drastically reducing the consumption of fresh sodium chloride and sulfuric acid while minimizing waste disposal fees. These efficiencies collectively contribute to a leaner, more agile production model that is less susceptible to fluctuations in raw material commodity prices.
- Cost Reduction in Manufacturing: The economic architecture of this process is fundamentally optimized for margin expansion through the removal of high-cost inputs and energy-intensive unit operations. By replacing expensive n-octyl bromide with inexpensive n-octanol and sodium chloride, the direct material cost per kilogram of product is significantly lowered. Additionally, the solvent-free nature of the main condensation reaction removes the need for energy-heavy distillation columns used for solvent recovery, resulting in drastically simplified utility consumption profiles. The qualitative impact on the bottom line is profound, as the process transforms a multi-step, solvent-reliant operation into a streamlined sequence that maximizes asset utilization and minimizes variable operating expenses without compromising on output quality.
- Enhanced Supply Chain Reliability: From a logistics and sourcing perspective, this method relies on commodity chemicals that are readily available in the global marketplace, reducing the risk of supply disruptions associated with specialty reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and catalyst loadings, ensures consistent batch-to-batch performance even when scaling up from pilot to full commercial production. This reliability is essential for maintaining continuous supply lines to downstream polymer manufacturers who depend on just-in-time delivery schedules. By mitigating the technical risks associated with complex solvent handling and hazardous gas feeding, the supply chain becomes more resilient, ensuring that delivery commitments are met consistently regardless of external market pressures.
- Scalability and Environmental Compliance: As regulatory frameworks regarding industrial emissions become increasingly stringent, the clean nature of this synthesis provides a significant compliance advantage. The process generates minimal hazardous waste, with the primary byproducts being recyclable salts and water, aligning perfectly with modern green chemistry principles. This environmental stewardship reduces the regulatory burden on the facility and lowers the long-term liability associated with waste treatment. Moreover, the simplicity of the equipment requirements—standard reactors without the need for specialized solvent recovery systems—facilitates rapid scale-up, allowing manufacturers to respond quickly to surges in market demand for UV stabilizers while maintaining a sustainable operational footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this clean synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, aiming to clarify the operational realities for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing production lines or for assessing the quality credentials of suppliers utilizing this method. The answers provided reflect a commitment to transparency and technical accuracy, ensuring that all stakeholders have a clear picture of the capabilities and limitations of the technology.
Q: What are the primary advantages of using phase transfer catalysts in UV-531 production?
A: The use of phase transfer catalysts eliminates the need for volatile organic solvents during the condensation step, significantly reducing environmental pollution and simplifying the post-reaction separation process while maintaining high reaction rates.
Q: How does this method improve safety compared to traditional chlorination processes?
A: By utilizing high-boiling sulfuric acid and sodium chloride to generate hydrogen chloride in situ, the process avoids the direct handling and feeding of volatile hydrochloric acid gas, thereby minimizing the risk of acid mist exposure to operators.
Q: Can the byproducts from this synthesis be recycled to reduce costs?
A: Yes, the wastewater generated during the washing phase contains recoverable salts like sodium sulfate and sodium chloride, which can be recycled back into the initial chlorination step, enhancing overall raw material utilization efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable UV-531 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN103508870A is key to delivering superior value to our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this clean technology are fully realized in practical, large-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of UV-531 we produce meets the exacting standards required for high-performance polymer applications. We are dedicated to leveraging our technical expertise to optimize this solvent-free process, delivering a product that combines exceptional UV protection capabilities with a sustainable manufacturing pedigree.
We invite procurement leaders and technical directors to engage with us to explore how this innovative synthesis can benefit your specific supply chain requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic advantages of switching to our high-efficiency production model. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume needs. Let us demonstrate how our commitment to clean chemistry and operational excellence can drive value and reliability in your polymer additive supply chain.
