Transforming Difenoconazole Waste into High-Value 3,4'-Dichlorodiphenyl Ether via Novel Catalytic Routes
The global fine chemical industry is increasingly pivoting towards circular economy models, where waste streams from established processes are repurposed into high-value intermediates. Patent CN112707799A introduces a groundbreaking methodology for synthesizing 3,4'-dichlorodiphenyl ether, a critical building block for pharmaceuticals and agrochemicals, directly from difenoconazole isomer waste. This innovation addresses a significant pain point in the production of triazole fungicides, where approximately 30% of the output consists of isomers that are traditionally difficult to separate and often incinerated as hazardous waste. By leveraging a sophisticated three-step sequence involving hydrolysis, haloform reaction, and decarboxylation, this technology transforms an environmental liability into a commercially viable asset. For R&D directors and procurement strategists, this represents a dual opportunity: securing a reliable source of high-purity intermediates while simultaneously aligning corporate sustainability goals with cost-reduction initiatives through waste valorization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 3,4'-dichlorodiphenyl ether typically rely on the nucleophilic substitution reaction between 1,3-dichlorobenzene and 4-chlorophenol. This classical approach necessitates the use of expensive catalysts such as basic copper carbonate and potassium carbonate in high-boiling solvents like N-methyl pyrrolidone. The reaction conditions are harsh, requiring temperatures upwards of 170°C for extended periods, which not only consumes substantial energy but also promotes the formation of complex byproduct profiles that are difficult to separate. Furthermore, the reliance on fresh petrochemical-derived starting materials exposes the supply chain to volatile raw material pricing and geopolitical instability. The downstream processing involves multiple distillation steps under reduced pressure to remove residual solvents and unreacted starting materials, leading to significant yield losses and generating large volumes of organic waste that require costly treatment. These factors collectively inflate the manufacturing cost and complicate the scale-up process for commercial production facilities.
The Novel Approach
In stark contrast, the method disclosed in CN112707799A utilizes difenoconazole isomer waste as the primary feedstock, effectively bypassing the need for expensive fresh halobenzenes. This route capitalizes on the existing carbon skeleton of the waste isomer, requiring only functional group transformations to achieve the target structure. The process operates under relatively milder conditions compared to traditional Ullmann-type couplings, particularly in the initial hydrolysis step which proceeds in an aqueous medium. By integrating waste utilization directly into the synthesis workflow, this approach drastically reduces the carbon footprint associated with raw material extraction and processing. The strategic shift from purchasing premium starting materials to utilizing low-cost waste streams offers a compelling economic argument for procurement managers seeking to optimize cost structures without compromising on the quality of the final pharmaceutical intermediates. Additionally, the simplified purification protocols inherent in this pathway enhance overall process efficiency.
Mechanistic Insights into the Three-Step Conversion Process
The core of this technology lies in a meticulously orchestrated three-step reaction sequence that ensures high selectivity and yield. The first stage involves an acid-catalyzed hydrolytic ring-opening of the difenoconazole isomer. In this step, the acetal moiety of the isomer is cleaved in the presence of strong mineral acids such as hydrochloric or sulfuric acid within an aqueous environment at temperatures ranging from 60°C to 120°C. This transformation yields [2-chloro-4-(4-chlorophenoxy)-phenyl]-2-[1,2,4]triazole-4-ethanone, a key ketone intermediate. The use of water as a solvent in this stage is particularly advantageous from a green chemistry perspective, eliminating the need for volatile organic compounds and simplifying the workup procedure. Precise control of the acid-to-isomer molar ratio is critical here; insufficient acid leads to incomplete conversion, while excessive acid offers no additional benefit and increases corrosion risks.

Following hydrolysis, the process advances to a haloform reaction, a classic organic transformation used to convert methyl ketones into carboxylic acids. The triazole-ethanone intermediate reacts with a hypohalite source, such as sodium hypochlorite, in the presence of an inorganic base like sodium hydroxide. This reaction occurs at controlled temperatures between -20°C and 50°C to manage the exothermic nature of the halogenation and cleavage steps. The mechanism involves the successive halogenation of the alpha-methyl group followed by nucleophilic attack by hydroxide, resulting in the cleavage of the carbon-carbon bond and the formation of the corresponding benzoate salt. Subsequent acidification precipitates 2-chloro-4-(4-chlorophenoxy)-benzoic acid. This step is crucial for removing the triazole moiety and establishing the carboxylic acid functionality required for the final decarboxylation.
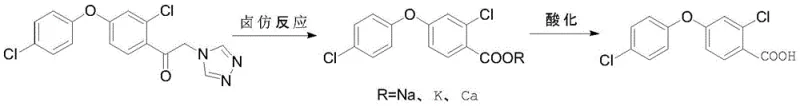
The final transformation is a copper-mediated decarboxylation, which removes the carboxyl group to yield the target 3,4'-dichlorodiphenyl ether. This reaction is conducted in quinoline, which serves as both a high-boiling solvent and a ligand stabilizer, with copper powder acting as the catalyst. The reaction temperature is maintained between 80°C and 250°C, a range optimized to drive the decarboxylation forward while minimizing thermal degradation of the sensitive diphenyl ether linkage. The copper catalyst facilitates the electron transfer necessary for the loss of carbon dioxide, regenerating the aromatic system. Optimization of the copper loading is essential; too little catalyst results in sluggish kinetics, whereas excessive amounts do not improve yield and complicate metal removal. This robust mechanistic pathway ensures the production of high-purity intermediates suitable for demanding applications in agrochemical intermediate synthesis.

How to Synthesize 3,4'-Dichlorodiphenyl Ether Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the preparation of the hydrolysis mixture, ensuring the correct stoichiometric balance of acid and water to facilitate complete ring opening without degrading the chlorophenoxy backbone. Following isolation of the ketone intermediate, the haloform reaction must be monitored closely to prevent over-halogenation or side reactions on the aromatic rings. Finally, the decarboxylation step demands rigorous temperature control and efficient removal of evolved carbon dioxide to drive the equilibrium towards the product. Detailed standard operating procedures regarding reagent addition rates, stirring speeds, and quenching protocols are essential for reproducible results at scale. For a comprehensive guide on the specific operational parameters and safety considerations, please refer to the standardized synthesis protocol below.
- Perform acid-catalyzed hydrolytic ring-opening of the difenoconazole isomer in water at 60-120°C to yield the triazole-ethanone intermediate.
- Execute a haloform reaction using inorganic base and hypohalite at -20 to 50°C, followed by acidification to isolate the benzoic acid derivative.
- Conduct decarboxylation using copper powder and quinoline solvent at 80-250°C to finalize the 3,4'-dichlorodiphenyl ether structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented method offers transformative benefits for supply chain resilience and cost management. By shifting the raw material base from virgin petrochemicals to industrial waste streams, manufacturers can insulate themselves from the volatility of global chemical markets. The availability of difenoconazole isomer waste is intrinsically linked to the production volume of a widely used fungicide, ensuring a consistent and abundant feedstock supply. This vertical integration of waste management and intermediate production creates a closed-loop system that significantly lowers the barrier to entry for new producers and enhances the competitiveness of existing ones. Furthermore, the reduction in hazardous waste disposal liabilities translates directly into improved bottom-line performance, making this route economically superior to conventional linear synthesis models.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic reduction in raw material costs. Since the primary feedstock is a waste byproduct, its acquisition cost is substantially lower than that of purified 1,3-dichlorobenzene or 4-chlorophenol. Additionally, the elimination of expensive transition metal catalysts often required in traditional coupling reactions, replacing them with inexpensive copper powder, further drives down the bill of materials. The aqueous nature of the first step also reduces solvent recovery costs, contributing to overall operational expenditure savings. These cumulative efficiencies allow for a more competitive pricing strategy in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Dependence on a single source of petrochemical precursors poses a risk to supply continuity. By diversifying the feedstock to include agricultural chemical waste, companies can mitigate the impact of supply disruptions in the oil and gas sector. The widespread production of difenoconazole globally ensures that the isomer waste stream is geographically distributed, allowing for regional sourcing strategies that reduce logistics lead times. This decentralization of raw material sources enhances the robustness of the supply chain, ensuring that production schedules for critical pharmaceutical intermediates are maintained even during periods of market turbulence.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as hydrolysis, filtration, and distillation that are standard in the fine chemical industry. The use of water as a primary solvent in the initial step aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. Moreover, by consuming hazardous waste as a resource, the process actively contributes to waste reduction targets, potentially qualifying for green manufacturing incentives. The simplified purification steps reduce the generation of secondary waste streams, facilitating easier compliance with environmental discharge standards and lowering the total cost of ownership for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this waste-to-value synthesis route. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: What is the primary advantage of using difenoconazole isomer waste for this synthesis?
A: The primary advantage is the conversion of hazardous waste, which typically constitutes about 30% of difenoconazole production byproducts, into a high-value organic synthetic raw material, significantly reducing disposal costs and raw material procurement expenses.
Q: What are the critical reaction conditions for the decarboxylation step?
A: The decarboxylation requires copper powder as a catalyst and quinoline as a solvent, operating within a temperature range of 80-250°C. Precise control of the copper-to-acid molar ratio (0.01-0.2:1) is essential to prevent incomplete reaction or excessive byproduct formation.
Q: How does this method compare to traditional diphenyl ether synthesis in terms of purity?
A: Experimental data from the patent indicates that this method achieves product purity exceeding 98% as analyzed by gas chromatography, which is comparable to or superior to traditional methods that often require extensive purification to remove unreacted halobenzenes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4'-Dichlorodiphenyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of sustainable and cost-effective synthesis routes in the modern chemical landscape. Our team of expert chemists has extensively evaluated the methodology described in CN112707799A and possesses the technical capability to scale this diverse pathway from laboratory benchtop quantities of 100 kgs to full commercial production capacities of 100 MT annually. We are committed to delivering high-purity 3,4'-dichlorodiphenyl ether that meets stringent purity specifications required for downstream pharmaceutical and agrochemical applications. Our state-of-the-art rigorous QC labs ensure that every batch undergoes comprehensive analysis to guarantee consistency and quality, providing our partners with the confidence needed to integrate this intermediate into their critical manufacturing processes.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through innovative chemical solutions. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex intermediate synthesis can drive value for your organization. Together, we can turn chemical challenges into commercial opportunities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
