Advanced Synthesis of Hexa-Alkoxy Triphenylene Intermediates for Commercial OLED Production
Advanced Synthesis of Hexa-Alkoxy Triphenylene Intermediates for Commercial OLED Production
The rapid evolution of the organic electronics sector demands intermediates of exceptional purity and structural precision, particularly for applications in organic light-emitting diodes (OLEDs) and photovoltaic devices. Patent CN103265436A introduces a transformative methodology for synthesizing 1,5,9-trinitro-2,3,6,7,10,11-hexa-alkoxy triphenylene, a critical precursor for triazatriangulene-based photoelectrical materials. This innovation addresses long-standing challenges in the nitration of electron-rich polycyclic aromatic hydrocarbons, shifting away from hazardous traditional reagents toward a more controlled, silver-mediated system. For R&D directors and procurement specialists in the electronic chemical supply chain, this patent represents a pivotal opportunity to optimize the manufacturing of high-value display materials. The strategic importance of this intermediate lies in its subsequent conversion into rigid, planar heterocyclic systems that exhibit superior charge transport properties.
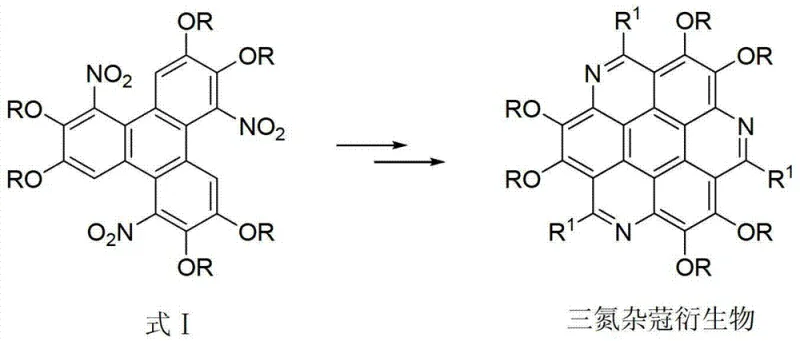
The core value proposition of this technology is its ability to deliver high-purity intermediates through a pathway that is inherently safer and more efficient than legacy methods. By utilizing a combination of metal nitrates and sulfonyl chlorides, the process achieves selective nitration at the 1,5,9-positions of the triphenylene core without degrading the sensitive alkoxy substituents. This selectivity is paramount for maintaining the electronic properties of the final material. As the global demand for high-resolution displays and flexible electronics grows, securing a reliable supply chain for such specialized aromatic intermediates becomes a competitive advantage. The following analysis dissects the technical merits and commercial implications of adopting this novel synthetic route for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrated triphenylene derivatives has relied heavily on the use of nitrosonitric acid or mixed acid systems involving concentrated sulfuric and nitric acids. These conventional approaches are fraught with significant technical and operational drawbacks that hinder efficient commercial manufacturing. The primary issue is the harsh reaction environment, which often leads to the oxidation of the electron-rich alkoxy groups on the triphenylene ring, resulting in complex impurity profiles that are difficult to separate. Furthermore, literature reports indicate that these traditional methods typically achieve product yields as low as 28%, meaning that nearly three-quarters of the valuable starting material is lost to side reactions or degradation. This low atom economy not only drives up raw material costs but also generates substantial quantities of acidic waste, posing severe environmental compliance challenges and increasing the burden on wastewater treatment facilities.
The Novel Approach
In stark contrast, the method disclosed in patent CN103265436A utilizes a mild nitrating system composed of a metal nitrate (such as silver nitrate or mercury pernitrate) and an alkyl or aryl sulfonyl chloride. This reagent combination generates the active nitrating species in situ under much gentler conditions, typically between 30°C and 100°C, avoiding the extreme acidity that plagues traditional routes. The visual representation of the core transformation highlights the structural integrity maintained during this process.
By employing this novel reagent system, the process achieves a dramatic improvement in yield, with optimized embodiments reporting yields up to 76%. This substantial increase in efficiency directly correlates to a reduction in the cost of goods sold (COGS) and a decrease in the volume of chemical waste generated per kilogram of product. Additionally, the absence of strong mineral acids during the reaction phase simplifies the downstream workup, as there is no need for extensive neutralization steps that can introduce inorganic salt contaminants. The versatility of the method is further demonstrated by its compatibility with various solvents, including trichloromethane and methylene dichloride, allowing manufacturers to select conditions that best fit their existing infrastructure and safety protocols.
Mechanistic Insights into Silver-Mediated Electrophilic Nitration
The success of this synthetic route relies on the unique mechanistic interaction between the metal nitrate and the sulfonyl chloride. In this system, the sulfonyl chloride acts as a dehydrating agent and a Lewis acid activator, facilitating the generation of the nitronium ion (NO2+) or a related electrophilic nitrate species from the silver nitrate. Unlike free nitric acid, which exists in a highly dissociated and aggressive state, this activated complex delivers the nitro group with greater specificity to the activated positions of the triphenylene ring. The electron-donating alkoxy groups direct the electrophilic attack to the 1,5,9-positions, and the mild nature of the reagent prevents over-nitration or oxidative damage to the aromatic core. This controlled reactivity is essential for preserving the optical and electronic characteristics required for high-performance OLED materials.
From an impurity control perspective, the mechanism offers distinct advantages. Traditional strong acid nitration often leads to ipso-substitution or cleavage of the ether bonds, generating phenolic by-products that are structurally similar to the target molecule and difficult to remove. The silver-mediated pathway minimizes these side reactions, resulting in a cleaner crude reaction mass. The use of silver nitrate also allows for the precipitation of silver chloride by-products, which can be easily filtered off before the organic workup, thereby reducing the load on the purification columns. This mechanistic clarity provides R&D teams with a robust framework for troubleshooting and optimizing the process further, ensuring consistent batch-to-bquality that meets the stringent specifications of the electronics industry.
How to Synthesize 1,5,9-Trinitro-2,3,6,7,10,11-hexa-methoxy Triphenylene Efficiently
Implementing this synthesis requires careful attention to stoichiometry and reaction parameters to maximize the yield benefits described in the patent. The general procedure involves dissolving the hexa-alkoxy triphenylene substrate in a chlorinated solvent, followed by the sequential addition of the sulfonyl chloride and the metal nitrate. Maintaining the optimal molar ratio of 1:6:6 (substrate:nitrate:sulfonyl chloride) is critical for driving the reaction to completion without excessive reagent waste. The detailed standardized synthesis steps for this high-efficiency route are provided below.
- Dissolve 2,3,6,7,10,11-hexa-methoxy triphenylene in an organic solvent such as trichloromethane or methylene dichloride within a reaction flask.
- Add benzene sulfonyl chloride and silver nitrate to the solution, maintaining a molar ratio of substrate to nitrate to sulfonyl chloride of approximately 1: 6:6.
- Stir the reaction mixture at a temperature between 60°C and 70°C for approximately 48 hours, followed by aqueous workup and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible benefits that extend beyond mere technical feasibility. The shift from a low-yield, high-hazard process to a high-yield, moderate-condition process fundamentally alters the cost structure and risk profile of producing these specialized intermediates. By eliminating the reliance on corrosive strong acids and improving material throughput, manufacturers can achieve significant operational efficiencies. These improvements translate directly into a more resilient supply chain capable of meeting the rigorous demands of the global display and semiconductor markets.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the drastic improvement in reaction yield. Moving from a historical baseline of roughly 28% to yields exceeding 70% effectively more than doubles the output from the same amount of raw starting material. This enhancement in atom economy significantly lowers the raw material cost per kilogram of the final intermediate. Furthermore, the elimination of strong acid reagents reduces the costs associated with corrosion-resistant equipment maintenance and the disposal of hazardous acidic waste streams. The simplified purification process also lowers solvent consumption and labor hours required for chromatography, contributing to a leaner overall manufacturing cost structure.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents such as silver nitrate and benzene sulfonyl chloride ensures a robust supply chain. Unlike specialized nitrating mixtures that may have short shelf-lives or require on-site generation, these components are standard fine chemicals with reliable global availability. The moderate reaction temperatures (60-70°C) reduce the energy intensity of the process compared to high-temperature alternatives, mitigating risks associated with thermal runaways. This operational stability ensures consistent production schedules and reduces the likelihood of batch failures that could disrupt downstream material supplies for OLED panel manufacturers.
- Scalability and Environmental Compliance: Scaling this process to multi-kilogram or tonnage levels is facilitated by the absence of gaseous nitrogen oxide emissions typically associated with nitric acid nitration. The solid by-products, primarily silver chloride, can be recovered and potentially recycled, aligning with green chemistry principles and reducing the environmental footprint. The process operates in common organic solvents that are easily managed in standard pharmaceutical and fine chemical reactors, removing the need for specialized high-pressure or exotic alloy vessels. This ease of scale-up allows suppliers to rapidly respond to increasing market demand for organic electronic materials without prohibitive capital expenditure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitration technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What are the primary advantages of this nitration method over traditional nitrosonitric acid processes?
A: The patented method utilizes silver nitrate and sulfonyl chloride instead of harsh nitrosonitric acid, eliminating the need for strong acid reagents. This results in significantly higher product yields (up to 76% versus 28%) and reduces environmental pollution caused by nitrogen oxide emissions.
Q: Can this synthesis be scaled for industrial production of OLED intermediates?
A: Yes, the reaction conditions are moderate (30-100°C) and do not require extreme pressures or corrosive strong acids, making the process highly suitable for commercial scale-up in standard stainless steel reactors with appropriate safety measures for silver salts.
Q: What is the purity profile of the resulting triphenylene derivative?
A: The method produces a faint yellow solid that can be purified via standard silica gel column chromatography. The absence of strong acid by-products simplifies the impurity profile, facilitating the attainment of high-purity specifications required for electronic grade materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5,9-Trinitro-2,3,6,7,10,11-hexa-alkoxy Triphenylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the performance of next-generation optoelectronic devices. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of regio-isomers and oxidative by-products that could compromise device efficiency.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in this patented nitration chemistry, we can help you optimize your supply chain for organic photoelectrical materials. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects, ensuring a secure and cost-effective supply of these vital electronic chemicals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
