Advanced Transesterification Technology for Commercial Scale Isooctyl Salicylate Production
The global demand for high-performance UVB absorbers in the personal care and pharmaceutical sectors has necessitated a re-evaluation of traditional synthetic routes for key intermediates like isooctyl salicylate. Patent CN102775311B introduces a transformative preparation method that shifts away from corrosive direct esterification towards a more efficient, base-catalyzed transesterification process. This technological leap addresses long-standing industry pain points regarding product color, equipment longevity, and overall process economics. By utilizing methyl salicylate and isooctyl alcohol as primary feedstocks in the presence of inexpensive solid inorganic bases, manufacturers can achieve recovery rates exceeding 90% with purity levels surpassing 99.0%. 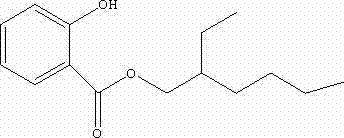 This structural integrity is paramount for its function as a UV filter, and the patented synthesis ensures minimal degradation of the aromatic ring during production. For R&D directors and procurement specialists seeking a reliable isooctyl salicylate supplier, understanding the mechanistic advantages of this transesterification pathway is essential for securing a stable, high-quality supply chain.
This structural integrity is paramount for its function as a UV filter, and the patented synthesis ensures minimal degradation of the aromatic ring during production. For R&D directors and procurement specialists seeking a reliable isooctyl salicylate supplier, understanding the mechanistic advantages of this transesterification pathway is essential for securing a stable, high-quality supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of isooctyl salicylate has relied heavily on the direct esterification of salicylic acid with isooctyl alcohol using concentrated sulfuric acid as a catalyst. This legacy approach is fraught with significant operational and quality control challenges that hinder modern lean manufacturing initiatives. The use of strong mineral acids inevitably leads to severe corrosion of reactor vessels and piping, necessitating expensive Hastelloy or glass-lined equipment and frequent maintenance schedules that disrupt production continuity. Furthermore, the oxidative nature of sulfuric acid at elevated temperatures often results in the formation of colored by-products, yielding a final product that is dark in appearance and requires extensive, yield-reducing bleaching or refining steps to meet the aesthetic standards of the cosmetic industry. Alternative acid catalysts like p-toluenesulfonic acid or solid superacids have been explored, yet they often require excessively high reaction temperatures exceeding 190°C and large excesses of alcohol to drive equilibrium, leading to substantial energy consumption and difficult solvent recovery processes.
The Novel Approach
In stark contrast, the methodology disclosed in CN102775311B employs a transesterification strategy using methyl salicylate and isooctyl alcohol catalyzed by solid inorganic bases such as sodium hydroxide or potassium hydroxide. This novel approach fundamentally alters the reaction thermodynamics and kinetics, allowing the process to proceed efficiently at temperatures ranging from 100°C to 200°C, which is significantly milder than many acid-catalyzed alternatives. The use of methyl salicylate as the acyl donor eliminates the generation of water during the reaction, thereby removing the need for water-carrying agents like toluene or benzene, which are increasingly regulated due to environmental and toxicity concerns. Instead, the by-product is methanol, a low-boiling solvent that is easily removed from the reaction mixture, driving the equilibrium towards the product side without the need for complex azeotropic distillation setups. This shift not only simplifies the reactor configuration but also drastically reduces the environmental footprint of the manufacturing process.
Mechanistic Insights into Base-Catalyzed Transesterification
The core of this technological advancement lies in the precise manipulation of nucleophilic acyl substitution mechanisms facilitated by the inorganic base catalyst. When solid NaOH or KOH is introduced to the mixture of methyl salicylate and isooctyl alcohol, it generates alkoxide species in situ which act as potent nucleophiles. These alkoxides attack the carbonyl carbon of the methyl salicylate ester, forming a tetrahedral intermediate that subsequently collapses to expel the methoxide leaving group. Unlike the reversible equilibrium often observed with weaker catalysts, the continuous removal of the volatile methanol by-product through distillation during the 4 to 10-hour reaction window effectively locks the reaction in the forward direction. This mechanistic pathway ensures that the conversion of methyl salicylate is nearly complete, minimizing the presence of starting materials in the crude product and reducing the burden on downstream purification units. The choice of inorganic bases over organic alkoxides like sodium methoxide is particularly strategic; while sodium methoxide is a strong nucleophile, it can sometimes induce side reactions or suffer from hydrolysis if moisture is present, whereas the solid inorganic bases offer robust stability and easier handling characteristics in large-scale reactors.
From an impurity control perspective, this base-catalyzed route offers superior selectivity compared to acid-catalyzed methods. Acidic conditions can promote electrophilic aromatic substitution or dehydration side reactions that lead to polymeric tars and colored impurities, which are notoriously difficult to separate from the target ester. The basic environment of the transesterification process preserves the integrity of the phenolic hydroxyl group on the salicylate ring, preventing unwanted etherification or condensation reactions. Consequently, the crude organic phase obtained after the reaction and hot water washing is remarkably clean, facilitating the final vacuum distillation step. The ability to wash the reaction mixture with hot water at 50-100°C effectively removes residual catalyst and any soap-like species formed, ensuring that the final distillation feed is free from non-volatile salts that could degrade under heat. This rigorous control over the reaction environment is what enables the consistent achievement of purity specifications greater than 99.0%, a critical metric for high-purity UVB absorber applications.
How to Synthesize Isooctyl Salicylate Efficiently
The synthesis protocol outlined in the patent provides a robust framework for scaling this chemistry from laboratory benchtop to multi-ton commercial production. The process begins with the precise blending of methyl salicylate and isooctyl alcohol, followed by the controlled addition of the solid catalyst and a programmed heating cycle that integrates reaction and by-product removal. Detailed standard operating procedures regarding mixing speeds, heating ramps, and vacuum levels are critical for reproducibility.
- Mix methyl salicylate and isooctyl alcohol (molar ratio 1: 1 to 1:3) and stir for 1-3 hours to form a homogeneous raw material solution.
- Add a solid inorganic base catalyst (NaOH or KOH, 0.2%-1.0% mass) to the solution to initiate the transesterification reaction.
- Heat the mixture to 100-200°C for 4-10 hours, recover methanol byproduct, wash with hot water, dry, and vacuum distill to collect the final fraction at 174-178°C/1.0kPa.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this transesterification technology translates directly into tangible operational efficiencies and risk mitigation strategies. The shift from corrosive liquid acids to solid inorganic bases eliminates the logistical hazards and storage costs associated with handling bulk sulfuric acid, thereby enhancing site safety and reducing insurance premiums. Furthermore, the simplified workup procedure, which relies on basic hot water washing rather than complex neutralization and extraction sequences, significantly reduces the volume of aqueous waste generated per kilogram of product. This reduction in waste treatment load is a major driver for cost reduction in cosmetic ingredient manufacturing, as effluent disposal costs continue to rise globally. The ability to recover methanol as a valuable by-product further improves the overall mass balance of the process, turning a potential waste stream into a revenue-generating asset or an internal utility for other synthesis steps.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of commodity-grade inorganic bases like NaOH or KOH, which are significantly cheaper and more readily available than specialized catalysts like sodium methoxide or solid superacids. The elimination of water-carrying solvents removes both the purchase cost of these solvents and the energy cost required to recover and dry them for reuse. Additionally, the milder reaction temperatures (100-200°C) compared to the >200°C required for some acid methods result in lower steam or thermal oil consumption, directly lowering the variable cost of production. These cumulative savings allow for a more competitive pricing structure without compromising on the stringent quality requirements of the personal care market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available raw materials; methyl salicylate and isooctyl alcohol are commodity chemicals with stable global supply networks, reducing the risk of raw material shortages. The robustness of the inorganic base catalyst means that the process is less sensitive to minor fluctuations in raw material quality or trace moisture content compared to moisture-sensitive alkoxide catalysts. This tolerance reduces the rate of batch failures and reworks, ensuring a consistent output of finished goods. For buyers seeking reducing lead time for high-purity cosmetic intermediates, this reliability means fewer delays and more predictable delivery schedules, which is crucial for just-in-time manufacturing environments in the downstream cosmetic formulation sector.
- Scalability and Environmental Compliance: The commercial scale-up of complex esters is often hindered by heat transfer limitations and safety concerns, but this transesterification process is inherently scalable due to its homogeneous nature and manageable exotherm. The absence of corrosive acids extends the lifespan of standard stainless steel reactors, reducing capital expenditure on specialized metallurgy. From an environmental compliance standpoint, the process aligns with green chemistry principles by avoiding toxic solvents and generating a recyclable methanol by-product. This eco-friendly profile is increasingly becoming a prerequisite for inclusion in the supply chains of major multinational personal care brands who are committed to sustainability goals, making this manufacturing route a strategic asset for long-term business partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of isooctyl salicylate using this advanced transesterification technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making.
Q: Why is the inorganic base catalyzed method superior to traditional sulfuric acid esterification?
A: Traditional sulfuric acid methods often result in dark-colored products due to oxidation and cause severe equipment corrosion. The inorganic base catalyzed transesterification described in patent CN102775311B operates under milder conditions, yielding a colorless to pale yellow product with purity exceeding 99.0%, while eliminating the need for corrosive acid handling and complex neutralization waste streams.
Q: How does this process improve yield compared to sodium methoxide catalysis?
A: While sodium methoxide catalysis often suffers from reaction equilibrium limitations leaving residual methyl salicylate, the solid inorganic base method drives the reaction more effectively at temperatures between 100-200°C. This approach achieves recovery rates exceeding 90% and allows for the efficient recovery of methanol byproduct, enhancing overall economic efficiency.
Q: What are the critical purification steps for achieving pharmaceutical grade purity?
A: The process utilizes a specific hot water washing step (50-100°C) to remove catalyst residues and polar impurities, followed by drying with anhydrous Na2SO4. The final critical step is vacuum distillation collecting the fraction at 174-178°C under 1.0kPa pressure, which ensures the removal of unreacted isooctyl alcohol and delivers high-purity isooctyl salicylate suitable for sensitive cosmetic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isooctyl Salicylate Supplier
The technical potential of the base-catalyzed transesterification route for isooctyl salicylate represents a significant opportunity for optimizing the cost and quality of UVB absorber supply chains. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of isooctyl salicylate meets the exacting standards required for sunscreen and cosmetic formulations. We understand that consistency is key in the fine chemical industry, and our process controls are designed to minimize batch-to-batch variation.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis capabilities, we can help you reduce total landed costs while securing a supply of high-purity materials. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us demonstrate how our commitment to advanced manufacturing technologies can become a cornerstone of your supply chain strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
