Advanced Heterocyclic Quinone Thiophene Materials for Next-Gen Organic Electronics
The rapid evolution of the organic electronics sector demands materials that balance high performance with manufacturability. Patent CN102574867B introduces a breakthrough class of heterocyclic quinone-type thiophene organic photoelectric materials, specifically designed to overcome the limitations of traditional inorganic semiconductors. These compounds, characterized by the general structure shown in Formula (1), feature a unique architecture comprising multiple quinoid thiophene rings and strong electron-withdrawing dicyanovinyl end groups. This structural innovation results in a moderate energy bandgap and an exceptionally wide spectral response, covering the visible light range from approximately 300nm to 750nm. For R&D directors seeking reliable organic photoelectric material suppliers, this technology represents a significant leap forward in developing efficient solar cells and organic field-effect transistors.
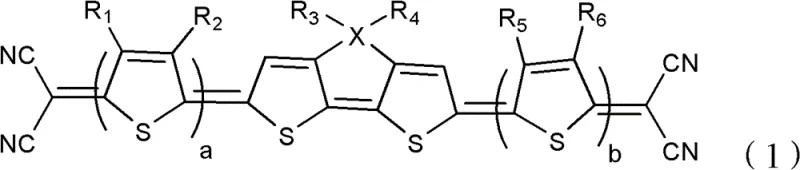
Beyond their optical properties, these materials exhibit superior thermal and environmental stability, which is critical for the longevity of commercial devices. The incorporation of alkyl or alkoxy chains at specific positions (R3 and R4) enhances solubility, facilitating solution processing techniques such as spin coating or inkjet printing. This solubility is a key factor in reducing manufacturing costs compared to vacuum deposition methods required for many inorganic counterparts. As the industry shifts towards flexible and lightweight electronics, the ability to process these materials from solution offers a distinct competitive advantage. The patent details a robust synthetic pathway that ensures high purity and structural integrity, making it an ideal candidate for scale-up in industrial settings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for conjugated organic semiconductors often suffer from complex multi-step procedures that result in low overall yields and difficult purification processes. Many conventional methods rely on harsh reaction conditions that can degrade sensitive functional groups or lead to irregular polymerization, creating a broad distribution of molecular weights that negatively impacts device performance. Furthermore, achieving the precise placement of electron-withdrawing groups necessary for optimal bandgap tuning is frequently challenging, leading to materials with inconsistent optoelectronic properties. The reliance on expensive catalysts and difficult-to-remove metal residues also poses significant challenges for commercial viability, particularly in applications requiring high purity standards. These factors collectively increase the cost of goods sold and extend the time-to-market for new electronic devices.
The Novel Approach
The methodology outlined in CN102574867B addresses these challenges through a streamlined three-step synthesis strategy centered around the versatile Stille coupling reaction. This approach allows for the modular assembly of the thiophene backbone, enabling precise control over the molecular architecture and the length of the conjugated system. By utilizing readily available precursors such as brominated thiophenes and stannylated dithienothiophene derivatives, the process minimizes the need for exotic reagents. The subsequent bromination and condensation steps are highly selective, ensuring that the critical dicyanovinyl end groups are installed efficiently without compromising the integrity of the core structure. This novel route not only simplifies the workflow but also significantly enhances the reproducibility of the final product, a crucial metric for any reliable organic photoelectric material supplier.
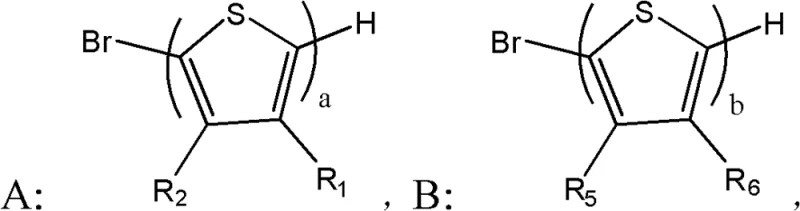
Mechanistic Insights into Stille-Catalyzed Assembly and Functionalization
The core of this synthesis lies in the palladium-catalyzed Stille coupling reaction, which serves as the foundation for constructing the extended pi-conjugated system. In this step, organopalladium catalysts such as Pd2(dba)3配合 with phosphine ligands facilitate the cross-coupling between the stannylated central unit and the brominated thiophene side chains. The mechanism involves oxidative addition of the aryl halide to the palladium center, followed by transmetallation with the organotin species and reductive elimination to form the new carbon-carbon bond. This cycle is highly efficient and tolerant of various functional groups, allowing for the introduction of solubilizing alkyl chains that are essential for processability. The use of tetrahydrofuran (THF) as a solvent under reflux conditions ensures complete conversion while maintaining the stability of the reactive intermediates.
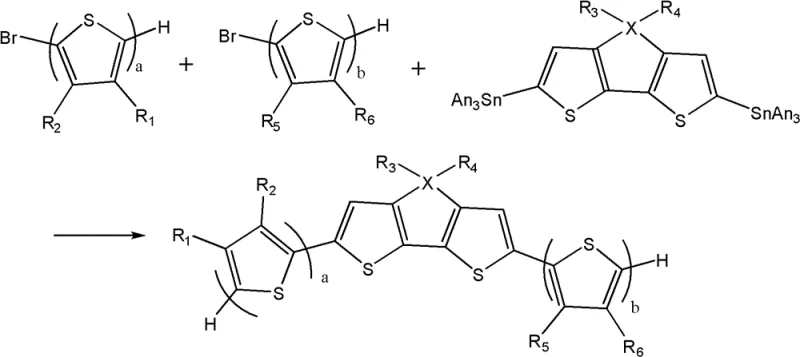
Following the backbone assembly, the material undergoes a strategic bromination using N-bromosuccinimide (NBS) to activate the terminal positions for final functionalization. This electrophilic substitution is carefully controlled to occur selectively at the alpha-positions of the terminal thiophene rings, avoiding over-bromination which could lead to cross-linking or defects. The final step involves a condensation reaction with malononitrile in the presence of a base such as sodium hydride. This Knoevenagel-type condensation installs the powerful dicyanovinyl electron-withdrawing groups, which are pivotal for lowering the LUMO energy level and enhancing electron affinity. The resulting push-pull electronic structure maximizes intramolecular charge transfer, directly translating to improved photocurrent generation in solar cell applications. This precise mechanistic control ensures that the final material meets the stringent purity specifications required for high-performance electronic devices.
How to Synthesize Heterocyclic Quinone Thiophene Efficiently
The synthesis of these advanced materials requires careful attention to reaction conditions and purification protocols to ensure optimal performance. The process begins with the preparation of high-purity precursors, followed by the critical coupling and functionalization steps described above. Each stage must be monitored closely to prevent the formation of byproducts that could act as charge traps in the final device. Detailed standard operating procedures regarding stoichiometry, temperature control, and workup methods are essential for consistent results. For a comprehensive breakdown of the specific experimental parameters and purification techniques, please refer to the standardized synthesis guide below.
- Prepare precursors including brominated thiophene derivatives and stannylated dithienothiophene units.
- Execute Stille coupling reaction using palladium catalysts to form the central conjugated backbone.
- Perform bromination followed by condensation with malononitrile to finalize the electron-withdrawing end groups.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route described in this patent offers substantial opportunities for cost optimization and supply chain stabilization. The reliance on commodity chemicals such as malononitrile, NBS, and common thiophene derivatives reduces dependency on specialized, single-source vendors. This diversification of raw material sources mitigates the risk of supply disruptions and price volatility, ensuring a more predictable cost structure for long-term production planning. Additionally, the high selectivity of the reactions minimizes waste generation and simplifies downstream purification, which directly contributes to lower operational expenditures. For procurement managers focused on cost reduction in electronic chemical manufacturing, this efficiency translates into a more competitive pricing model for the final active materials.
- Cost Reduction in Manufacturing: The streamlined three-step synthesis eliminates the need for complex protection-deprotection sequences often found in alternative routes. By utilizing robust catalytic systems that operate under relatively mild conditions, the process reduces energy consumption and equipment wear. The high yields associated with Stille coupling mean that less starting material is required to produce a given amount of final product, effectively lowering the material cost per kilogram. Furthermore, the ability to purify intermediates using standard chromatographic techniques avoids the need for expensive preparative HPLC or recrystallization from exotic solvents, driving down overall processing costs significantly.
- Enhanced Supply Chain Reliability: The precursors used in this synthesis, including various brominated thiophenes and organotin compounds, are widely available from multiple global chemical suppliers. This availability ensures that production schedules are not held hostage by the lead times of niche reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain fluctuations. For supply chain heads concerned with reducing lead time for high-purity organic semiconductors, this resilience is a critical factor in maintaining continuous manufacturing operations.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram and ton-scale commercial production. The use of standard solvents like THF and DMF allows for integration into existing chemical infrastructure without major capital investment. Moreover, the efficient atom economy of the coupling reactions reduces the volume of hazardous waste generated per unit of product. This alignment with green chemistry principles facilitates easier regulatory compliance and reduces the environmental footprint of the manufacturing process, a growing priority for multinational corporations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this material class is essential for making informed sourcing decisions. The following questions address common inquiries regarding the performance, stability, and application of these heterocyclic quinone-type thiophene compounds. The answers are derived directly from the technical data and experimental results presented in the patent documentation, ensuring accuracy and relevance for potential partners.
Q: What are the primary applications of heterocyclic quinone-type thiophene materials?
A: These materials are primarily designed for organic solar cells, organic field-effect transistors (OFETs), and organic light-emitting diodes (OLEDs) due to their wide spectral response and stability.
Q: How does the Stille coupling reaction benefit the synthesis of these materials?
A: Stille coupling allows for the modular assembly of complex thiophene backbones under mild conditions, ensuring high yields and facilitating the introduction of solubilizing alkyl chains.
Q: Why is the dicyanovinyl group important in this molecular structure?
A: The dicyanovinyl group acts as a strong electron-withdrawing unit that extends the conjugation system, lowers the energy gap, and shifts absorption into the red and near-infrared regions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Quinone Thiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in CN102574867B for the next generation of organic electronic devices. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of material meets the exacting standards required for high-efficiency solar cells and transistors. Our commitment to quality assurance means that you can rely on us for consistent supply without compromising on performance metrics.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development cycles. By leveraging our expertise in organic synthesis and process engineering, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Let us help you engineer the future of organic electronics with materials that deliver both performance and value.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
