Advanced Synthesis of Single Iodo Perfluoro Alkanes for Commercial Scale Production
Advanced Synthesis of Single Iodo Perfluoro Alkanes for Commercial Scale Production
The global demand for high-performance fluorinated materials continues to surge, driven by their unique thermal stability and chemical inertness in pharmaceutical and electronic applications. A pivotal advancement in this sector is detailed in patent CN101456789B, which discloses a robust method for preparing single iodo perfluoro alkanes. This technology represents a significant departure from traditional telomerization processes, offering a streamlined pathway to critical intermediates like perfluoroethyl iodide (PFEI). By leveraging a specific combination of iodine, chlorine, and anhydrous hydrofluoric acid under Lewis acid catalysis, the process achieves exceptional purity levels exceeding 99 percent. For R&D directors and procurement strategists, understanding this synthetic breakthrough is essential for securing a reliable supply chain of high-purity fluorine materials. The method not only simplifies the operational workflow but also drastically reduces the dependency on scarce and hazardous reagents, positioning it as a cornerstone for next-generation fluorochemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of single iodo perfluoro alkanes has been plagued by significant economic and technical hurdles. Traditional routes often rely on the telomerization of micromolecular single iodo perfluoro alkanes with tetrafluoroethylene, a process that is inherently difficult to control and often results in broad molecular weight distributions. Furthermore, established addition reactions frequently utilize iodine pentafluoride (IF5) or iodine chloride (ICl) as key reagents. The preparation of iodine pentafluoride is notoriously complex, requiring specialized equipment and posing severe safety risks due to its high reactivity. Similarly, iodine chloride is expensive and difficult to source in bulk quantities, creating a bottleneck for large-scale manufacturing. These legacy methods also suffer from lower yields and generate substantial waste streams, making them increasingly unsustainable in a modern regulatory environment focused on green chemistry and cost efficiency.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a highly efficient direct addition strategy that circumvents the need for pre-functionalized iodine reagents. By utilizing elemental iodine and chlorine or sulfuryl chloride in the presence of anhydrous hydrofluoric acid, the reaction proceeds smoothly to form the target carbon-iodine bond. This approach eliminates the costly upstream synthesis of IF5 or ICl, directly translating to substantial raw material savings. The reaction is conducted in an oxygen-free atmosphere within a standard autoclave, heating the mixture to temperatures between 130°C and 190°C. This simplicity allows for precise control over the reaction kinetics, minimizing side reactions and maximizing the formation of the desired mono-iodo species. The versatility of this system is further enhanced by its compatibility with various perfluoroolefins, enabling the production of a wide range of chain lengths from C4 to C12.
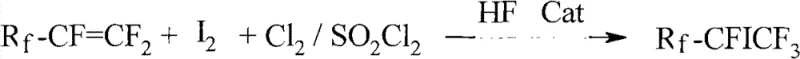
Mechanistic Insights into Lewis Acid-Catalyzed Halogenation
The core of this technological breakthrough lies in the sophisticated interplay between the Lewis acid catalyst and the reaction medium. Anhydrous hydrofluoric acid serves a dual role in this system, acting both as a reactant providing the fluorine source and as a polar solvent that stabilizes ionic intermediates. The Lewis acid catalyst, which can be selected from halogenated boron, niobium, tantalum, aluminum, iron, or tin halides, activates the halogen molecules (chlorine or sulfuryl chloride) to generate electrophilic species. These activated species then attack the electron-deficient double bond of the perfluoroolefin. The presence of iodine facilitates the formation of the carbon-iodine bond through a mechanism that likely involves radical or ionic addition pathways, depending on the specific catalyst and temperature profile employed. Strict exclusion of oxygen is critical, as oxidative degradation of the iodine species or the perfluoroolefin would lead to the formation of unwanted byproducts and reduced yields.
Impurity control is paramount in the synthesis of pharmaceutical and electronic grade intermediates, and this process excels in minimizing side products. One of the primary challenges in fluoroalkane synthesis is the formation of di-iodo byproducts or telomers, which are difficult to separate and degrade product performance. By optimizing the molar ratios of reactants—specifically maintaining a chlorine to perfluoroolefin ratio of 1.0 to 1.5—the process selectively favors the formation of the mono-iodo product. The subsequent workup procedure, involving neutralization with aqueous sodium hydroxide and drying with calcium chloride, effectively removes acidic residues and unreacted halogens. Finally, low-temperature distillation at cryogenic conditions (e.g., -78°C cold wells) ensures the isolation of the target compound with a purity greater than 99 percent, as confirmed by 19F NMR spectroscopy. This rigorous purification protocol ensures that the final material meets the stringent specifications required for high-value applications.
How to Synthesize Perfluoroethyl Iodide Efficiently
Implementing this synthesis route requires careful attention to reactor preparation and parameter control to ensure safety and reproducibility. The process begins with the thorough deoxygenation of the reaction vessel, typically a stainless steel autoclave equipped with induction stirring, to prevent oxidative side reactions. Precise weighing of the iodine, chlorine source, and catalyst is followed by the introduction of anhydrous hydrofluoric acid and the perfluoroolefin substrate. The reaction mixture is then subjected to a controlled heating ramp, reaching the target temperature of 130-190°C over the course of one hour. Maintaining this temperature for a duration of 3 to 7 hours under autogenous pressure allows the reaction to reach completion. Detailed standardized operating procedures regarding specific catalyst loading and pressure management are critical for scaling this technology from the laboratory to commercial production.
- Charge a deoxygenated autoclave with iodine, chlorine or sulfuryl chloride, anhydrous hydrofluoric acid, Lewis acid catalyst, and perfluoroolefin.
- Heat the mixture to 130-190°C over one hour and maintain temperature for 3-7 hours under 0.1-20MPa pressure.
- Cool the product, absorb gas phase with sodium hydroxide, dry with calcium chloride, and purify via low-temperature distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this novel synthesis method offers compelling strategic advantages that extend beyond simple technical metrics. The shift away from exotic reagents like iodine pentafluoride to commodity chemicals such as elemental iodine and chlorine fundamentally alters the cost structure of production. This change mitigates the risk of supply disruptions associated with niche chemical suppliers and leverages the robust global availability of basic halogens. Furthermore, the simplified operational workflow reduces the burden on facility maintenance and safety compliance teams, as the handling of highly unstable intermediates is eliminated. These factors combine to create a more resilient and cost-effective supply chain for critical fluorine intermediates.
- Cost Reduction in Manufacturing: The economic impact of replacing expensive iodine pentafluoride with low-cost iodine and chlorine cannot be overstated. By removing the need for complex upstream synthesis of halogenating agents, the overall variable cost of goods sold is significantly reduced. Additionally, the high yield of the reaction, consistently exceeding 92 percent, minimizes raw material waste and maximizes throughput per batch. The elimination of transition metal catalysts in favor of recyclable or less toxic Lewis acids further contributes to long-term cost savings by reducing waste disposal expenses and catalyst replenishment frequency.
- Enhanced Supply Chain Reliability: Relying on widely available commodity chemicals like iodine and sulfur dioxide derivatives ensures a stable supply base that is less susceptible to market volatility. Unlike specialized reagents that may have single-source suppliers or long lead times, the raw materials for this process are produced by multiple global manufacturers. This diversity in sourcing options empowers procurement teams to negotiate better terms and secure continuous inventory flow. Moreover, the robustness of the reaction conditions allows for flexible production scheduling, enabling manufacturers to respond rapidly to fluctuations in downstream demand without compromising product quality.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in stainless steel autoclaves which are standard equipment in the fine chemical industry. The absence of heavy metal contaminants simplifies the purification process and ensures the final product meets strict environmental and regulatory standards for pharmaceutical and electronic use. Waste streams are primarily composed of manageable salts and acids that can be neutralized and treated using standard effluent protocols. This alignment with green chemistry principles reduces the environmental footprint of the manufacturing site and lowers the regulatory burden associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs. The answers highlight the balance between operational simplicity and high-performance output that defines this technology.
Q: Why is this method superior to using Iodine Pentafluoride?
A: Conventional methods often rely on Iodine Pentafluoride (IF5) or Iodine Chloride (ICl), which are expensive, difficult to prepare, and complicate the reaction process. This novel method utilizes inexpensive and readily available elemental iodine and chlorine/sulfuryl chloride, significantly reducing raw material costs and operational complexity while maintaining high yields above 92%.
Q: What catalysts are suitable for this reaction?
A: The process employs a variety of Lewis acid catalysts to drive the reaction efficiently. Suitable catalysts include halogenated boron, niobium, tantalum, aluminum, iron, and tin halides. These catalysts can be used individually or in combination, typically at a molar ratio of 0.02-0.2 relative to the perfluoroolefin, ensuring optimal conversion rates.
Q: What purity levels can be achieved with this synthesis route?
A: The patented method is designed to produce high-purity single iodo perfluoro alkanes suitable for sensitive applications. Through strict control of reaction conditions and effective purification steps such as low-temperature distillation, the final product consistently achieves a purity greater than 99%, with yields reaching 92% or higher.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perfluoroethyl Iodide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity fluorine materials play in the development of advanced pharmaceuticals and electronic chemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the efficiencies of the synthesis method described in CN101456789B, we can offer our partners a competitive advantage through consistent quality and optimized manufacturing processes.
We invite you to collaborate with us to explore how this advanced technology can enhance your product portfolio and reduce your overall manufacturing costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can build a sustainable and efficient supply chain for the next generation of fluorinated innovations.
