Advanced Green Synthesis of 3-Oxo-N-(4-Trifluoromethylphenyl)Butanamide for High-Volume API Production
Introduction to Next-Generation Teriflunomide Intermediate Manufacturing
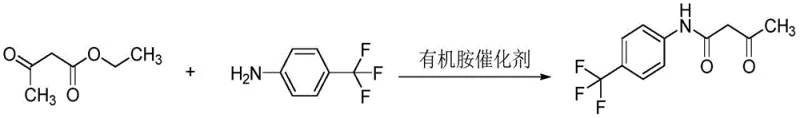
The pharmaceutical landscape for Multiple Sclerosis treatments continues to evolve, driving an intense demand for high-quality intermediates such as 3-oxo-N-(4-trifluoromethylphenyl)butanamide. Patent CN112457210A introduces a transformative preparation method that addresses critical bottlenecks in the synthesis of this key precursor for Teriflunomide. Unlike legacy processes that rely on hazardous solvents and extended reaction times, this innovation utilizes an excess of p-trifluoromethylaniline acting simultaneously as a reactant and a solvent. This dual-function approach, coupled with efficient organic base catalysis, creates a closed-loop system that maximizes atom economy while minimizing environmental impact. For R&D directors and supply chain leaders, this represents a pivotal shift towards sustainable, cost-effective manufacturing that does not compromise on the stringent purity standards required for active pharmaceutical ingredient (API) production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-oxo-N-(4-trifluoromethylphenyl)butanamide has been plagued by inefficient protocols that hinder large-scale commercialization. Prior art, such as the methods disclosed in WO 2010013159 A1, typically employs xylene as a solvent, necessitating reflux conditions for up to 48 hours to drive the reaction to completion. Such prolonged exposure to high thermal energy inevitably leads to the decomposition of sensitive starting materials and the target product itself, resulting in a complex impurity profile that is difficult to purge. Furthermore, the reliance on volatile organic compounds like xylene, toluene, or nitromethane introduces significant safety hazards and environmental liabilities, requiring expensive solvent recovery systems and rigorous waste gas treatment protocols. The necessity for post-reaction purification via column chromatography further exacerbates operational costs and limits throughput, making these traditional routes economically unviable for modern, high-volume pharmaceutical supply chains.
The Novel Approach
The methodology outlined in CN112457210A fundamentally reengineers the reaction environment to overcome these thermodynamic and kinetic barriers. By employing a significant molar excess of p-trifluoromethylaniline (ranging from 1:1.2 to 1:10 relative to ethyl acetoacetate), the process leverages Le Chatelier's principle to drive the equilibrium forward without the need for external, toxic solvents. This excess amine serves as the reaction medium, effectively solubilizing the reactants while facilitating the continuous removal of the ethanol byproduct through distillation. The strategic use of organic base catalysts, such as sodium methoxide or tri-n-butylamine, accelerates the amidation rate at moderate temperatures between 80°C and 140°C. This optimization not only drastically shortens the reaction cycle to between 10 and 30 hours but also preserves the structural integrity of the molecule, yielding a crude product with exceptional purity that often bypasses the need for chromatographic purification.
Mechanistic Insights into Organic Base Catalyzed Amidation
At the core of this technological advancement lies a sophisticated understanding of nucleophilic acyl substitution mechanisms facilitated by organic base catalysis. The reaction initiates with the deprotonation of the amine or the activation of the carbonyl group of ethyl acetoacetate by the organic base catalyst, such as N-methylpyrrolidone or amino acid derivatives. This activation lowers the energy barrier for the nucleophilic attack of the p-trifluoromethylaniline nitrogen on the ester carbonyl carbon. The formation of the tetrahedral intermediate is stabilized by the reaction medium, which consists primarily of the aromatic amine itself. Crucially, the continuous distillation of ethanol shifts the reaction equilibrium irreversibly towards the product side. This dynamic removal of the leaving group prevents the reverse hydrolysis reaction, ensuring high conversion rates even without the extreme thermal stress associated with traditional solvent reflux methods. The result is a clean reaction profile with minimal formation of side products such as diketenes or polymerized species.
Impurity control is inherently built into this mechanistic design, addressing a primary concern for R&D directors focused on downstream processing. In conventional xylene-based syntheses, thermal degradation often generates structurally similar byproducts that co-elute during purification, requiring resource-intensive recrystallization or chromatography steps. In contrast, the solvent-free nature of this new method reduces the thermal load on the reaction mixture, thereby suppressing decomposition pathways. The excess p-trifluoromethylaniline can be easily recovered via vacuum distillation after the reaction is complete, leaving behind the target amide with high assay values, often exceeding 98% purity as demonstrated in experimental examples. This inherent purity simplifies the isolation process, allowing for direct crystallization or simple concentration, which significantly reduces the overall processing time and solvent consumption per kilogram of product manufactured.
How to Synthesize 3-Oxo-N-(4-Trifluoromethylphenyl)Butanamide Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal gradients to maximize yield and safety. The process begins by charging ethyl acetoacetate into a dry reactor, followed by the sequential addition of the excess amine and the selected organic base catalyst under an inert atmosphere. The reaction mixture is then subjected to a controlled heating ramp, initially holding at 80°C to initiate the reaction before gradually increasing to the optimal range of 100°C to 140°C. Throughout this phase, the evolved ethanol is continuously distilled off, serving as a real-time indicator of reaction progress. Detailed standardized operating procedures regarding specific catalyst loading ratios, exact temperature ramping rates, and work-up protocols are essential for reproducible commercial success.
- Charge ethyl acetoacetate and excess p-trifluoromethylaniline into a reactor equipped with mechanical stirring and distillation capabilities.
- Add a catalytic amount of organic base (e.g., sodium methoxide or tri-n-butylamine) under inert gas protection.
- Heat gradually to 80-140°C to facilitate amidation while continuously distilling off the generated ethanol byproduct to drive equilibrium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers profound strategic advantages beyond mere technical feasibility. The elimination of hazardous solvents like xylene and nitromethane directly correlates to a substantial reduction in raw material costs and waste disposal fees. By removing the dependency on column chromatography, the manufacturing footprint is significantly reduced, allowing for higher batch frequencies within existing infrastructure. This process intensification translates to a more resilient supply chain capable of responding rapidly to market fluctuations in Teriflunomide demand. Furthermore, the ability to recover and reuse the excess amine reactant creates a circular material flow that insulates production costs from volatile raw material pricing, ensuring long-term economic stability for the manufacturing operation.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the drastic simplification of the downstream processing workflow. Traditional methods incur heavy costs associated with solvent purchase, recovery, and the extensive labor and materials required for chromatographic purification. By utilizing the reactant as the solvent and achieving high crude purity, this method eliminates the need for expensive stationary phases and large volumes of elution solvents. Additionally, the recovery of unreacted p-trifluoromethylaniline allows for near-quantitative utilization of raw materials, effectively lowering the cost of goods sold (COGS) per unit. The reduction in energy consumption, due to shorter reaction times and lower overall thermal requirements compared to 48-hour refluxes, further contributes to significant operational expenditure savings.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the regulatory scrutiny surrounding the use of Class 2 and Class 3 solvents. By adopting a solvent-free or self-solvating approach, manufacturers mitigate the risk of supply disruptions caused by environmental compliance issues or solvent shortages. The simplified process flow reduces the number of unit operations, thereby decreasing the probability of equipment failure or batch deviations. This robustness ensures a consistent and reliable output of high-purity intermediates, enabling pharmaceutical partners to maintain steady inventory levels without the need for excessive safety stock. The ease of scaling this technology from pilot to commercial scale further guarantees that supply can be ramped up quickly to meet surging global demand for Multiple Sclerosis therapies.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology aligns perfectly with modern green chemistry mandates. The avoidance of volatile, toxic organic solvents minimizes the emission of VOCs, reducing the burden on废气 treatment facilities and lowering the carbon footprint of the manufacturing site. The process generates significantly less hazardous waste, simplifying disposal logistics and reducing associated liability costs. Scalability is enhanced because the reaction does not rely on specialized equipment for handling large volumes of flammable solvents; standard stainless steel reactors equipped with distillation columns are sufficient. This compatibility with existing general-purpose chemical infrastructure allows for rapid technology transfer and immediate capacity expansion without the need for capital-intensive retrofitting or new construction projects.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on how this method compares to established industry standards. Understanding these nuances is critical for stakeholders evaluating the feasibility of integrating this technology into their current manufacturing portfolios to achieve competitive advantages in the pharmaceutical intermediate market.
Q: How does this new method improve purity compared to traditional xylene reflux?
A: Traditional methods using xylene require prolonged reflux (up to 48 hours), leading to thermal decomposition of raw materials and products. The new method uses excess amine as a solvent at controlled temperatures (80-140°C) with continuous ethanol removal, significantly reducing degradation impurities and achieving purities over 98%.
Q: What are the environmental benefits of eliminating volatile organic solvents?
A: By replacing toxic solvents like xylene, toluene, or nitromethane with excess reactant (p-trifluoromethylaniline), the process eliminates VOC emissions and simplifies waste treatment. The excess amine is easily recovered and reused, aligning with green chemistry principles and reducing hazardous waste disposal costs.
Q: Is this process scalable for industrial manufacturing of Teriflunomide intermediates?
A: Yes, the process is designed for industrial scalability. It avoids complex column chromatography purification, relying instead on simple concentration and recovery steps. The use of robust organic base catalysts and standard heating/distillation equipment makes it highly suitable for multi-ton production campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Oxo-N-(4-Trifluoromethylphenyl)Butanamide Supplier
As the global demand for Teriflunomide continues to rise, securing a supply partner with deep technical expertise in advanced intermediate synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging cutting-edge methodologies like the one described in CN112457210A to deliver superior value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every gram of 3-oxo-N-(4-trifluoromethylphenyl)butanamide meets stringent purity specifications. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee batch-to-batch consistency, providing our clients with the confidence needed to navigate complex regulatory filings and commercial launches successfully.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and comprehensive route feasibility assessments, allowing you to validate the quality and economic viability of our offerings against your internal benchmarks. Let us help you engineer a more efficient, sustainable, and profitable future for your pharmaceutical projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
