Advanced Catalytic Strategy for High-Purity 2-Phenylacrylonitrile Production and Commercial Scale-Up
The global demand for high-performance antimicrobial agents and specialized polymer additives has intensified the search for efficient synthetic routes to key building blocks like 2-phenylacrylonitrile. A pivotal advancement in this domain is detailed in patent CN109134312B, which discloses a robust preparation method utilizing phenylacetonitrile and paraformaldehyde as primary feedstocks. This technology represents a significant leap forward in organic synthesis, specifically addressing the longstanding challenges of chemical selectivity and yield optimization in methylene homologation reactions. By employing a unique catalytic system based on bicarbonate salts and phase transfer catalysts, the process achieves yields up to 79%, surpassing traditional methodologies that often struggle with byproduct formation. For R&D directors and procurement strategists, this patent offers a compelling blueprint for cost reduction in pharmaceutical intermediates manufacturing, ensuring a stable supply of high-purity materials essential for downstream applications in agrochemicals and industrial biocides.
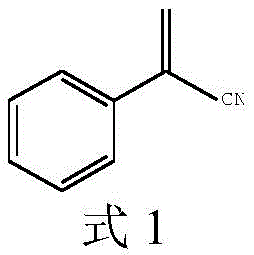
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
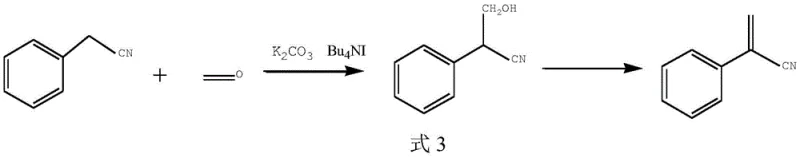
Historically, the synthesis of 2-phenylacrylonitrile has been plagued by severe selectivity issues arising from the harsh reaction conditions required to drive the condensation of phenylacetonitrile with formaldehyde sources. Early literature, such as the method described in the Journal of Organic Chemistry (1956), relied heavily on sodium methoxide in methanol at temperatures between 50°C and 60°C. While this approach established the foundational chemistry, it suffered from a critical flaw: the excessive alkalinity of sodium methoxide promoted the polymerization of the product and facilitated unwanted side reactions. Specifically, the hydroxymethyl intermediate is highly sensitive to strong basic conditions, leading to the formation of complex oligomers and making product isolation arduous. Furthermore, more recent attempts using potassium carbonate and tetrabutylammonium iodide in toluene at 80°C still required rigorous nitrogen protection to prevent yield degradation, indicating a lack of process robustness suitable for large-scale industrial application.
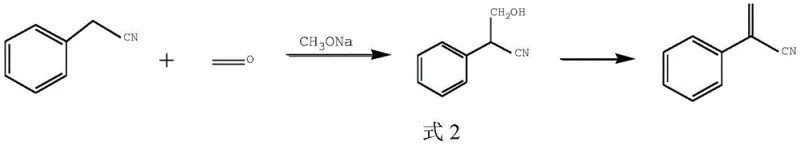
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in CN109134312B introduces a paradigm shift by utilizing mild acid binding agents such as potassium bicarbonate or sodium bicarbonate. This strategic substitution fundamentally alters the reaction landscape, creating a buffered environment that is sufficiently basic to drive the methylene addition but mild enough to preserve the integrity of the sensitive hydroxymethyl intermediate. The process operates efficiently at moderate temperatures ranging from 50°C to 80°C without the need for inert gas shielding, drastically simplifying the engineering requirements for commercial reactors. By eliminating the necessity for expensive strong bases and complex atmospheric controls, this method not only enhances the chemical selectivity but also streamlines the workflow, making it an ideal candidate for commercial scale-up of complex nitriles where operational simplicity translates directly to margin improvement.
Mechanistic Insights into Bicarbonate-Catalyzed Methylene Homologation
The core innovation of this technology lies in the precise modulation of basicity to control the reaction pathway. In traditional strong-base catalysis, the deprotonation of phenylacetonitrile generates a highly reactive carbanion that attacks paraformaldehyde to form 3-hydroxy-2-phenylpropionitrile. However, under strongly alkaline conditions, this intermediate retains significant acidity at the alpha-position, allowing it to undergo a second deprotonation and react with another molecule of phenylacetonitrile or formaldehyde. This cascade leads to the formation of 2,4-bishydroxymethyl-2,4-diphenyl-glutaronitrile, a heavy byproduct that consumes valuable starting materials and complicates purification. The use of bicarbonate salts creates a thermodynamic ceiling on the pH of the reaction medium, effectively suppressing this secondary deprotonation event while still permitting the initial condensation to proceed.

Furthermore, the inclusion of a phase transfer catalyst, specifically tetrabutylammonium bromide, plays a pivotal role in enhancing the mass transfer between the aqueous bicarbonate phase and the organic toluene phase. This ensures that the active catalytic species are available at the interface where the reaction occurs, maximizing the collision frequency between the nucleophilic phenylacetonitrile anion and the electrophilic formaldehyde. The result is a highly selective transformation that minimizes the generation of polymeric tars and dimers. For quality assurance teams, this mechanistic control translates to a cleaner crude profile, reducing the load on downstream distillation columns and ensuring that the final high-purity 2-phenylacrylonitrile meets stringent specifications for trace impurities, which is critical for its application in sensitive antimicrobial formulations.
How to Synthesize 2-Phenylacrylonitrile Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation, requiring standard chemical processing equipment without the need for specialized high-pressure or cryogenic setups. The protocol involves a straightforward mixing of phenylacetonitrile, paraformaldehyde, the bicarbonate catalyst, and the phase transfer agent in a solvent like toluene or xylene. Following a controlled heating period, the reaction mixture undergoes a simple workup involving phase separation and acid neutralization.
- Mix phenylacetonitrile, toluene solvent, saturated potassium bicarbonate solution, and tetrabutylammonium bromide in a reactor.
- Heat the mixture to 60°C and add paraformaldehyde, maintaining the temperature for 2.5 hours to complete the methylene homologation.
- Cool the reaction, separate the organic layer, neutralize with dilute hydrochloric acid, and purify via vacuum distillation to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this bicarbonate-catalyzed process offers profound advantages in terms of raw material security and operational expenditure. Traditional methods relying on alkali metal alkoxides or strong carbonates often necessitate strict moisture control and inert atmospheres, which increase capital expenditure on reactor infrastructure and raise the barrier to entry for contract manufacturing organizations. By contrast, the tolerance of the new method to ambient conditions allows for greater flexibility in production scheduling and reduces the risk of batch failures due to atmospheric leaks. This robustness ensures a more reliable supply chain, positioning manufacturers as a reliable agrochemical intermediate supplier capable of meeting volatile market demands without compromising on delivery timelines.
- Cost Reduction in Manufacturing: The substitution of expensive and hazardous strong bases with commodity-grade bicarbonate salts results in a direct reduction in raw material costs. Additionally, the elimination of nitrogen purging systems reduces utility consumption and simplifies safety protocols, leading to substantial operational savings. The higher selectivity of the reaction means less waste generation and lower solvent usage per kilogram of product, further driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: The use of widely available and stable reagents like potassium bicarbonate and paraformaldehyde mitigates the risk of supply disruptions associated with specialty catalysts. The simplified process flow, which avoids complex filtration steps required to remove polymeric byproducts, shortens the overall cycle time. This efficiency allows for faster turnover of production assets, enabling suppliers to respond more agilely to procurement requests and maintain consistent inventory levels.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup align well with green chemistry principles, reducing the environmental footprint of the manufacturing process. The absence of heavy metal catalysts or toxic reagents simplifies wastewater treatment and regulatory compliance, facilitating smoother permitting for capacity expansion. This sustainability profile is increasingly valued by multinational corporations seeking to optimize their Scope 3 emissions and partner with environmentally responsible vendors.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for process engineers aiming to replicate the high yields reported in the laboratory setting at an industrial scale.
Q: Why is potassium bicarbonate preferred over sodium methoxide for this synthesis?
A: Potassium bicarbonate acts as a mild acid binding agent that prevents the polymerization of the product and suppresses side reactions like dimerization, which are common with strong bases like sodium methoxide.
Q: Does this process require inert gas protection?
A: No, unlike previous methods requiring nitrogen protection to maintain yield, this novel bicarbonate-catalyzed process operates effectively under ambient atmosphere, simplifying operational complexity.
Q: What is the primary impurity formed if strong bases are used?
A: When strong bases are employed, the intermediate 3-hydroxy-2-phenylpropionitrile reacts further to form 2,4-bishydroxymethyl-2,4-diphenyl-glutaronitrile, significantly reducing the selectivity for the target acrylonitrile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenylacrylonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that 2-phenylacrylonitrile plays in the value chains of the agrochemical and pharmaceutical industries. Our technical team has extensively analyzed the pathway described in CN109134312B and possesses the expertise to translate this laboratory-scale innovation into a commercially viable manufacturing process. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that adheres to stringent purity specifications. Our rigorous QC labs are equipped to monitor trace impurities and ensure that every batch meets the exacting standards required for downstream antimicrobial and polymer applications.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis capabilities, we can help you reduce lead time for high-purity 2-phenylacrylonitrile and secure a competitive advantage in your market. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
