Advanced Synthesis of Fluorene-Based Non-Fullerene Acceptors for Next-Gen Organic Photovoltaics
Advanced Synthesis of Fluorene-Based Non-Fullerene Acceptors for Next-Gen Organic Photovoltaics
The rapid evolution of the organic photovoltaic (OPV) sector demands electron acceptor materials that surpass the limitations of traditional fullerene derivatives. Patent CN112608309B introduces a significant breakthrough in this domain by disclosing a class of non-condensed ring organic small molecular materials featuring a fluorene ring group. These materials, specifically designated as Flu-R1 and BTT6IC-R2, represent a strategic shift towards A-D-A (Acceptor-Donor-Acceptor) architectures that offer superior tunability of energy levels and enhanced light absorption capabilities. As a leading entity in fine chemical manufacturing, we recognize the immense potential of these compounds to drive the next generation of flexible and translucent solar devices. The patent details a robust synthetic methodology that leverages well-established cross-coupling reactions to construct complex conjugated systems with high precision.
The core innovation lies in the molecular engineering strategy where a 2,7-disubstituted-9,9-dioctylfluorene serves as the central donor unit, flanked by thiophene linkers and terminated with strong electron-withdrawing cyanoindanone groups. This structural arrangement facilitates effective intramolecular charge transfer, a critical parameter for high power conversion efficiency. Furthermore, the optional introduction of fluorine atoms at the terminal positions allows for fine-tuning of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) levels, thereby optimizing the open-circuit voltage of the resulting devices. For procurement specialists and R&D teams seeking reliable organic photovoltaic material suppliers, understanding the synthesis and scalability of these specific architectures is paramount for securing a stable supply chain for future electronic chemical manufacturing.
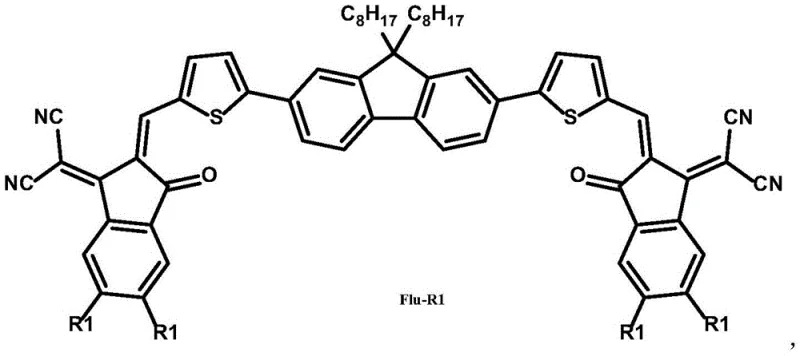
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the organic photovoltaic industry has relied heavily on fullerene-based acceptors such as PCBM. While these materials offered reasonable electron mobility, they suffered from intrinsic drawbacks that hindered the commercial viability of OPV technology. Fullerene derivatives typically exhibit weak absorption in the visible region, limiting the short-circuit current density of the devices. Moreover, their spherical geometry often leads to morphological instability within the bulk heterojunction active layer, causing phase separation over time and degrading device performance. From a synthesis perspective, functionalizing fullerenes to improve compatibility often involves complex, low-yielding reactions that are difficult to scale. The rigidity of the fullerene cage also limits the ability to tune energy levels through simple structural modifications, forcing manufacturers to rely on blending strategies that add complexity to the formulation process.
The Novel Approach
The methodology outlined in CN112608309B presents a compelling alternative by utilizing a planar, non-condensed ring system that addresses these fundamental issues. By employing a fluorene core, the material gains inherent photo-thermal stability due to the rigid planar interconnected benzene units. The synthetic route allows for the modular insertion of electron-deficient units like benzothiadiazole, which significantly enhances the electron affinity of the molecule without compromising solubility. The introduction of thiophene units expands the pi-conjugation system, effectively narrowing the bandgap and extending light absorption into the near-infrared region. Crucially, the presence of alkyl side chains on the fluorene and thiophene moieties ensures excellent solubility in common processing solvents, enabling the fabrication of high-quality thin films via solution processing techniques. This approach not only improves device efficiency but also simplifies the manufacturing workflow, offering a clear path toward cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Suzuki-Miyaura and Knoevenagel Coupling Strategies
The synthesis of these advanced materials relies on a sequence of palladium-catalyzed cross-coupling reactions, primarily the Suzuki-Miyaura coupling, which is renowned for its tolerance to functional groups and mild reaction conditions. In the initial stage, 2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-9,9-dioctylfluorene reacts with 5-bromothiophene-2-carbaldehyde. This step is critical as it constructs the central D-A-D framework. The use of tetrakis(triphenylphosphine)palladium(0) as the catalyst, in conjunction with potassium carbonate as the base, facilitates the transmetallation and reductive elimination steps necessary to form the carbon-carbon bonds between the fluorene core and the thiophene aldehyde units. The reaction is typically conducted in a biphasic system of tetrahydrofuran and water under an inert nitrogen atmosphere to prevent oxidation of the sensitive organometallic intermediates.
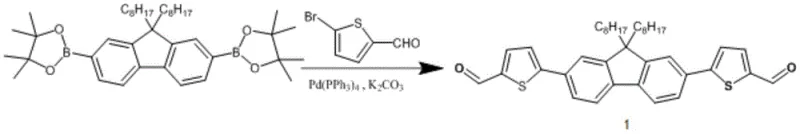
Following the construction of the central core, the final functionalization is achieved through a Knoevenagel condensation reaction. This step involves the reaction of the dialdehyde intermediate with active methylene compounds, specifically 3-(dicyanomethylene)indanone or its fluorinated analog. The mechanism proceeds via the deprotonation of the active methylene group by a base such as pyridine, generating a nucleophilic carbanion that attacks the carbonyl carbon of the aldehyde. Subsequent dehydration yields the conjugated double bond linkage that extends the pi-system to the terminal electron-withdrawing groups. This condensation is pivotal for establishing the push-pull electronic character of the molecule. The patent highlights that controlling the stoichiometry and reaction temperature (typically around 65°C) is essential to minimize side reactions and ensure high conversion rates. The resulting materials exhibit strong absorption coefficients and favorable energy level alignment, making them ideal candidates for high-performance OPV active layers.
How to Synthesize Flu-R1 and BTT6IC-R2 Efficiently
The preparation of these non-condensed ring organic small molecules involves a multi-step sequence that requires precise control over reaction parameters to ensure high purity and yield. The process begins with the synthesis of key intermediates via palladium-catalyzed coupling, followed by formylation and final condensation. Each step necessitates rigorous exclusion of oxygen and moisture to maintain catalyst activity and prevent degradation of the conjugated system. The detailed standardized synthetic steps for producing these high-purity organic photovoltaic materials are outlined in the guide below, providing a roadmap for laboratory scale-up and process optimization.
- Perform Suzuki-Miyaura coupling between 2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-9,9-dioctylfluorene and 5-bromothiophene-2-carbaldehyde using Pd(PPh3)4 catalyst to form the central aldehyde intermediate.
- Execute Knoevenagel condensation by reacting the central aldehyde intermediate with 3-(dicyanomethylene)indanone or its fluorinated derivative in chloroform with pyridine catalysis.
- Purify the final non-condensed ring small molecule receptor material via column chromatography followed by recrystallization from a chloroform-n-hexane system to ensure electronic grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to non-fullerene acceptors like those described in CN112608309B offers significant strategic advantages beyond mere performance metrics. The synthetic route utilizes commercially available starting materials such as fluorene derivatives, thiophenes, and indanones, which reduces dependency on exotic or single-source precursors. This abundance of raw materials contributes to a more resilient supply chain, mitigating the risks associated with geopolitical disruptions or raw material shortages. Furthermore, the reactions employed, such as Suzuki and Knoevenagel condensations, are well-understood industrial processes that can be readily scaled from gram to kilogram quantities using standard reactor setups. This scalability ensures that production lead times can be minimized, supporting the rapid deployment of new OPV technologies.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive and difficult-to-remove fullerene derivatives, replacing them with planar organic molecules that are cheaper to synthesize on a large scale. The use of efficient palladium catalysts, which can be recovered and recycled in industrial settings, further drives down the cost of goods sold. Additionally, the high solubility of the final products reduces the need for aggressive high-temperature processing or specialized solvents, lowering energy consumption and waste disposal costs during device fabrication. The overall process design prioritizes atom economy and operational simplicity, translating directly into substantial cost savings for mass production.
- Enhanced Supply Chain Reliability: By relying on robust C-C bond-forming reactions that are less sensitive to minor fluctuations in reaction conditions compared to fullerene functionalization, the manufacturing process achieves higher consistency and batch-to-batch reproducibility. The availability of diverse building blocks, such as fluorinated and non-fluorinated end groups, allows for flexible production scheduling based on market demand for specific performance characteristics. This flexibility ensures that supply chain managers can respond quickly to changing customer requirements without significant retooling or long qualification periods, thereby enhancing overall supply chain agility and reliability.
- Scalability and Environmental Compliance: The purification methods described, involving column chromatography followed by recrystallization, are amenable to large-scale implementation using continuous chromatography or crystallization tanks. The solvents used, such as chloroform and hexane, are standard industrial chemicals with established recovery and recycling protocols, minimizing environmental impact. The avoidance of heavy metal contaminants in the final product, achieved through rigorous purification, ensures compliance with strict electronic industry standards regarding hazardous substances. This focus on green chemistry principles and scalable purification makes the technology highly attractive for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these fluorene-based non-condensed ring materials. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating these materials into your existing product development pipelines.
Q: What are the key structural advantages of the Flu-R1 and BTT6IC-R2 materials described in CN112608309B?
A: These materials utilize a rigid 9,9-dioctylfluorene core which provides photo-thermal stability. The introduction of benzothiadiazole units enhances electron-deficiency, while terminal fluorine atoms effectively adjust energy levels and induce a red-shift in absorption, crucial for maximizing photon harvesting in the visible spectrum.
Q: How does the synthesis route address the solubility issues common in organic semiconductors?
A: The synthetic design incorporates long alkyl side chains, specifically octyl groups on the fluorene core and hexyl chains on the thiophene units in the BTT series. These lipophilic chains significantly improve solubility in common organic solvents like chlorobenzene and chloroform, facilitating solution-processable film formation for device fabrication.
Q: What purification methods are recommended to achieve the purity required for OPV applications?
A: The patent specifies a rigorous purification protocol involving initial column chromatography to remove catalyst residues and by-products, followed by a critical recrystallization step using a chloroform-n-hexane solvent system. This dual-stage purification is essential for removing trace impurities that could act as charge traps in the active layer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Non-Condensed Ring Organic Small Molecule Material Supplier
As the global demand for high-efficiency organic photovoltaics continues to surge, the need for a dependable partner capable of delivering complex electronic chemicals at scale has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the stringent purity specifications required for OPV active layers, utilizing rigorous QC labs to verify every batch against exacting standards. We understand that the performance of your solar devices depends critically on the quality of the raw materials, and we are committed to providing the consistency and reliability that your R&D and production teams demand.
We invite you to collaborate with us to optimize your supply chain for next-generation solar materials. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to our deep expertise in process chemistry and scale-up engineering. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for Flu-R1, BTT6IC-R2, or other custom organic semiconductor structures. Let us help you accelerate your path to commercialization with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
