Advanced Synthesis of Imidazopyridazine Derivatives for Anti-Asthmatic Pharmaceutical Applications
Introduction to Novel Imidazopyridazine Therapeutics
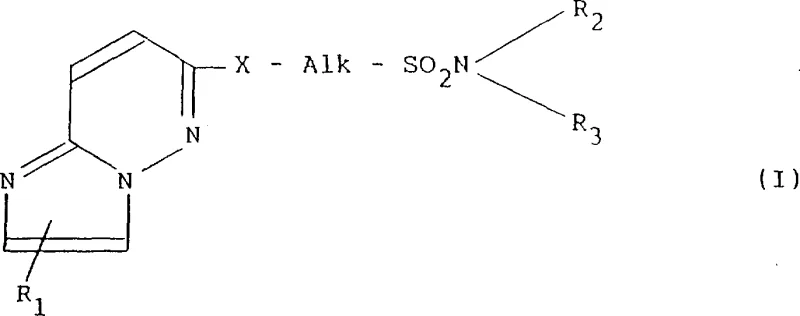
The pharmaceutical landscape for respiratory diseases continuously demands novel chemical entities that offer improved efficacy and safety profiles. Patent CN1032361C introduces a significant advancement in this field through the disclosure of novel imidazo[1,2-b]pyridazine compounds. These derivatives are specifically engineered to possess potent anti-allergic, anti-inflammatory, and anti-PAF (platelet activating factor) activities. The core innovation lies in the strategic substitution at the 6-position of the imidazopyridazine ring system, which has been historically underexplored for anti-asthmatic applications compared to its cardiovascular counterparts. This structural modification allows for the modulation of bronchospasm and bronchoconstriction, addressing a critical unmet need in asthma management.
The general formula (I) encompasses a broad range of substituents, allowing for fine-tuning of pharmacokinetic properties. As illustrated in the structural diagram below, the molecule features a fused heterocyclic core linked via an alkylene chain to a sulfonamide moiety. This specific architecture is crucial for binding affinity and metabolic stability. For procurement specialists and R&D teams, understanding this scaffold is the first step towards securing a reliable supply of high-purity pharmaceutical intermediates capable of driving next-generation respiratory therapies forward.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of imidazopyridazine derivatives focused heavily on cardiovascular indications, as noted in prior art such as Japanese Patent Application Laid-Open No. 61-152684. While these earlier compounds demonstrated cardiotonic activity, they lacked the specific profile required for effective asthma treatment. Conventional synthetic routes often struggled with regioselectivity issues when attempting to functionalize the 6-position of the pyridazine ring without affecting other reactive sites. Furthermore, traditional methods for introducing sulfonamide groups frequently involved harsh conditions that could degrade the sensitive heterocyclic core, leading to complex impurity profiles and reduced overall yields. The inability to efficiently couple bulky or branched alkyl sulfonamide chains also limited the chemical space available for optimization, restricting the potential for discovering compounds with superior bioavailability.
The Novel Approach
The methodology presented in CN1032361C overcomes these hurdles through a modular and versatile synthetic strategy. By employing distinct coupling methods (Method A, B, and C), the inventors provide robust pathways to access a diverse library of derivatives. Method A, for instance, utilizes a nucleophilic substitution between a mercapto-imidazopyridazine and a reactive sulfonamide precursor. This approach allows for the introduction of various alkylene linkers and sulfonamide terminations under relatively mild conditions. The flexibility to use different leaving groups (halogens, sulfonates) and bases (alkoxides, hydrides) ensures that the process can be adapted for different substrate solubilities and reactivities. This adaptability is a key factor in cost reduction in pharmaceutical intermediate manufacturing, as it minimizes the need for specialized reagents and allows for the use of commodity chemicals.
Mechanistic Insights into Nucleophilic Substitution and Coupling
The core chemical transformation driving this synthesis is a nucleophilic substitution reaction, typically occurring at the 6-position of the imidazopyridazine ring. In Method A, the sulfur or oxygen atom of the side chain acts as the nucleophile, attacking the electron-deficient carbon at the 6-position of the chloro-imidazopyridazine substrate. The presence of a base, such as sodium methoxide or sodium hydride, is critical for deprotonating the thiol or hydroxyl group, thereby generating the active nucleophilic species. The reaction mechanism proceeds through a transition state where the leaving group (typically chloride) is displaced. The choice of solvent plays a pivotal role in stabilizing the transition state and solubilizing the ionic intermediates; polar aprotic solvents like dimethylformamide (DMF) or dimethylsulfoxide (DMSO) are often preferred to enhance reaction rates.
Impurity control is managed through careful selection of reaction temperatures and stoichiometry. The patent specifies reaction temperatures ranging from 10°C to 200°C, with a preferred range of 50°C to 100°C for optimal conversion while minimizing side reactions such as over-alkylation or hydrolysis of the sulfonamide group. Post-reaction workup involves standard extraction techniques followed by purification via silica gel column chromatography. This step is essential for removing unreacted starting materials and by-products, ensuring the final intermediate meets stringent purity specifications required for API synthesis. The ability to recrystallize the final products from solvents like methanol or ethanol further enhances the purity profile, delivering a high-quality material suitable for downstream pharmaceutical processing.

How to Synthesize 6-Substituted Imidazopyridazine Sulfonamides Efficiently
The synthesis of these valuable intermediates follows a logical progression of activation, coupling, and purification. The process begins with the preparation of the sulfonamide side chain, which can be derived from readily available alkyl halides or alcohols. Once the side chain is activated (either as a thiol, alcohol, or halide), it is coupled with the imidazopyridazine core under basic conditions. The detailed standardized synthesis steps provided in the patent examples serve as a robust blueprint for scaling this chemistry. For R&D teams looking to replicate or optimize this pathway, adherence to the specified molar ratios and temperature controls is paramount to achieving reproducible results.
- Preparation of the nucleophilic precursor by converting hydroxy-alkyl-sulfonamides to mercapto-alkyl-sulfonamides or activating them as halides.
- Condensation of the activated sulfonamide precursor with 6-chloroimidazo[1,2-b]pyridazine in the presence of a base like sodium methoxide.
- Purification of the crude reaction mixture via silica gel column chromatography followed by recrystallization from methanol or ether to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthetic routes described in this patent offer substantial advantages in terms of raw material availability and process robustness. The starting materials, such as chloro-imidazopyridazines and various alkyl sulfonamides, are accessible through established chemical supply chains, reducing the risk of bottlenecks associated with exotic reagents. The use of common solvents like methanol, ethanol, and dichloromethane simplifies solvent recovery and waste management protocols, contributing to a more sustainable manufacturing process. Furthermore, the reaction conditions are compatible with standard stainless steel reactor equipment, eliminating the need for specialized corrosion-resistant vessels that would otherwise drive up capital expenditure.
- Cost Reduction in Manufacturing: The synthetic pathways avoid the use of expensive transition metal catalysts or precious metal reagents, which are often cost-prohibitive in large-scale production. By relying on base-mediated nucleophilic substitutions, the process significantly lowers the bill of materials. Additionally, the high selectivity of the reactions reduces the burden on downstream purification, leading to higher overall yields and less waste generation. This efficiency translates directly into lower production costs per kilogram, making the final API more economically viable for market entry.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the decoupling of side-chain production from the core heterocycle coupling. This means that key intermediates can be stockpiled independently, providing a buffer against supply disruptions. The robustness of the chemistry ensures consistent quality across batches, which is critical for maintaining regulatory compliance and avoiding costly production delays. Suppliers can confidently commit to long-term contracts knowing that the process is stable and scalable.
- Scalability and Environmental Compliance: The processes described operate at atmospheric pressure and moderate temperatures, facilitating safe scale-up from pilot plant to commercial tonnage. The absence of hazardous high-pressure hydrogenation steps or cryogenic conditions simplifies operational safety requirements. Moreover, the waste streams generated are primarily organic solvents and inorganic salts, which can be treated using standard effluent treatment protocols, ensuring alignment with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these imidazopyridazine derivatives. The answers are derived directly from the technical disclosures within the patent documentation, providing clarity on stability, formulation, and regulatory considerations. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating these intermediates into their development pipelines.
Q: What are the primary biological activities of these imidazopyridazine compounds?
A: According to patent CN1032361C, these compounds exhibit significant anti-allergic, anti-inflammatory, and anti-PAF (platelet activating factor) activities, making them potent candidates for anti-asthmatic therapies.
Q: Can this synthesis route be scaled for commercial production?
A: Yes, the described methods utilize standard organic synthesis techniques such as refluxing in common solvents (methanol, DMF) and standard purification methods like chromatography and recrystallization, which are highly amenable to scale-up from laboratory to industrial manufacturing.
Q: What forms do the final pharmaceutical products take?
A: The patent describes various pharmaceutically acceptable salts, including hydrochlorides and methanesulfonates, and formulations such as tablets, capsules, and injectable solutions, ensuring versatility in drug delivery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazopyridazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving respiratory medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of imidazopyridazine intermediate meets the highest industry standards. Our commitment to quality assurance means that you can trust our materials to perform consistently in your downstream API synthesis and formulation studies.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your next-generation asthma therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your rapid development timelines and help you bring effective anti-asthmatic agents to market faster.
