Advanced Synthesis of Tegaserod Maleate: Technical Upgrade and Commercial Scalability
Advanced Synthesis of Tegaserod Maleate: Technical Upgrade and Commercial Scalability
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for the production of critical active pharmaceutical ingredients (APIs) and their intermediates. Patent CN103467358A introduces a significant breakthrough in the preparation method for Tegaserod Maleate, a potent 5-HT4 receptor partial agonist used primarily for treating irritable bowel syndrome with constipation (IBS-C). This innovative technical disclosure outlines a robust synthetic route that fundamentally shifts away from hazardous solvent systems, specifically replacing methanol with safer alternatives like ethanol and ethyl acetate. For R&D directors and process chemists, this represents a pivotal opportunity to enhance the purity profile of the final product while simultaneously addressing stringent environmental, health, and safety (EHS) regulations. The methodology described not only streamlines the operational workflow by reducing the number of purification steps but also ensures a more consistent quality of the intermediate N-amyl-N-aminoguanidine, which is crucial for the downstream condensation reactions.
From a strategic supply chain perspective, the adoption of this improved synthesis protocol offers substantial advantages in terms of cost reduction in API manufacturing and operational reliability. By eliminating the reliance on high-toxicity reagents and optimizing the crystallization processes through mixed solvent systems, manufacturers can achieve a more stable production cycle with reduced downtime for waste treatment. The patent details a comprehensive approach that covers everything from the initial alkylation of thiosemicarbazide to the final salt formation with maleic acid, providing a complete blueprint for industrial implementation. As a reliable pharmaceutical intermediate supplier, understanding these nuanced process improvements is essential for maintaining competitiveness in the global market, where regulatory compliance and cost-efficiency are paramount drivers of procurement decisions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Tegaserod and its precursors have historically been plagued by significant operational challenges that hinder large-scale efficiency and safety. A primary concern in legacy methods is the extensive use of methanol as a reaction and processing solvent, which poses severe toxicity risks to personnel and complicates waste management protocols due to its volatility and flammability. Furthermore, conventional techniques often generate substantial amounts of mercaptan byproducts, which are not only malodorous but also highly toxic, requiring complex and expensive tail gas absorption systems to prevent environmental contamination. The purification stages in older methodologies frequently involve multiple recrystallization steps using单一 solvents that fail to effectively remove trace impurities, leading to lower overall yields and inconsistent product quality that may not meet the rigorous specifications required for pharmaceutical applications. Additionally, the harsh reaction conditions often necessitated by these outdated routes can lead to the degradation of sensitive intermediates, further exacerbating yield losses and increasing the cost of goods sold (COGS) for the final API.
The Novel Approach
The novel approach detailed in the patent data presents a transformative solution by engineering a synthesis pathway that prioritizes both chemical efficiency and environmental stewardship. By substituting methanol with ethanol and utilizing a mixed solvent system of ethanol and ethyl acetate for the final salt formation, the new method drastically reduces the toxicity profile of the entire manufacturing process, thereby lowering the barrier for regulatory approval and facility compliance. This strategic solvent switch not only enhances the solubility of the target compound during crystallization but also facilitates easier solvent recovery and recycling, contributing to significant cost reduction in pharmaceutical intermediate manufacturing. The process incorporates a specialized tail gas treatment step using saturated sodium hydroxide solution to effectively neutralize mercaptan gases generated during the aminolysis step, ensuring a safer working environment and minimizing atmospheric emissions. Moreover, the optimization of reaction parameters, such as precise temperature control and catalyst selection, allows for higher conversion rates and cleaner reaction profiles, ultimately delivering a high-purity tegaserod maleate product that exceeds industry standards for impurity levels.
Mechanistic Insights into Base-Catalyzed Alkylation and Condensation
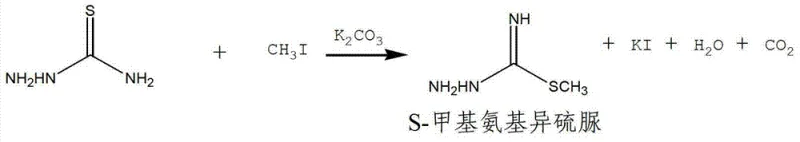
The core of this synthetic innovation lies in the meticulous control of nucleophilic substitution and condensation mechanisms, which are fundamental to constructing the guanidine scaffold of Tegaserod. The initial step involves the alkylation of thiosemicarbazide with methyl iodide in the presence of a base catalyst such as potassium carbonate or sodium carbonate. In this reaction, the base deprotonates the sulfur atom of the thiosemicarbazide, generating a highly nucleophilic thiolate anion that attacks the methyl group of the alkylating agent via an SN2 mechanism. This step is critical because the choice of base and the maintenance of low temperatures (0-15°C) prevent over-alkylation and side reactions, ensuring the selective formation of S-methylamino isothiourea with high fidelity. The use of anhydrous ethanol as the solvent in this stage is particularly advantageous as it stabilizes the ionic intermediates while remaining inert to the reactive species, thus preserving the integrity of the thioamide functionality for subsequent transformations.
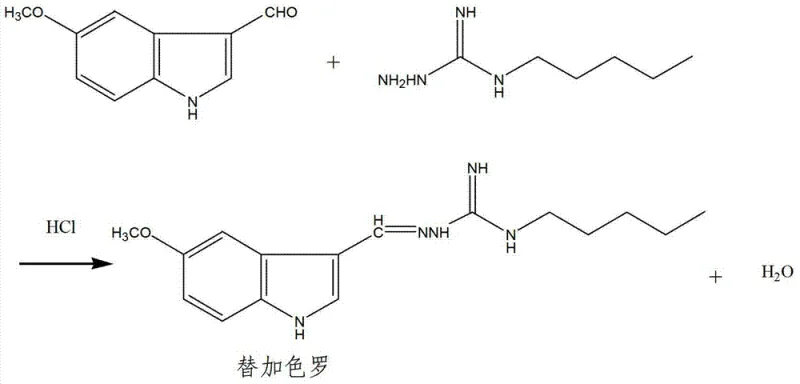
Following the formation of the S-methyl intermediate, the process proceeds to a nucleophilic substitution with n-amylamine to construct the N-amyl-N-aminoguanidine core. This transformation is driven by the strong nucleophilicity of the primary amine, which displaces the methylthio group, releasing methanethiol as a byproduct. The mechanistic elegance of this step is enhanced by the in-situ absorption of the evolved gas, which shifts the equilibrium towards product formation according to Le Chatelier's principle, thereby driving the reaction to completion without the need for excessive reagent loading. The subsequent condensation with 5-methoxyindole-3-aldehyde under mildly acidic conditions (pH 5-6) involves the formation of a hydrazone linkage, a reaction that is highly sensitive to pH and temperature. Maintaining the pH within the narrow window of 5-6 ensures that the amine nucleophile remains sufficiently unprotonated to attack the aldehyde carbonyl while preventing the acid-catalyzed hydrolysis of the sensitive guanidine moiety, resulting in the efficient assembly of the Tegaserod backbone with minimal impurity generation.
How to Synthesize Tegaserod Maleate Efficiently
Implementing this synthesis route requires strict adherence to the optimized reaction parameters to maximize yield and purity while ensuring operational safety. The process begins with the careful preparation of the S-methylamino isothiourea intermediate, followed by its conversion to the guanidine derivative, and concludes with the condensation and salt formation steps. Each stage demands precise control over stoichiometry, temperature, and solvent composition to avoid the pitfalls associated with traditional methods. For process engineers and laboratory teams, the following guide outlines the critical operational milestones derived from the patent data, serving as a foundational reference for scaling this technology from benchtop to pilot plant. Detailed standardized synthesis steps are provided in the section below to facilitate immediate technical evaluation and trial runs.
- Alkylation of thiosemicarbazide with methyl iodide in ethanol using potassium carbonate catalyst at 0-15°C.
- Substitution reaction with n-amylamine in ethanol at 55-60°C to form N-amyl-N-aminoguanidine, absorbing mercaptan byproduct.
- Condensation with 5-methoxyindole-3-aldehyde under acidic conditions (pH 5-6) at 25-50°C.
- Salt formation with maleic acid in ethanol/ethyl acetate mixture followed by crystallization and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this patented synthesis method offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of high-toxicity solvents like methanol in favor of ethanol and ethyl acetate not only reduces the cost associated with hazardous waste disposal but also simplifies the regulatory burden related to solvent residue limits in the final drug product. This shift translates into a more streamlined supply chain where raw material sourcing is less constrained by volatile chemical markets, as ethanol is a widely available and cost-effective commodity chemical. Furthermore, the improved yield and purity profiles mean that less raw material is wasted per kilogram of finished product, effectively lowering the unit cost of production and enhancing the overall margin potential for the manufacturing entity. The robustness of the process also implies fewer batch failures and less rework, leading to more predictable delivery schedules and stronger relationships with downstream API manufacturers.
- Cost Reduction in Manufacturing: The substitution of expensive or heavily regulated solvents with common industrial grades like ethanol significantly lowers the direct material costs associated with the synthesis. By optimizing the crystallization process through the use of mixed solvents, the method reduces the volume of solvent required for purification, which in turn decreases energy consumption for solvent recovery and distillation. The higher reaction yields observed in this pathway mean that the throughput of the reactor train is effectively increased without the need for capital expansion, allowing for greater production capacity within existing infrastructure. Additionally, the reduced generation of hazardous byproducts minimizes the expenditure on waste treatment facilities and compliance monitoring, contributing to substantial long-term operational savings.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials such as thiosemicarbazide and n-amylamine ensures a stable supply base that is less susceptible to geopolitical or logistical disruptions. The simplified workup procedures, which avoid complex extraction and chromatography steps, reduce the turnaround time between batches, enabling a more responsive production schedule that can adapt to fluctuating market demands. The inherent safety of the process, characterized by milder reaction conditions and effective gas scrubbing, reduces the risk of unplanned shutdowns due to safety incidents, thereby guaranteeing a continuous flow of high-quality intermediates to customers. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery windows imposed by major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor equipment and conditions that can be easily translated from laboratory scale to multi-ton commercial production. The effective management of mercaptan emissions through alkaline absorption demonstrates a proactive approach to environmental compliance, mitigating the risk of regulatory fines and community opposition. By adopting greener chemistry principles, such as atom economy and safer solvents, the manufacturing process aligns with the sustainability goals of modern pharmaceutical companies, enhancing the brand value and marketability of the final product. This alignment with ESG (Environmental, Social, and Governance) criteria is increasingly becoming a key differentiator in supplier selection processes for global healthcare corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Tegaserod Maleate using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of the process for potential partners and stakeholders. Understanding these details is vital for making informed decisions about technology transfer and long-term supply agreements. The answers provided reflect the current state of the art in guanidine derivative synthesis and highlight the specific improvements offered by this invention.
Q: How does the new synthesis route improve environmental safety compared to traditional methods?
A: The novel process replaces toxic methanol with ethanol and ethyl acetate, significantly reducing operator exposure risks and environmental impact while simplifying solvent recovery systems.
Q: What are the key yield improvements in this patented method?
A: The optimized conditions, particularly the use of specific base catalysts and controlled temperature profiles, result in substantially higher yields across all steps, minimizing raw material waste.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method utilizes common industrial solvents and standard reaction conditions without requiring extreme pressures or temperatures, making it highly scalable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tegaserod Maleate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity tegaserod maleate that adheres to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex organic syntheses with the utmost attention to safety and quality, making us an ideal partner for companies seeking to secure a stable and compliant supply of this vital intermediate.
We invite you to collaborate with us to leverage the full potential of this patented synthesis route for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can drive down your total cost of ownership. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to evaluate the superior quality and reliability of our Tegaserod Maleate offerings. Let us be your trusted partner in navigating the complexities of pharmaceutical manufacturing and achieving your commercial objectives together.
