Scalable Enzymatic Resolution of Chiral Tetrahydrofuran Derivatives for Commercial Production
Scalable Enzymatic Resolution of Chiral Tetrahydrofuran Derivatives for Commercial Production
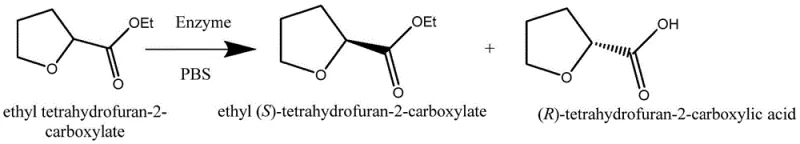
The pharmaceutical and fine chemical industries are constantly seeking sustainable and efficient pathways to access high-value chiral building blocks, and the technology disclosed in patent CN107828831B represents a significant leap forward in this domain. This patent details a novel method for preparing chiral 2-tetrahydrofurfuryl acid derivatives through protease resolution, offering a robust alternative to traditional chemical synthesis. By leveraging the specificity of alkaline proteases, this process achieves high optical purity under mild reaction conditions, specifically within a temperature range of 0-20 ℃ and at a neutral to slightly alkaline pH of 8. For R&D directors and process chemists, this biocatalytic approach solves critical pain points associated with energy consumption and environmental pollution, positioning it as a superior choice for the manufacturing of complex pharmaceutical intermediates. The ability to simultaneously obtain both (S)-ethyl ester and (R)-acid configurations from a single racemic starting material drastically improves the overall atom economy of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure tetrahydrofuran-2-carboxylic acid derivatives has relied heavily on chemical resolution methods involving chiral resolving agents and repeated recrystallization steps. These conventional techniques are fraught with inefficiencies, including the requirement for massive quantities of organic solvents and the generation of substantial chemical waste, which poses severe environmental challenges and increases disposal costs. Furthermore, traditional chemical resolution often suffers from a theoretical maximum yield of 50% for the desired enantiomer, as the unwanted isomer is frequently discarded or requires complex racemization steps to be reused. The operational complexity is another major drawback, often necessitating more than five crystallization operations to achieve acceptable optical purity, which extends production cycles and ties up valuable reactor capacity. For procurement managers, these factors translate into volatile supply chains and inflated raw material costs due to the low utilization rate of the starting racemate.
The Novel Approach
In stark contrast, the enzymatic resolution method described in the patent utilizes a biphasic system comprising a phosphate buffer and toluene, catalyzed by alkaline protease, to selectively hydrolyze the racemic ethyl ester. This biological catalysis operates under exceptionally mild conditions, typically between 0-20 ℃, which minimizes energy expenditure and eliminates the risk of thermal degradation of sensitive functional groups. The process is remarkably streamlined, requiring simple filtration to recover the enzyme and straightforward pH adjustments to separate the product phases, thereby removing the need for complex chromatographic separations. By achieving high conversion rates and excellent stereoselectivity, this novel approach ensures that both enantiomeric forms—the (S)-ester and the (R)-acid—are recovered in high purity, effectively doubling the utility of the raw material compared to single-isomer isolation methods. This shift from harsh chemical reagents to biocatalysts aligns perfectly with modern green chemistry principles, offering a safer and more sustainable manufacturing route.
Mechanistic Insights into Alkaline Protease-Catalyzed Hydrolysis
The core of this technological breakthrough lies in the precise stereoselectivity of the alkaline protease, which acts as a chiral catalyst to differentiate between the enantiomers of the racemic ethyl 2-tetrahydrofurfuryl acid ester. In the reaction mechanism, the enzyme's active site preferentially binds to one specific enantiomer, facilitating the hydrolysis of the ester bond to form the corresponding carboxylic acid, while leaving the other enantiomer largely untouched in its ester form. This kinetic resolution is conducted in a phosphate buffer solution maintained at pH 8, which is critical for maintaining the ionization state of the enzyme and ensuring optimal catalytic activity. The addition of toluene creates a biphasic system that not only solubilizes the organic substrate but also facilitates the subsequent separation of products, as the unreacted ester partitions into the organic phase while the hydrolyzed acid remains in the aqueous phase as a salt. This elegant mechanistic design prevents side reactions and ensures that the reaction proceeds with high fidelity, yielding products with minimal impurities.
Controlling impurities in this system is inherently managed by the specificity of the biocatalyst and the mild reaction parameters. Unlike chemical catalysts that might promote non-selective hydrolysis or degradation of the tetrahydrofuran ring, the protease exhibits high substrate specificity, ensuring that only the target ester bond is cleaved. The patent data indicates that by optimizing the mass ratio of toluene, substrate, and enzyme, along with controlling the reaction time between 24 to 72 hours, the process achieves an ee value of up to 99.1% for the (S)-ester and high GC purity for the (R)-acid. The workup procedure further enhances purity; washing the organic layer with saturated sodium carbonate or bicarbonate solutions removes any trace acidic impurities, while drying over anhydrous sodium sulfate ensures a water-free final product. This rigorous control over the reaction environment and downstream processing guarantees a consistent quality profile that meets the stringent specifications required for pharmaceutical intermediate manufacturing.
How to Synthesize Ethyl Tetrahydrofuran-2-Carboxylate Efficiently
Implementing this enzymatic resolution process in a laboratory or pilot plant setting requires careful attention to the preparation of the reaction medium and the maintenance of physiological conditions for the enzyme. The protocol begins with the dissolution of the racemic ethyl ester in a mixture of phosphate buffer and toluene, followed by the addition of the alkaline protease, such as Alcalase 2.4L or Protease P6. It is crucial to maintain the pH at 8.0 throughout the reaction, often requiring the dropwise addition of sodium hydroxide solution to counteract the acid generated during hydrolysis. The detailed standardized synthetic steps, including specific reagent quantities, stirring speeds, and isolation techniques, are outlined in the structured guide below to ensure reproducibility and safety.
- Prepare the reaction mixture by adding racemic ethyl 2-tetrahydrofurfuryl acid ester, alkaline protease, and toluene into a phosphate buffer solution adjusted to pH 8.
- Maintain the hydrolysis reaction at a controlled temperature between 0-20 ℃ for a duration of 24 to 72 hours with continuous stirring.
- Separate the products by filtering to recover the enzyme, adjusting pH to extract the (S)-ester into the organic layer, and acidifying the aqueous layer to isolate the (R)-acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic process offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of expensive chiral resolving agents and the reduction in solvent usage directly contribute to a significant reduction in raw material costs, making the final product more price-competitive in the global market. Furthermore, the simplicity of the operation—requiring standard reactors without the need for specialized high-pressure or cryogenic equipment—lowers the barrier to entry for manufacturing and reduces capital expenditure. The ability to recover and potentially reuse the enzyme from the filter cake adds another layer of cost efficiency, while the mild reaction conditions ensure a safer working environment, reducing insurance and compliance costs associated with hazardous chemical handling.
- Cost Reduction in Manufacturing: The enzymatic route fundamentally alters the cost structure by replacing stoichiometric amounts of chiral auxiliaries with catalytic amounts of enzyme, which are generally more cost-effective on a per-kg basis. Since the process avoids the need for multiple recrystallization steps and extensive solvent exchanges, the operational expenses related to energy and waste treatment are drastically simplified. This lean manufacturing approach allows for substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final API or intermediate.
- Enhanced Supply Chain Reliability: Reliance on complex chemical resolving agents often introduces supply chain vulnerabilities due to the limited number of global suppliers for such specialty chemicals. By switching to widely available alkaline proteases and common solvents like toluene and ethyl acetate, the manufacturing process becomes far more resilient to market fluctuations. The robustness of the enzyme under the specified conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale experiments to multi-ton commercial production without significant re-engineering. The use of aqueous buffers and the absence of heavy metal catalysts simplify the wastewater treatment process, ensuring compliance with increasingly strict environmental regulations. This 'green' credential is not just a regulatory box to tick but a valuable asset for companies aiming to reduce their carbon footprint and appeal to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the enzymatic resolution of tetrahydrofuran derivatives, based on the specific data and embodiments provided in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios. The answers reflect the proven capabilities of the method to deliver high-purity chiral compounds efficiently.
Q: What are the advantages of using alkaline protease over chemical resolving agents?
A: Using alkaline protease eliminates the need for expensive chiral chemical resolving agents and reduces solvent consumption. The enzymatic method operates under mild conditions (0-20 ℃) and allows for the simultaneous recovery of both (S)-ester and (R)-acid configurations, significantly improving atom economy compared to traditional recrystallization methods.
Q: What optical purity can be achieved with this enzymatic resolution process?
A: According to patent data, the process yields (S)-ethyl 2-tetrahydrofurfuryl acid with an optical purity (ee value) exceeding 99%, and (R)-2-tetrahydrofurfuryl acid with an ee value around 80%, demonstrating high stereoselectivity suitable for pharmaceutical applications.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process is highly scalable due to its simple operation, low equipment requirements, and the use of robust alkaline proteases. The absence of harsh chemicals and the ability to recover the enzyme from the filter cake further support large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl Tetrahydrofuran-2-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalytic processes like the one described in CN107828831B for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, including the precise measurement of ee values and GC purity required for pharmaceutical applications. We are committed to delivering reliable supply chain solutions that leverage the latest advancements in green chemistry to meet your project timelines.
We invite you to collaborate with our technical team to explore how this enzymatic resolution technology can optimize your specific synthesis requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this biocatalytic route for your specific volume needs. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
