Advanced One-Step Iodolactonization for High-Purity Beta-Iodobutenolide Manufacturing
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A pivotal advancement in this domain is documented in patent CN1328271C, which details a novel method for synthesizing beta-iodobutenolide, a critical structural motif found in numerous bioactive natural products and pharmaceutical intermediates. This technology represents a significant departure from conventional multi-step procedures, offering a streamlined one-pot iodolactonization strategy that operates under remarkably mild conditions. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for optimizing supply chains and reducing the overall cost of goods sold (COGS) for complex lactone derivatives. The core innovation lies in the direct utilization of 2,3-allenoate esters, bypassing the unstable free acid intermediates that have historically plagued this chemical transformation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of five-membered ring unsaturated lactones, such as beta-iodobutenolide, relied heavily on the iodocyclization of 2,3-allenoic acids. This classical approach necessitates a preliminary hydrolysis step where the corresponding 2,3-allenoate ester is converted into the free carboxylic acid using acid or base catalysis. However, this hydrolysis step is fraught with significant chemical challenges that negatively impact overall process efficiency and yield. Under the acidic or basic conditions required for hydrolysis, the sensitive 2,3-allenoate structure is prone to undesirable isomerization, often converting into stable alkyne by-products or generating other complex impurities. Consequently, the yield of the desired 2,3-allenoic acid is frequently compromised, creating a bottleneck that propagates through the subsequent cyclization step. Furthermore, the isolation and purification of these unstable free acids add unnecessary operational complexity, requiring stringent control over reaction parameters and often resulting in substantial material loss before the final ring-closing event can even occur.
The Novel Approach
In stark contrast to the cumbersome traditional pathways, the methodology outlined in CN1328271C introduces a direct iodolactonization of 2,3-allenoate esters, effectively eliminating the problematic hydrolysis step entirely. By reacting the ester directly with iodine in a mixed solvent system comprising water and an organic co-solvent, the process achieves cyclization in a single operational unit. This approach not only simplifies the workflow but also dramatically improves the atom economy and overall yield of the transformation. The reaction is versatile, accommodating a wide range of substituents including alkyl, aryl, and benzyl groups at various positions on the allene backbone, making it a robust platform technology for diverse chemical libraries. The ability to proceed directly from the stable ester precursor to the final lactone product without isolating reactive intermediates signifies a major leap forward in process chemistry, offering a cleaner, faster, and more reliable route for industrial production.
Mechanistic Insights into Iodine-Participated Cyclization
The success of this novel synthetic route hinges on the unique reactivity of iodine within a water-organic biphasic or mixed solvent environment. Mechanistically, the reaction initiates with the electrophilic attack of molecular iodine on the electron-rich allene system of the 2,3-allenoate ester. Unlike traditional methods that might require harsh Lewis acids or elevated temperatures to activate the substrate, the presence of water in the solvent mixture appears to facilitate the generation of the active iodinating species while simultaneously stabilizing the transition state. The nucleophilic carbonyl oxygen of the ester group then attacks the activated allene-iodine complex, leading to the formation of the five-membered lactone ring and the incorporation of the iodine atom at the beta-position. This intramolecular cyclization is highly regioselective, ensuring that the iodine atom is positioned precisely to allow for further downstream functionalization, such as palladium-catalyzed cross-coupling reactions which are vital for building complex pharmaceutical architectures.
From an impurity control perspective, the use of a water-containing solvent system plays a crucial role in suppressing side reactions. In strictly anhydrous organic solvents, competing pathways such as polymerization or alternative halogenation patterns might dominate. However, the specific solvation effects provided by the water-organic mixture help to moderate the reactivity of the iodine, preventing over-iodination or degradation of the sensitive allene moiety. Additionally, the reaction produces a single major product with high selectivity, which greatly simplifies the downstream purification process. For quality control teams, this means that the crude reaction mixture typically requires only standard workup procedures, such as quenching with sodium thiosulfate to remove excess iodine followed by extraction, to achieve high purity levels. This inherent cleanliness of the reaction profile reduces the burden on analytical resources and minimizes the risk of carrying over difficult-to-remove impurities into the final API intermediate.
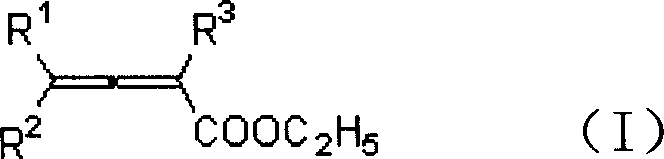
How to Synthesize Beta-Iodobutenolide Efficiently
Implementing this synthesis on a laboratory or pilot scale requires careful attention to solvent ratios and reagent stoichiometry to maximize the benefits described in the patent literature. The process is designed to be operationally simple, avoiding the need for specialized equipment like gloveboxes or high-pressure reactors. Detailed below is the strategic framework for executing this transformation, focusing on the critical parameters that ensure reproducibility and high yield. Operators should note that while the reaction is robust, optimization of the organic co-solvent (such as acetonitrile, ethanol, or DMF) may be necessary depending on the solubility profile of the specific allenoate substrate being used. The standardized protocol ensures that the reaction proceeds smoothly at ambient temperatures, aligning with green chemistry principles by reducing energy consumption.
- Prepare a reaction mixture containing water, an organic solvent such as acetonitrile, and the 2,3-allenoate ester substrate in a suitable vessel.
- Add elemental iodine to the stirred solution at room temperature, maintaining a molar ratio of iodine to ester between 1: 1 and 3:1.
- Stir the reaction mixture for 0.5 to 24 hours, then quench with water and treat with saturated sodium thiosulfate to remove excess iodine before purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic advantages that extend beyond mere technical elegance. The elimination of the hydrolysis step translates directly into a reduction in raw material consumption, as there is no longer a need for additional hydrolysis reagents or the associated neutralization agents. Furthermore, the simplified workflow reduces the total processing time, allowing manufacturing facilities to increase throughput without expanding physical infrastructure. The use of common, inexpensive solvents like acetonitrile and water, combined with the avoidance of exotic catalysts, ensures that the variable costs associated with production remain low and predictable. This stability in input costs is crucial for long-term contract negotiations and budget forecasting in the volatile fine chemical market.
- Cost Reduction in Manufacturing: The most significant economic driver here is the telescoping of two distinct chemical steps into one. By removing the hydrolysis and isolation of the free acid, manufacturers save on labor, energy, and solvent usage associated with that intermediate stage. The reaction operates at room temperature, which drastically cuts down on heating and cooling costs compared to processes requiring reflux or cryogenic conditions. Additionally, the high yield range of 65-91% reported in the patent examples indicates minimal material waste, ensuring that expensive starting materials are converted efficiently into valuable product. The simplicity of the workup, involving basic aqueous quenching and extraction, further lowers the operational expenditure related to waste treatment and solvent recovery.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. Since the process does not require strictly anhydrous or oxygen-free environments, it is less susceptible to failures caused by minor deviations in plant operations or equipment integrity. The starting materials, 2,3-allenoate esters, are generally stable and commercially accessible, reducing the risk of supply disruptions associated with handling unstable free acids. The short reaction time, ranging from 0.5 to 24 hours, allows for flexible scheduling and faster turnaround times, enabling suppliers to respond more敏捷 ly to fluctuating market demands. This agility is a key differentiator when serving just-in-time manufacturing models prevalent in the pharmaceutical industry.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage quantities is facilitated by the use of benign solvent systems and the absence of hazardous exotherms typically associated with strong acid hydrolysis. The environmental footprint is significantly reduced due to the lower volume of waste generated per kilogram of product, aligning with increasingly stringent global environmental regulations. The use of water as a co-solvent not only improves safety by reducing flammability risks but also simplifies effluent treatment protocols. For companies aiming to meet sustainability goals, adopting this greener synthetic route demonstrates a commitment to responsible manufacturing practices, which can enhance brand reputation and compliance standing with regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodolactonization technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing production portfolios. The answers reflect the balance between theoretical potential and practical application observed in the reported examples.
Q: What are the primary advantages of this iodolactonization method over traditional hydrolysis routes?
A: This method eliminates the need for a separate hydrolysis step to generate the free acid, which traditionally suffers from low yields due to isomerization into alkynes. By reacting the ester directly, the process achieves higher yields (65-91%) under milder conditions.
Q: Does this synthesis require strict anhydrous or oxygen-free conditions?
A: No, one of the key operational benefits is that the reaction does not require anhydrous or oxygen-free environments. The use of a water-organic mixed solvent system makes the process more robust and easier to handle on an industrial scale.
Q: What is the typical reaction time and temperature profile for this cyclization?
A: The reaction proceeds efficiently at room temperature, typically requiring between 0.5 to 24 hours depending on the specific substituents on the allenoate ester, significantly reducing energy consumption compared to heated processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Iodobutenolide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN1328271C for producing high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise to adapt and optimize such routes for large-scale commercial production, ensuring that our clients benefit from both innovation and reliability. Our facilities are equipped to handle complex organic syntheses, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-iodobutenolide meets the exacting standards required for downstream drug synthesis, providing a secure foundation for your supply chain.
We invite you to collaborate with us to explore how this efficient synthesis can drive value for your specific projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our capabilities align with your strategic sourcing goals. Let us help you engineer a more resilient and cost-effective supply chain for your critical chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
