Revolutionizing Pharmaceutical Intermediate Production via One-Step Acylation-Esterification
The pharmaceutical and agrochemical industries rely heavily on the consistent supply of high-purity intermediates to maintain robust production pipelines for active pharmaceutical ingredients (APIs). A significant technological advancement in this domain is detailed in patent CN1283612C, which discloses a novel process for producing 4-phenyl-4-oxo-2-butenoate derivatives. These compounds serve as critical building blocks in the synthesis of various therapeutic agents and crop protection chemicals. Traditionally, the manufacturing of these esters has been plagued by inefficiencies, including multi-step procedures, prolonged reaction times, and difficult purification protocols. The disclosed invention addresses these challenges by introducing a streamlined one-pot methodology that simultaneously performs Friedel-Crafts acylation and esterification. By leveraging a specific combination of acid catalysts and alkylating agents, this process achieves superior yields and purity levels while drastically reducing the operational complexity associated with conventional synthesis routes.
For procurement managers and supply chain directors, the implications of this technology extend beyond mere chemical curiosity; it represents a tangible opportunity for cost reduction in pharmaceutical intermediate manufacturing. The ability to produce these key precursors in a single step translates directly to lower energy consumption, reduced solvent usage, and minimized labor costs. Furthermore, the high purity achieved directly from the reaction crystallization reduces the burden on downstream purification units, thereby enhancing the overall throughput of the manufacturing facility. As a reliable pharmaceutical intermediates supplier, understanding and adopting such process intensification technologies is essential for maintaining competitiveness in a market that demands both speed and quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the method described in CN1283612C, the synthesis of 4-phenyl-4-oxo-2-butenoate derivatives was typically accomplished through a disjointed two-step sequence. The initial step involved a Friedel-Crafts reaction between an aromatic compound and maleic anhydride in the presence of aluminum chloride to generate the corresponding 4-phenyl-4-oxo-2-butenoic acid. This step was notoriously inefficient, often requiring reaction times exceeding 48 hours to reach completion, as documented in comparative examples within the patent literature. Following the acylation, the reaction mixture contained a complex slurry of aluminum hydroxide complexes, necessitating cumbersome workup procedures such as steam distillation or extensive aqueous extractions to isolate the acid intermediate. Even after successful isolation, a second distinct chemical transformation was required: the esterification of the acid to form the final target ester. This additional step introduced further opportunities for yield loss, increased raw material costs, and extended the total production cycle time to over 50 hours, creating a significant bottleneck for industrial scale-up.
The Novel Approach
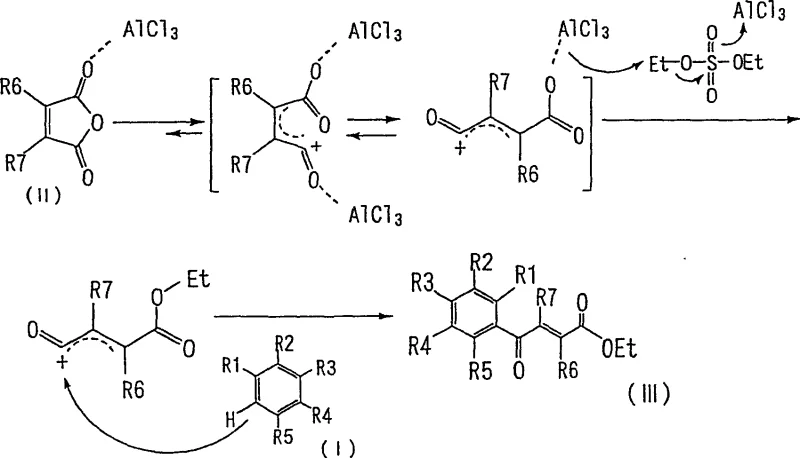
The innovative approach presented in the patent fundamentally reimagines this synthetic pathway by merging the acylation and esterification events into a concurrent, one-pot operation. By introducing an alkylating agent, such as diethyl sulfate or dimethyl sulfate, directly into the reaction system alongside the acid catalyst and maleic anhydride derivative, the process bypasses the isolation of the free acid entirely. The alkylating agent acts as a chemical trap, reacting with the intermediate species to form the ester in situ. Experimental data from the patent demonstrates that this modification reduces the total reaction time from over 50 hours to merely 4 hours while simultaneously boosting the isolated yield from approximately 54% to over 70%. This dramatic improvement in efficiency is not merely incremental; it represents a paradigm shift in process chemistry that allows for the rapid generation of high-purity materials suitable for immediate use in subsequent synthetic steps without extensive purification.
Mechanistic Insights into Alkylating Agent-Promoted Friedel-Crafts Acylation
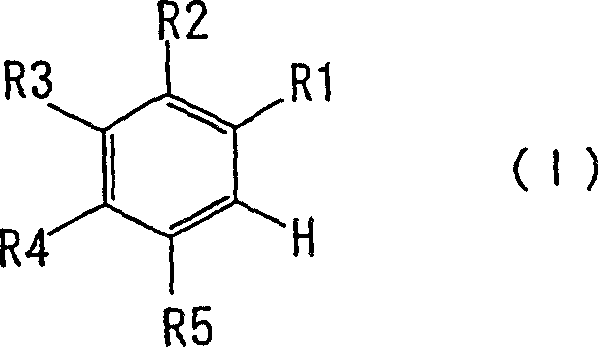
To fully appreciate the technical superiority of this method, one must examine the underlying reaction mechanism, particularly the role of the alkylating agent in manipulating chemical equilibrium. In a standard Friedel-Crafts acylation using maleic anhydride and aluminum chloride, the reaction exists in a dynamic equilibrium where the maleic anhydride tends to remain in its closed-ring cyclic form or revert to it after opening. This equilibrium limits the concentration of the reactive acyl cation species necessary for the electrophilic attack on the aromatic ring, resulting in sluggish kinetics. The presence of the alkylating agent disrupts this equilibrium by reacting with the open-chain carboxylate intermediate. Once the alkyl group is attached, the resulting ester functionality prevents the molecule from cyclizing back into the anhydride form. This irreversible trapping effect continuously pulls the equilibrium towards the open-chain acyl cation, effectively increasing its steady-state concentration within the reaction vessel.
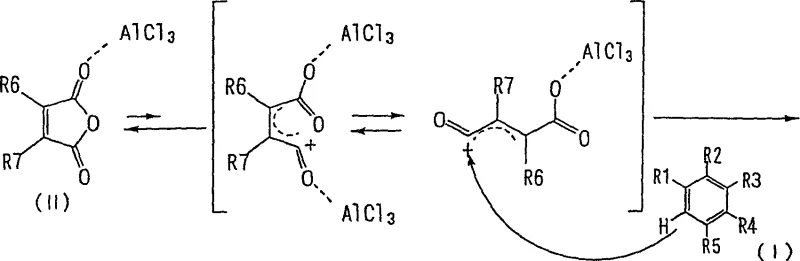
This mechanistic advantage ensures a much faster reaction rate and higher conversion of the starting aromatic hydrocarbon. Furthermore, the in-situ formation of the ester prevents the formation of aluminum carboxylate salts that typically complicate the workup in conventional acid syntheses. Instead, the product remains in the organic phase or precipitates cleanly upon quenching, facilitating a much simpler isolation procedure. The control over impurities is also enhanced because the shorter reaction time minimizes the opportunity for side reactions such as polymerization or over-acylation, which are common pitfalls in prolonged Friedel-Crafts processes. This precise control over the reaction trajectory is what enables the achievement of purity levels exceeding 99%, a critical specification for pharmaceutical grade intermediates where impurity profiles are strictly regulated.
How to Synthesize 4-Phenyl-4-Oxo-2-Butenoate Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the tandem reaction. The process begins with the preparation of a reaction mixture containing the aromatic substrate, maleic anhydride derivative, and the chosen alkylating agent in an inert solvent such as chlorobenzene or dichloromethane. The addition of the acid catalyst, typically aluminum chloride, must be performed under controlled conditions, usually at temperatures below 10°C, to manage the exotherm and prevent degradation of sensitive functional groups. The reaction is then allowed to proceed at mild temperatures, typically between 10°C and 50°C, for a duration of 1 to 4 hours. Upon completion, the catalyst is decomposed using dilute acid, and the product is isolated through standard extraction and crystallization techniques, yielding the target ester in high purity.
- Prepare the reaction mixture by combining maleic anhydride derivatives, aromatic hydrocarbons, and a suitable alkylating agent such as diethyl sulfate in an inert solvent.
- Add an acid catalyst, preferably aluminum chloride, under controlled low-temperature conditions to initiate the acylation and prevent side reactions.
- Maintain the reaction temperature between 0°C and 80°C for 1 to 4 hours, allowing the alkylating agent to trap the intermediate and drive ester formation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot synthesis technology offers substantial strategic advantages for organizations managing the supply of fine chemical intermediates. The most immediate benefit is the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By eliminating the discrete esterification step and the associated isolation of the acid intermediate, manufacturers can significantly reduce the consumption of solvents, reagents, and energy. This streamlining of the process flow also reduces the physical footprint required for production, as fewer reactors and separation units are needed to achieve the same output volume. For procurement teams, this translates into a more resilient supply chain with lower vulnerability to bottlenecks caused by complex multi-step processing.
- Cost Reduction in Manufacturing: The elimination of the separate esterification stage removes the need for additional reagents, catalysts, and solvents specifically dedicated to that second step. Furthermore, the reduction in reaction time from nearly two days to just a few hours dramatically increases reactor turnover rates, allowing facilities to produce more batches within the same timeframe without capital investment in new equipment. The simplified workup procedure also reduces waste disposal costs, as there is less aluminum sludge and aqueous waste to treat compared to the conventional acid isolation method.
- Enhanced Supply Chain Reliability: Shorter cycle times inherently improve supply chain responsiveness. With a reaction time of only 4 hours compared to the conventional 50+ hours, manufacturers can react more quickly to fluctuations in demand. The robustness of the reaction, evidenced by high yields across various substrates including electron-rich and electron-deficient aromatics, ensures consistent output quality. This reliability minimizes the risk of batch failures and supply interruptions, providing downstream API manufacturers with a dependable source of critical intermediates.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind. The avoidance of steam distillation and complex aqueous extractions simplifies the scale-up process, reducing the engineering challenges associated with moving from pilot plant to commercial production. Additionally, the higher atom economy and reduced solvent usage contribute to a greener manufacturing profile, helping companies meet increasingly stringent environmental regulations and sustainability goals without compromising on production efficiency or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these details is crucial for R&D teams planning to integrate this methodology into their existing manufacturing platforms.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: The primary advantage is the consolidation of two distinct steps—acylation and esterification—into a single one-pot reaction. Conventional methods require isolating the acid intermediate followed by a separate esterification step, which significantly increases processing time and reduces overall yield.
Q: How does the addition of an alkylating agent improve reaction kinetics?
A: The alkylating agent reacts with the open-chain maleic acid derivative intermediate, forming an ester that cannot easily revert to the cyclic anhydride form. This shifts the chemical equilibrium towards the open-chain acyl cation, accelerating the Friedel-Crafts reaction rate significantly.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process is highly scalable. It eliminates the need for complex separation techniques like steam distillation required in older methods to remove aluminum hydroxide sludge, simplifying the workup and making it suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Phenyl-4-Oxo-2-Butenoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern chemical industry. Our team of expert chemists has extensively evaluated the technology described in CN1283612C and possesses the capability to implement this advanced one-pot acylation-esterification process on a commercial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4-phenyl-4-oxo-2-butenoate derivatives meets the highest standards required for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain. By leveraging our expertise, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate the tangible economic and operational advantages of switching to this superior manufacturing method. Let us help you engineer a more efficient and cost-effective supply chain for your critical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
