Industrial Scale-Up of 2-Hydroxyphenoxazin-3-one via Green Aqueous Oxidation Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways for synthesizing complex heterocyclic scaffolds. Patent CN111233781A introduces a groundbreaking methodology for the production of 2-hydroxyphenoxazin-3-one compounds, a class of molecules renowned for their potent biological activities including anti-tumor, antiviral, and anti-inflammatory properties. This innovation represents a paradigm shift from traditional organic solvent-based syntheses to a greener, aqueous-phase catalytic oxidation process. By leveraging the synergistic effects of natural gallic acid and transition metal salts under mild oxygen pressure, this technology addresses critical pain points in modern chemical manufacturing, specifically targeting the reduction of hazardous waste and the simplification of downstream processing. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable supply chain of high-value pharmaceutical intermediates while adhering to increasingly stringent global environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenoxazinone alkaloids has been plagued by inefficient multi-step sequences and the reliance on harsh reaction conditions. Early literature, such as the work by C. W. Bird, described routes originating from 2-nitrodiphenyl ether compounds which necessitated reductive cyclization and demethylation steps, resulting in low overall atom economy and poor industrial applicability. Furthermore, the oxidative condensation method reported by H.W. Wanzlick, while more direct, relied heavily on the use of stoichiometric amounts of potassium ferricyanide (K3Fe(CN)6) as an oxidant in acetic acid solvent. This conventional approach generates substantial quantities of salty wastewater and requires cumbersome post-treatment procedures to remove inorganic salts and residual solvents. The use of acetic acid also poses significant safety and cost challenges regarding solvent recovery and VOC emissions, making these legacy methods economically unviable for large-scale commercial production of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a catalytic system comprising gallic acid and metal salts to facilitate molecular oxygen oxidation directly in an aqueous phase. This approach completely eliminates the need for organic solvents, thereby drastically reducing the environmental footprint and operational costs associated with solvent purchase, recovery, and disposal. The reaction proceeds efficiently under mild temperatures ranging from 10°C to 60°C and moderate oxygen pressures of 0.1 to 1.0 MPa, utilizing air or pure oxygen as the terminal oxidant. This shift not only enhances the safety profile of the manufacturing process but also simplifies the isolation of the final product, as the absence of complex organic matrices allows for straightforward extraction and recrystallization. The versatility of this system is demonstrated by its compatibility with various substituted o-aminophenols and catechols, providing a robust platform for the diverse synthesis of phenoxazinone derivatives.
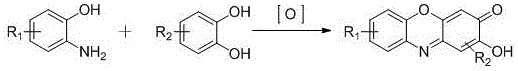
Mechanistic Insights into Gallic Acid-Metal Salt Catalyzed Oxidation
The core of this technological advancement lies in the unique catalytic cycle driven by the combination of gallic acid and transition metal salts such as Cu, Fe, Co, or Mn. Gallic acid, a naturally occurring polyphenol, acts as a primary catalyst that likely facilitates electron transfer processes, while the metal salt serves as a co-catalyst to activate molecular oxygen. This dual-catalyst system creates a highly active oxidative environment capable of driving the condensation of o-aminophenols and catechols into the tricyclic phenoxazinone core with exceptional efficiency. The mechanism avoids the formation of radical species that typically lead to polymerization or tar formation in non-catalyzed aerobic oxidations, ensuring a clean reaction profile. The patent data indicates that the molar ratio of substrates is tightly controlled between 1:0.9 and 1:1.2, optimizing the collision frequency of reactants while minimizing the accumulation of unreacted starting materials that could complicate purification.
From an impurity control perspective, the aqueous nature of the reaction medium plays a pivotal role in suppressing side reactions. In organic solvents like acetic acid, over-oxidation or non-selective coupling can occur, leading to difficult-to-remove byproducts. However, in the aqueous system described, the solubility differences between the organic product and inorganic byproducts allow for high selectivity. Experimental results from the patent demonstrate that products can be achieved with liquid chromatography purities reaching 98%, as seen in Example 1 where 2-hydroxyphenoxazin-3-one was isolated in 87% yield. The use of mild bases such as NaOH, Na2CO3, or K2CO3 further buffers the reaction environment, preventing the degradation of sensitive functional groups on the aromatic rings and ensuring the structural integrity of the final API intermediate.
How to Synthesize 2-Hydroxyphenoxazin-3-one Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and minimize catalyst loading. The process begins with the preparation of an aqueous slurry containing the o-aminophenol and catechol substrates, followed by the addition of gallic acid (0.01-10% molar ratio) and the selected metal salt co-catalyst. The reaction mixture is then basified and subjected to oxygen pressure while maintaining strict temperature control. Detailed standard operating procedures regarding specific substrate loadings, stirring rates, and work-up protocols are critical for reproducibility. For a comprehensive guide on executing this synthesis with optimal parameters, please refer to the standardized technical documentation below.
- Prepare the reaction mixture by combining o-aminophenol and catechol derivatives in water with gallic acid catalyst and metal salt co-catalyst.
- Add an inorganic base such as NaOH or K2CO3 to adjust the pH and facilitate the oxidative condensation.
- Introduce oxygen or air at 0.1-1.0 MPa pressure and maintain temperature between 10-60°C until reaction completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous oxidation technology offers profound strategic benefits beyond mere technical feasibility. The elimination of organic solvents and stoichiometric oxidants translates directly into a simplified supply chain architecture, reducing dependency on volatile petrochemical-derived solvents and specialized oxidizing agents. This simplification mitigates risks associated with raw material price fluctuations and availability, ensuring a more stable and predictable manufacturing timeline. Furthermore, the reduction in hazardous waste generation significantly lowers the burden on waste management infrastructure, allowing for faster throughput and reduced regulatory compliance costs. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The economic implications of replacing stoichiometric oxidants like potassium ferricyanide with catalytic molecular oxygen are substantial. By shifting to a catalytic regime, the consumption of expensive reagents is minimized, and the cost associated with treating heavy metal-laden or saline wastewater is drastically curtailed. Additionally, the use of water as the sole reaction medium removes the capital and operational expenditures linked to solvent recovery systems and explosion-proof infrastructure required for organic solvents. This leaner operational model allows for significant margin improvement in the production of complex pharmaceutical intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: The raw materials required for this process, specifically o-aminophenols and catechols, are commodity chemicals with well-established global supply networks. Unlike specialized reagents that may face sourcing bottlenecks, these precursors are readily available from multiple vendors, reducing single-source dependency risks. The robustness of the catalytic system, which tolerates a range of metal salts (acetates, carbonates, chlorides), provides further flexibility; if one specific metal salt faces supply constraints, the process can often be adapted to use an alternative salt of the same metal or a different metal entirely, ensuring uninterrupted production continuity for critical drug substances.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes thermal and safety limitations, particularly with exothermic oxidations in organic media. The aqueous nature of this reaction provides superior heat capacity and thermal stability, making the scale-up to 100 MT annual production volumes inherently safer and more manageable. Moreover, the alignment with Green Chemistry principles—specifically the use of safer solvents and design for energy efficiency—positions manufacturers favorably against tightening environmental regulations. This proactive compliance reduces the risk of future shutdowns due to regulatory changes and enhances the corporate sustainability profile, a key metric for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous oxidation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of using water as a solvent for phenoxazinone synthesis?
A: Using water eliminates the need for expensive and hazardous organic solvents like acetic acid, significantly reducing raw material costs and simplifying waste treatment processes associated with salty wastewater.
Q: Which metal salts are effective co-catalysts in this oxidation process?
A: The patent specifies that salts of Copper (Cu), Iron (Fe), Cobalt (Co), and Manganese (Mn), such as acetates, carbonates, chlorides, or sulfates, function effectively as co-catalysts to enhance reaction efficiency.
Q: How does this method improve product purity compared to traditional routes?
A: By utilizing molecular oxygen as the terminal oxidant instead of stoichiometric oxidants like potassium ferricyanide, the process minimizes side reactions and inorganic salt byproducts, yielding products with purity up to 98%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxyphenoxazin-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous oxidation technology described in CN111233781A for the production of high-value phenoxazinone intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific requirements of aqueous high-pressure reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the global pharmaceutical industry. We are committed to delivering not just a product, but a reliable, compliant, and cost-effective supply solution.
We invite forward-thinking R&D and procurement leaders to collaborate with us to optimize their supply chains for phenoxazinone derivatives. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our advanced manufacturing capabilities can support your next-generation drug development programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
