Scalable Synthesis of Malachite Green Borate Precursors for High-Performance AIE Fluorescent Dyes
Scalable Synthesis of Malachite Green Borate Precursors for High-Performance AIE Fluorescent Dyes
The rapid evolution of optoelectronic materials has placed immense pressure on R&D teams to discover fluorophores that defy the limitations of traditional aggregation-caused quenching (ACQ). In response to this critical industry challenge, recent intellectual property developments, specifically patent CN115991717A, have introduced a groundbreaking class of molecules known as malachite green borates. These compounds serve as versatile precursors for aggregation-induced emission (AIE) luminophores, offering a structural solution to the efficiency losses typically seen in solid-state lighting and display technologies. By integrating a twisted, propeller-like molecular architecture, these borate esters effectively restrict intramolecular motion and prevent detrimental pi-pi stacking interactions.
This technological leap provides a reliable OLED material supplier with the necessary chemical building blocks to engineer next-generation emissive layers. The core innovation lies in the conversion of conventional planar dye structures into three-dimensional AIE-active scaffolds through a streamlined synthetic pathway. As illustrated in the general structural formula below, the molecule features a central carbon atom bonded to three aromatic rings, one of which is functionalized with a pinacol boronate ester group, enabling further diversification through cross-coupling reactions.
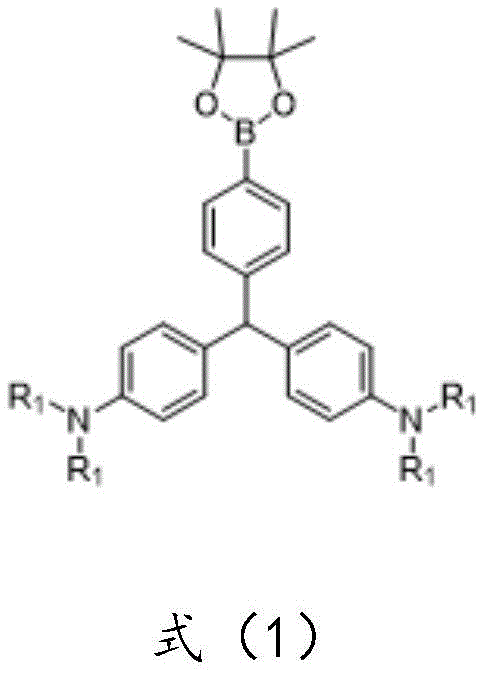
The strategic placement of the boron functionality allows for extensive post-synthetic modification, making this scaffold invaluable for creating a library of high-performance electronic chemicals. For procurement managers and supply chain heads, understanding the robustness of this synthesis is key to securing long-term material availability for display manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the design of fluorescent dyes for organic light-emitting diodes (OLEDs) and bio-imaging has been plagued by the ACQ effect, where fluorophores lose their emissive properties when concentrated or in the solid state due to strong intermolecular interactions. Traditional synthetic routes often yield planar molecules that pack tightly in crystal lattices, facilitating non-radiative decay pathways that drastically reduce quantum yields. Furthermore, modifying these conventional dyes to prevent aggregation often requires complex, multi-step syntheses involving bulky substituents that are difficult to source and expensive to install. This creates a significant bottleneck in cost reduction in electronic chemical manufacturing, as the yield penalties and purification challenges associated with rigid planar systems drive up the final price per gram of the active material.
The Novel Approach
The methodology disclosed in the patent data presents a paradigm shift by utilizing a malachite green derivative as a universal building block for AIEgens. Instead of struggling to force planar molecules to twist, this approach starts with a inherently non-planar triphenylmethane backbone that naturally adopts a propeller shape. The novel synthetic route involves a direct borylation of the bromomalachite green intermediate, introducing a reactive handle for Suzuki coupling without disrupting the crucial twisted geometry. This strategy not only preserves the AIE characteristics but also enhances the solubility and processability of the resulting dyes. The versatility of this approach is evident in the ability to generate various derivatives by coupling with different aryl halides, as shown in the generalized reaction scheme below, allowing for fine-tuning of emission wavelengths and energy levels.
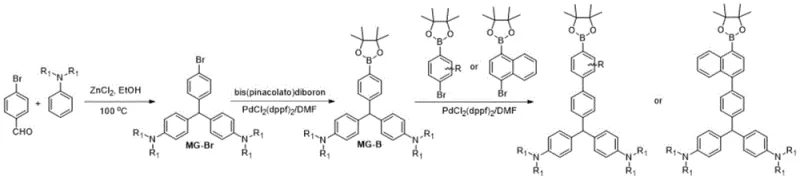
By adopting this modular design, manufacturers can rapidly prototype new emissive materials with tailored properties, significantly reducing the lead time for high-purity electronic chemicals to reach the market. The elimination of complex structural distortion steps simplifies the overall process flow, directly addressing the scalability concerns of supply chain负责人.
Mechanistic Insights into Zinc Chloride Catalyzed Condensation and Pd-Catalyzed Borylation
The synthesis of malachite green borate proceeds through a sophisticated two-stage mechanism that ensures high regioselectivity and yield. The first stage involves the condensation of 4-bromobenzaldehyde with N,N-disubstituted aniline, catalyzed by anhydrous zinc chloride in an ethanol solvent system at elevated temperatures ranging from 80 to 100°C. In this electrophilic aromatic substitution, the zinc chloride acts as a Lewis acid, activating the carbonyl group of the aldehyde towards nucleophilic attack by the electron-rich aniline derivatives. This step is critical for establishing the triphenylmethane core, and the use of a 1:3 molar ratio of aldehyde to aniline drives the equilibrium towards the formation of the leuco base, which is subsequently oxidized or stabilized to form the bromomalachite green intermediate (MG-Br).
Following the isolation of the bromo-intermediate, the second stage employs a palladium-catalyzed Miyaura borylation to install the pinacol boronate ester. This transformation utilizes bis(pinacolato)diboron as the boron source and [1,1'-bis(diphenylphosphino)ferrocene]palladium(II) dichloride (PdCl2(dppf)2) as the catalyst in dimethylformamide (DMF). The mechanism involves the oxidative addition of the aryl bromide to the Pd(0) species, followed by transmetallation with the diboron reagent assisted by potassium acetate, and finally reductive elimination to release the desired borate ester. This catalytic cycle is highly efficient and tolerant of the amine functionalities present in the molecule, ensuring that the final product retains its optical integrity. The detailed reaction pathway for the primary example is depicted below, highlighting the transition from the bromo-precursor to the final borate ester.
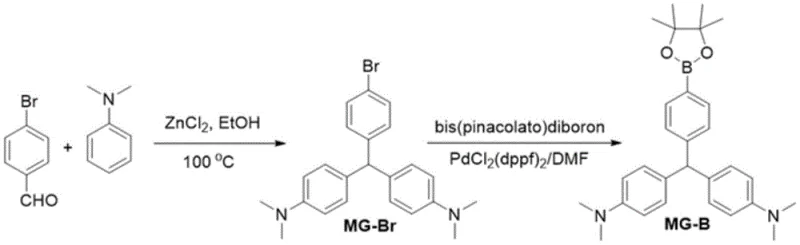
Understanding these mechanistic nuances is vital for R&D directors aiming to optimize impurity profiles, as controlling the stoichiometry of the diboron reagent and the activity of the palladium catalyst are key to minimizing homocoupling byproducts.
How to Synthesize Malachite Green Borate Efficiently
Implementing this synthesis in a pilot or production plant requires strict adherence to the optimized parameters defined in the patent to ensure consistent quality and safety. The process leverages commodity chemicals that are readily available in the global supply chain, mitigating risks associated with raw material scarcity. Operators must maintain precise temperature control during the condensation phase to prevent side reactions, while the borylation step demands an inert atmosphere to protect the sensitive palladium catalyst from oxidation. The following guide outlines the standardized operational procedure derived from the experimental examples, serving as a blueprint for technical teams preparing for technology transfer.
- Condense 4-bromobenzaldehyde with N,N-disubstituted aniline using anhydrous zinc chloride in ethanol at 100°C to form the bromomalachite green intermediate.
- React the crude intermediate with bis(pinacolato)diboron and potassium acetate in DMF using a PdCl2(dppf)2 catalyst at 80°C.
- Purify the final malachite green borate product via silica gel column chromatography to ensure high purity for optical applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this malachite green borate synthesis route offers substantial strategic benefits for organizations managing the procurement of advanced electronic materials. The reliance on widely available starting materials such as 4-bromobenzaldehyde and N,N-dimethylaniline ensures a stable supply chain that is less susceptible to the volatility often seen with exotic heterocyclic building blocks. Furthermore, the reaction conditions are relatively mild, avoiding the need for cryogenic cooling or ultra-high pressure equipment, which translates to lower capital expenditure (CAPEX) for facility upgrades and reduced operational expenditure (OPEX) regarding energy consumption.
- Cost Reduction in Manufacturing: The streamlined two-step process eliminates the need for multiple protection and deprotection sequences often required in complex dye synthesis, thereby significantly reducing solvent usage and waste generation. By utilizing a robust palladium catalyst system that operates efficiently at moderate loadings, the process minimizes the cost associated with precious metal recovery, leading to a more economically viable production model for high-value OLED intermediates.
- Enhanced Supply Chain Reliability: Since the key reagents are bulk commodities produced by numerous global chemical suppliers, the risk of single-source dependency is drastically minimized. This diversity in sourcing options allows procurement managers to negotiate better terms and secure long-term contracts, ensuring uninterrupted production schedules for downstream display manufacturers who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, as demonstrated by the successful execution of the reaction on gram scales in the patent examples, with clear pathways to kilogram and tonne-scale production. The use of standard organic solvents like ethanol and DMF, which have well-established recycling protocols, facilitates compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste disposal.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled answers to common inquiries regarding the chemical properties and processing requirements of malachite green borate. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: What is the primary advantage of Malachite Green Borate over conventional fluorescent dyes?
A: Unlike conventional dyes that suffer from Aggregation-Caused Quenching (ACQ), Malachite Green Borate features a twisted propeller structure that enables Aggregation-Induced Emission (AIE), maintaining high fluorescence intensity even in solid states or aggregated forms.
Q: Can the synthesis of MG-B be scaled for industrial production?
A: Yes, the synthesis utilizes standard reaction conditions (80-100°C) and commercially available starting materials like 4-bromobenzaldehyde, making it highly suitable for commercial scale-up without requiring specialized cryogenic equipment.
Q: What catalysts are required for the borylation step?
A: The borylation step employs [1,1'-bis(diphenylphosphino)ferrocene]palladium(II) dichloride (PdCl2(dppf)2) as the catalyst, which facilitates efficient coupling under mild thermal conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Malachite Green Borate Supplier
As the demand for high-efficiency AIE materials continues to surge in the display and lighting sectors, partnering with an experienced chemical manufacturing partner is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of malachite green borate meets the exacting standards required for optoelectronic applications.
We invite you to collaborate with our technical procurement team to explore how this novel synthetic route can optimize your material costs and accelerate your product development cycles. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into the economic benefits of switching to this AIE precursor. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments tailored to your unique project requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
