Revolutionizing Pharmaceutical Intermediate Production: Scalable Synthesis of Indolizine Mannich Base Compounds
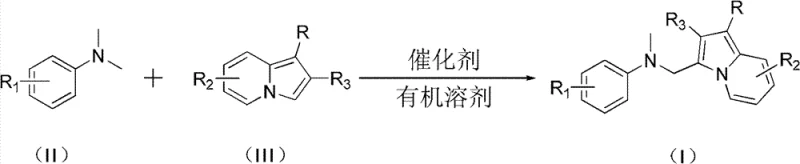
The Chinese patent CN102093354B introduces a groundbreaking methodology for synthesizing indolizine Mannich base compounds, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach addresses long-standing challenges in the field by providing a streamlined pathway to these structurally complex molecules, which serve as essential building blocks for novel drug development. The patent details a copper-catalyzed reaction system that operates under remarkably mild conditions compared to conventional methods, offering pharmaceutical manufacturers a more efficient route to high-value intermediates. By eliminating the need for aldehyde substrates and expanding substrate scope beyond traditional limitations, this technology opens new possibilities for medicinal chemistry research and development. The process demonstrates exceptional versatility across various substituent patterns while maintaining high product purity standards required in pharmaceutical applications, positioning it as a valuable asset for companies seeking reliable intermediate supply chains.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional Mannich reaction methodologies have historically faced significant constraints that limit their practical utility in pharmaceutical intermediate production, as documented in established literature including Angewandte Chemie International Edition (1998, 37, 1044–1070) and Synthesis (1973, 703–775). These conventional approaches typically require extended reaction durations often exceeding 24 hours under harsh conditions that pose safety concerns and increase operational complexity. Furthermore, they are fundamentally restricted by their dependence on aldehyde substrates and limited compatibility with aliphatic amine compounds, severely constraining the structural diversity of accessible products. The narrow substrate scope creates substantial barriers for medicinal chemists seeking to explore novel molecular architectures, while the demanding reaction parameters complicate scale-up efforts and increase production costs through energy-intensive processing requirements. These inherent limitations have long hindered the efficient synthesis of indolizine-based intermediates despite their recognized importance in pharmaceutical development pipelines.
The Novel Approach
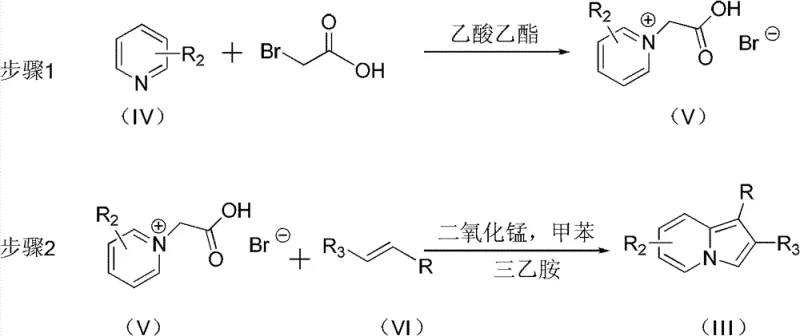
The patented methodology presented in CN102093354B overcomes these historical limitations through an innovative copper-catalyzed system that operates under significantly milder conditions between 30–70°C with dramatically reduced reaction times of 8–20 hours. This breakthrough eliminates the requirement for aldehyde substrates while expanding compatibility to diverse aromatic amine compounds, thereby enabling access to a broader range of structurally complex indolizine Mannich base derivatives. The process utilizes commercially available monovalent copper catalysts such as CuCl, CuBr, or CuI at optimized molar ratios (catalyst:substrate = 0.05–0.2:1), ensuring efficient transformation without the need for specialized equipment or hazardous reagents. By simplifying the reaction setup and workup procedures while maintaining high yields across various substituent patterns, this approach delivers substantial operational advantages that directly address the pain points experienced by pharmaceutical manufacturers in intermediate production.
Mechanistic Insights into Copper-Catalyzed Indolizine Mannich Reaction
The core innovation lies in the copper-mediated activation mechanism that facilitates the direct coupling between N,N-dimethyl aromatic amines and indolizine compounds without traditional aldehyde involvement. The monovalent copper catalyst forms a transient complex with the indolizine nitrogen atom, lowering the activation energy barrier for electrophilic attack on the aromatic amine's methyl group. This unique pathway proceeds through a proposed iminium ion intermediate that subsequently undergoes nucleophilic addition by the indolizine system, followed by rearomatization to yield the final Mannich base product. The reaction demonstrates remarkable regioselectivity across various substitution patterns on both reactant classes, with the copper catalyst playing a dual role in substrate activation and stereochemical control. This mechanistic pathway represents a significant departure from conventional Mannich chemistry by eliminating the need for pre-formed imines or aldehydes while maintaining excellent functional group tolerance.
Impurity profile management is inherently addressed through the reaction's precise mechanistic control and optimized conditions that minimize side reactions. The mild temperature range (30–70°C) prevents thermal degradation pathways common in traditional methods, while the selective copper catalysis suppresses unwanted polymerization or over-reaction products. The well-defined reaction stoichiometry (N,N-dimethyl aromatic amine:indolizine:catalyst = 2–6:1:0.05–0.2) ensures complete conversion without excess reagents that could lead to impurities. Post-reaction processing through straightforward filtration and silica gel-assisted column chromatography effectively removes catalyst residues and minor byproducts, consistently delivering products meeting stringent pharmaceutical purity requirements without requiring additional purification steps that could compromise yield or introduce new contaminants.
How to Synthesize Indolizine Mannich Base Compounds Efficiently
This patented synthesis route represents a significant advancement in pharmaceutical intermediate manufacturing, offering researchers and production teams an optimized pathway to high-value indolizine derivatives with exceptional operational efficiency. The methodology provides clear advantages over conventional approaches through its simplified reaction setup, reduced processing requirements, and compatibility with standard laboratory equipment. Detailed standardized synthesis steps are provided below to facilitate seamless implementation in both research and manufacturing environments.
- Combine catalyst (CuCl, CuBr, or CuI), N,N-dimethyl aromatic amine compound, and indolizine compound in organic solvent at precise molar ratios (2–6: 1:0.05–0.2) under controlled conditions.
- Heat the reaction mixture to 30–70°C and maintain stirring for 8–20 hours to ensure complete conversion while monitoring reaction progress through standard analytical methods.
- Perform post-reaction processing including filtration, silica gel mixing, and column chromatography purification using appropriate eluents to isolate high-purity indolizine Mannich base compounds.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial strategic advantages for procurement and supply chain decision-makers by addressing critical pain points in pharmaceutical intermediate sourcing. The process eliminates dependency on specialized or restricted reagents while utilizing readily available starting materials from multiple global suppliers, significantly reducing supply chain vulnerability. The simplified workflow requires minimal specialized equipment investment and reduces operator training requirements compared to traditional methods, enabling faster technology transfer between facilities and more flexible production scheduling to meet dynamic demand patterns.
- Cost Reduction in Manufacturing: The elimination of aldehyde substrates and specialized reaction conditions reduces raw material costs while the simplified workup procedure minimizes solvent consumption and waste generation. The use of cost-effective copper catalysts instead of precious metal alternatives provides significant economic advantages without compromising product quality, while the high yields achieved across diverse substrates maximize resource utilization efficiency throughout the production cycle.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials from multiple global sources creates robust supply chain resilience against single-source dependencies. The process's compatibility with standard manufacturing equipment enables rapid technology transfer between production sites, while the consistent high yields across various substituent patterns ensure reliable output regardless of specific structural requirements for different drug development programs.
- Scalability and Environmental Compliance: The mild reaction conditions (30–70°C) and straightforward processing requirements facilitate seamless scale-up from laboratory to commercial production without requiring specialized infrastructure modifications. The reduced energy consumption compared to traditional high-temperature methods lowers environmental impact while generating less hazardous waste through simplified purification protocols, aligning with increasingly stringent environmental regulations in pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology and its implementation in pharmaceutical intermediate production environments.
Q: How does this copper-catalyzed method overcome traditional Mannich reaction limitations?
A: The patented process eliminates the need for aldehyde substrates and aliphatic amines required in conventional Mannich reactions, while operating under milder conditions (30–70°C) with significantly reduced reaction times compared to traditional methods requiring harsh conditions and extended durations.
Q: What specific advantages does this synthesis route offer for pharmaceutical intermediate production?
A: The method provides exceptional operational simplicity with straightforward workup procedures, high product yields without complex purification steps, and compatibility with diverse substituents enabling tailored intermediate production for various drug development pathways.
Q: How does this technology support supply chain reliability for pharmaceutical manufacturers?
A: The process utilizes commercially available starting materials and standard laboratory equipment, operates under safe temperature ranges, and delivers consistent high-purity outputs, thereby ensuring stable production capacity and reduced vulnerability to supply chain disruptions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Mannich Base Compound Supplier
Our company leverages this patented technology to deliver exceptional value through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs. As a specialized CDMO partner, we combine deep expertise in copper-catalyzed methodologies with comprehensive quality systems to ensure consistent delivery of high-purity indolizine intermediates meeting exacting pharmaceutical standards. Our dedicated technical teams work collaboratively with clients to optimize processes for specific structural variants while maintaining the core advantages of this innovative synthesis route.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can enhance your specific supply chain requirements. Please contact us for specific COA data and route feasibility assessments tailored to your pharmaceutical development program needs.
