Scalable Synthesis of Asymmetric 9-Alkoxy Phenanthrene Dimers for Advanced Optoelectronics
Scalable Synthesis of Asymmetric 9-Alkoxy Phenanthrene Dimers for Advanced Optoelectronics
The rapid evolution of the organic light-emitting diode (OLED) and asymmetric catalysis sectors demands increasingly sophisticated polycyclic aromatic hydrocarbon architectures. A pivotal breakthrough in this domain is documented in patent CN109111351B, which details a robust synthetic methodology for constructing asymmetric 9-alkoxy phenanthrene dimers. Unlike traditional approaches that often result in symmetrical structures with limited functionalization potential, this invention leverages a persulfate-mediated oxidative coupling strategy to selectively forge C-C bonds between the 1-position and 9'-position of phenanthrene derivatives. This technical advancement is critical for manufacturers seeking a reliable optoelectronic material supplier capable of delivering complex, non-symmetrical building blocks. By shifting the reaction locus from the conventional C10 site to the C1/C9' axis, the process unlocks new avenues for designing large conjugated systems with tailored electronic properties, addressing a long-standing bottleneck in the synthesis of high-performance photoelectric materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidative dimerization of 9-hydroxy phenanthrene and its alkoxy derivatives has been plagued by inherent regioselectivity issues driven by the electronic nature of the phenanthrene core. In standard oxidative conditions, the electron density is highest at the C10 position, leading almost exclusively to homocoupling at this site. This results in the formation of completely symmetrical 9,9'-dihydroxy or 9,9'-dialkoxy phenanthrene dimers, as illustrated in the reaction scheme below. Such symmetrical products present a significant drawback for downstream applications; once the C10 positions are consumed in the dimerization, the resulting molecule lacks reactive sites for further derivatization. This structural rigidity limits the utility of these compounds in the construction of complex chiral ligands or extended pi-conjugated systems required for next-generation display technologies. Furthermore, many traditional methods rely on transition metal catalysts, such as copper complexes, which introduce the risk of metal contamination—a critical failure mode in semiconductor and OLED manufacturing where trace metals can quench luminescence.
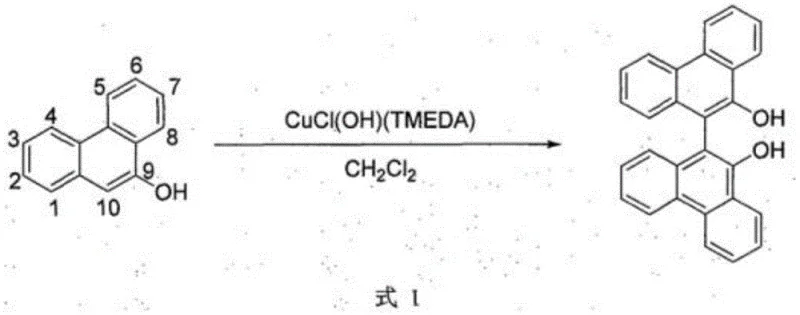
The Novel Approach
To overcome these structural and processing limitations, the disclosed invention introduces a paradigm shift by utilizing inorganic persulfates as potent oxidants within a specific acidic environment. This method successfully redirects the coupling trajectory from the C10 position to the C1 and C9' positions, yielding asymmetric 9,10'-dialkoxy-1,9'-biphenanthrene compounds. The general reaction pathway is depicted below, showcasing the versatility of the method across different alkoxy substituents. This approach not only preserves the C10 active site for potential future functionalization but also constructs a rigid, asymmetric backbone that is highly desirable for inducing chirality or tuning band gaps in organic semiconductors. By avoiding transition metals and utilizing inexpensive, commercially available persulfate salts, the process offers a streamlined route to high-value intermediates. This represents a substantial improvement in cost reduction in display material manufacturing, as it simplifies the purification workflow and eliminates the need for expensive metal scavengers.
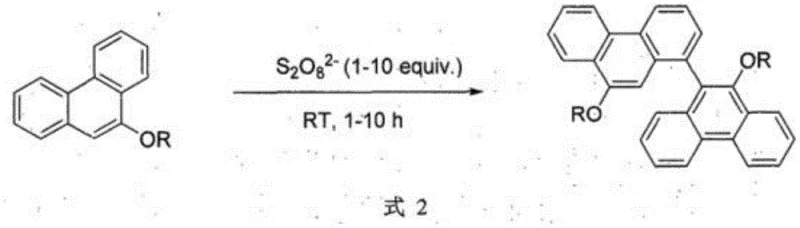
Mechanistic Insights into Persulfate-Mediated Oxidative Coupling
The success of this synthetic route lies in the unique mechanistic pathway facilitated by the persulfate anion (S2O8 2-) in the presence of a strong acid like trifluoroacetic acid (TFA). Under these conditions, the persulfate acts as a single-electron oxidant, generating a radical cation intermediate on the electron-rich phenanthrene ring. The acidic medium plays a dual role: it protonates the alkoxy oxygen to modulate the electron density of the aromatic system and stabilizes the radical intermediates, preventing uncontrolled polymerization. The steric and electronic environment created by the 9-alkoxy group directs the radical attack to the C1 position of one molecule and the C9' position of another, rather than the thermodynamically favored C10 site. This regioselective control is crucial for obtaining the asymmetric dimer with high fidelity. The reaction proceeds through a radical-radical coupling or a radical-nucleophilic substitution mechanism, eventually rearomatizing to form the stable biaryl bond. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as temperature and oxidant stoichiometry, to maximize the yield of the desired isomer while suppressing side reactions.
From an impurity control perspective, the absence of transition metals significantly simplifies the impurity profile. In copper-catalyzed systems, residual metal ions and ligand byproducts are common contaminants that require rigorous and costly removal steps, often involving silica gel treatment or specialized chelating resins. In contrast, the byproducts of the persulfate oxidation are primarily inorganic sulfates and acids, which are easily removed during the aqueous workup and neutralization steps described in the patent examples. The use of sodium carbonate for neutralization effectively quenches the acidic reaction mixture and precipitates inorganic salts, allowing for a clean separation of the organic phase. This results in a crude product with high purity, which can be further polished to >95% purity via standard column chromatography. This clean reaction profile is essential for maintaining the integrity of the supply chain for high-purity optoelectronic materials, ensuring that the final product meets the stringent specifications required by end-users in the electronics industry.
How to Synthesize 9,10'-Dialkoxy-1,9'-biphenanthrene Efficiently
The practical implementation of this chemistry is straightforward and amenable to standard laboratory and pilot-scale equipment. The protocol involves dissolving the 9-alkoxyphenanthrene substrate in a chlorinated or aromatic solvent, cooling the mixture to control the exotherm upon oxidant addition, and maintaining an inert atmosphere to prevent side oxidation. The specific examples in the patent demonstrate the robustness of the method across methoxy, ethoxy, and propoxy substrates, yielding the corresponding asymmetric dimers in moderate to good yields (53%-64%). For detailed operational parameters and safety considerations regarding the handling of strong oxidants and acids, please refer to the standardized synthesis guide below.
- Dissolve 9-alkoxyphenanthrene substrate in an organic solvent such as dichloromethane or toluene under a nitrogen atmosphere.
- Add persulfate oxidant (1.0-10.0 equivalents) at 0°C, followed by the injection of trifluoroacetic acid to initiate the reaction.
- Stir the mixture for 1-3 hours until substrate consumption is confirmed by TLC, then neutralize with sodium carbonate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this persulfate-mediated synthesis offers compelling economic and logistical benefits over traditional transition-metal catalyzed routes. The primary advantage stems from the drastic simplification of the raw material bill of materials (BOM). By replacing expensive and often supply-constrained transition metal catalysts with commodity chemicals like sodium persulfate and trifluoroacetic acid, the direct material costs are significantly reduced. Furthermore, the elimination of heavy metals from the process flow removes the need for specialized metal scavenging resins and the associated validation testing for residual metals, which are time-consuming and costly regulatory hurdles in the pharmaceutical and electronic materials sectors. This streamlining translates directly into faster batch turnover times and lower overall production costs, enhancing the competitiveness of the final product in the global market.
- Cost Reduction in Manufacturing: The shift to a metal-free oxidative coupling system fundamentally alters the cost structure of producing these complex aromatics. Traditional methods often require stoichiometric or near-stoichiometric amounts of copper salts and specialized ligands, which represent a significant portion of the variable cost. By utilizing inexpensive inorganic oxidants, the process achieves substantial cost savings without compromising on yield or selectivity. Additionally, the simplified workup procedure reduces the consumption of solvents and stationary phases during purification, further driving down the cost of goods sold (COGS). This economic efficiency makes the commercial scale-up of complex optoelectronic intermediates much more viable, allowing suppliers to offer competitive pricing for high-performance materials.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like persulfates and common organic solvents mitigates the risk of supply chain disruptions often associated with specialized catalysts. Transition metal catalysts can be subject to geopolitical supply constraints or fluctuating market prices, whereas the reagents used in this novel method are produced globally in massive volumes. This ensures a stable and continuous supply of critical raw materials, reducing the lead time for high-purity phenanthrene dimers. For supply chain heads, this reliability is paramount in maintaining production schedules for downstream OLED or pharmaceutical clients who cannot afford delays due to raw material shortages. The robustness of the reaction conditions also implies a wider operating window, reducing the risk of batch failures and ensuring consistent delivery performance.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than that of heavy-metal catalyzed alternatives. The waste stream consists primarily of benign inorganic salts and aqueous acid, which are easier to treat and dispose of compared to heavy metal-contaminated waste. This aligns with increasingly stringent global environmental regulations and corporate sustainability goals. From a scalability perspective, the reaction does not require exotic equipment or extreme conditions (such as high pressure), making it easily transferable from gram-scale laboratory synthesis to multi-kilogram or ton-scale commercial production. The ability to scale this process efficiently ensures that suppliers can meet growing demand for advanced materials without encountering the technical bottlenecks often associated with complex organometallic chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these asymmetric phenanthrene dimers. The answers are derived directly from the experimental data and technical disclosures found in the underlying patent literature, providing a factual basis for evaluating the technology's potential.
Q: Why is asymmetric coupling preferred over symmetric coupling for phenanthrene derivatives?
A: Symmetric coupling typically occurs at the C10 position, which deactivates the molecule for further derivatization. Asymmetric coupling at the C1/C9' positions retains the C10 active site, allowing for the creation of complex, multi-ring conjugated systems essential for advanced optoelectronic applications.
Q: What are the typical yields and purity levels for this persulfate-mediated method?
A: According to patent CN109111351B, the method achieves isolated yields ranging from 53% to 64% with product purity exceeding 95% as confirmed by NMR spectroscopy, making it suitable for high-grade electronic material applications.
Q: Does this process require transition metal catalysts?
A: No, this novel method utilizes persulfate salts (such as sodium, potassium, or ammonium persulfate) as the oxidant in an acidic system, eliminating the need for expensive transition metal catalysts like copper and the subsequent heavy metal removal steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,10'-Dialkoxy-1,9'-biphenanthrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation electronic and pharmaceutical products. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the persulfate-mediated coupling described above can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 9,10'-dialkoxy-1,9'-biphenanthrene meets the exacting standards required for optoelectronic applications. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply of advanced aromatic building blocks.
We invite R&D directors and procurement specialists to collaborate with us on optimizing their supply chains for these valuable materials. Whether you require custom synthesis of specific alkoxy derivatives or assistance with process optimization to further enhance yield and purity, our technical team is ready to support your goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
