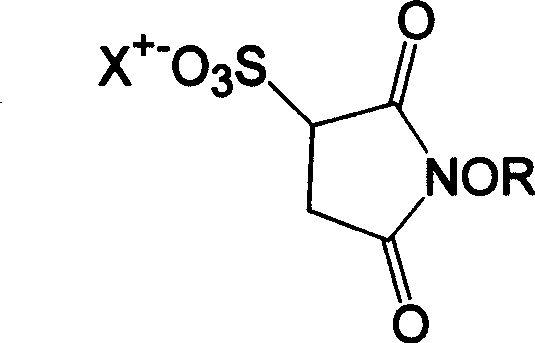
Scalable Synthesis of High-Purity N-Hydroxy Thiosuccinimide Salt for Advanced Bioconjugation
The landscape of bioconjugation chemistry relies heavily on the availability of high-purity cross-linking reagents, specifically N-hydroxy thiosuccinimide salts, often referred to industrially as Sulfo-NHS. Patent CN100480239C introduces a transformative synthetic methodology that addresses long-standing stability and purity issues inherent in traditional manufacturing routes. By utilizing N-acyloxymaleimide as a robust starting material rather than the unstable N-hydroxymaleimide, this innovation enables the direct reaction with thiosulfate salts to produce the target compound with exceptional efficiency. This technical breakthrough is pivotal for pharmaceutical manufacturers seeking reliable sources of activated esters that maintain integrity during storage and transport. The process eliminates the formation of complex isomeric mixtures that plague older cyclization techniques, thereby streamlining the purification workflow and ensuring a consistent supply of critical reagents for protein modification and labeling applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-hydroxy thiosuccinimide salts has been fraught with significant chemical challenges that impact both yield and product quality. The first conventional technique involves the reaction of N-hydroxymaleimide with sulfothiorine; however, N-hydroxymaleimide is notoriously unstable, exhibiting tangible decomposition within just a few days even when stored at room temperature. This instability leads to a synthetic complexity that drastically lowers the productive rate and invariably results in a final product containing between 2% and 5% impurities, which is unacceptable for high-grade pharmaceutical applications. A second existing technology attempts to generate sulfo-succinyl oxide through the cyclization of thiosuccinate in acetic anhydride, followed by a reaction with N-azanol. This pathway is equally problematic as it generates a plurality of sulfo-hydroxamic acid isomers, creating a messy mixture that requires rigorous separation. Furthermore, the overall yield of this entire technological sequence is reported to be merely 30%, representing a massive inefficiency in raw material utilization and processing time that drives up costs for downstream users.
The Novel Approach
In stark contrast to these inefficient legacy methods, the novel approach detailed in the patent utilizes N-acyloxymaleimide, a raw material characterized by its relative stability, ease of preparation, and inherently higher purity profile. This method allows for the direct reaction and simultaneous hydrolysis with sodium thiosulfate or potassium thiosulfate in a mixed solvent system comprising water and lower alcohols. By bypassing the unstable N-hydroxymaleimide intermediate entirely, the process achieves a quantitative generation of the product, effectively eliminating the yield losses associated with isomeric byproducts. The reaction conditions are mild, often proceeding at room temperature or with gentle heating around 50 degrees Celsius, which reduces energy consumption and thermal stress on the reagents. This streamlined pathway not only simplifies the operational procedure but also ensures that the final N-hydroxy thiosuccinimide salt is obtained in a highly purified state, ready for immediate use in sensitive biological systems without extensive downstream purification.
Mechanistic Insights into Thiosulfate-Maleimide Addition and Hydrolysis
The core of this synthetic advancement lies in the nucleophilic addition of the thiosulfate anion to the electron-deficient double bond of the N-acyloxymaleimide ring. In this mechanism, the sulfur atom of the thiosulfate acts as a potent nucleophile, attacking the alkene functionality of the maleimide derivative to form a stable carbon-sulfur bond. This step creates the N-acyloxy thiosuccinimide salt intermediate, which precipitates out of the water-alcohol solvent mixture due to its reduced solubility, driving the equilibrium forward and ensuring high conversion rates. The presence of the acyloxy group at the nitrogen position serves as a protecting group that stabilizes the molecule during this initial addition phase, preventing the premature degradation observed in unprotected hydroxyl variants. This mechanistic stability is crucial for maintaining the structural integrity of the succinimide ring throughout the reaction, allowing for the isolation of a clean intermediate that can be stored or processed further without significant loss of quality.
Following the formation of the intermediate, the process proceeds to a hydrolysis step where the N-acyloxy group is cleaved to reveal the active N-hydroxy functionality. This is achieved by refluxing the intermediate salt in a mixture of water and a short-chain organic acid, such as acetic acid or propionic acid. The acidic environment facilitates the nucleophilic attack of water on the carbonyl carbon of the acyloxy group, resulting in the release of the corresponding carboxylic acid and the formation of the target N-hydroxy thiosuccinimide. The final product, now bearing the free hydroxyl group adjacent to the carbonyl, is highly reactive towards amines, making it an ideal activated ester for bioconjugation. The ability to control this hydrolysis precisely ensures that the sulfonate group remains intact while the activating group is perfectly positioned, resulting in a reagent that combines water solubility with high reactivity, a dual property essential for modifying proteins in aqueous environments.
How to Synthesize N-Hydroxy Thiosuccinimide Salt Efficiently
The synthesis protocol outlined in the patent offers a flexible operational framework that can be adapted for both batch and continuous processing environments. The procedure begins with the suspension of equimolar amounts of N-acyloxymaleimide and a thiosulfate salt in a solvent system typically composed of water and ethanol or propanol. This mixture is stirred, often overnight or for approximately three hours depending on the temperature, allowing the nucleophilic addition to reach completion. The resulting precipitate can be filtered and dried to isolate the intermediate, or the process can be telescoped into a single pot where the solvent is removed via rotary evaporation before adding the acidic hydrolysis medium. This operational flexibility allows manufacturers to optimize their workflow based on available equipment and throughput requirements, ensuring that the transition from laboratory scale to commercial production is seamless and efficient.
- Mix equimolar N-acyloxymaleimide and sodium or potassium thiosulfate in a water-alcohol mixed solvent at room temperature or 50°C to form N-acyloxy thiosuccinimide salt.
- Filter and dry the precipitated intermediate salt, or proceed directly to hydrolysis if using a one-pot method.
- Reflux the intermediate in water and organic acid (e.g., acetic acid) to hydrolyze and separate out the final high-purity N-hydroxy thiosuccinimide salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a compelling value proposition centered on cost efficiency and supply reliability. The shift from unstable, low-yield starting materials to stable, high-purity N-acyloxymaleimides fundamentally alters the cost structure of production by minimizing waste and maximizing output per batch. Because the reaction proceeds to quantitative generation, there is a drastic reduction in the volume of raw materials required to produce a specific mass of the final active ingredient, leading to substantial cost savings in material procurement. Furthermore, the simplicity of the workup, which relies on standard filtration and crystallization techniques rather than complex chromatographic separations, significantly reduces processing time and labor costs. This efficiency translates directly into a more competitive pricing structure for the end customer while maintaining healthy margins for the manufacturer.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps and the avoidance of low-yield cyclization reactions result in a significantly streamlined manufacturing process. By utilizing common, inexpensive solvents like water and ethanol instead of specialized anhydrides or hazardous reagents, the overall operational expenditure is drastically lowered. The quantitative nature of the reaction means that nearly every mole of starting material is converted into valuable product, effectively removing the financial burden of disposing of large quantities of unreacted feedstock or byproducts. This economic efficiency allows for a more aggressive pricing strategy in the global market for bioconjugation reagents.
- Enhanced Supply Chain Reliability: The use of stable raw materials such as N-acyloxymaleimide ensures that production schedules are not disrupted by the degradation of inventory prior to use. Unlike N-hydroxymaleimide, which requires careful handling and rapid usage to avoid decomposition, the precursors in this new method have a long shelf life, enabling manufacturers to maintain robust safety stocks. This stability guarantees a continuous supply of the final N-hydroxy thiosuccinimide salt, reducing lead times for customers who depend on these reagents for time-sensitive research and development projects. The predictability of the synthesis also aids in accurate demand forecasting and inventory management.
- Scalability and Environmental Compliance: The process is inherently green, utilizing water as a primary solvent and generating minimal hazardous waste compared to traditional organic synthesis routes. The absence of heavy metal catalysts and the use of benign organic acids for hydrolysis simplify wastewater treatment and regulatory compliance, reducing the environmental overhead associated with production. This eco-friendly profile is increasingly important for multinational corporations aiming to meet strict sustainability goals. Additionally, the straightforward nature of the reaction conditions, which do not require extreme pressures or temperatures, facilitates easy scale-up from pilot plants to multi-ton commercial facilities without the need for specialized high-pressure reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of N-hydroxy thiosuccinimide salts using this advanced methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this reagent into their supply chains and research workflows.
Q: Why is N-acyloxymaleimide preferred over N-hydroxymaleimide as a starting material?
A: N-acyloxymaleimide is significantly more stable and easier to prepare with higher purity compared to N-hydroxymaleimide, which decomposes rapidly at room temperature and often contains 2%-5% impurities.
Q: What represents the primary advantage of this synthesis route regarding yield?
A: The process allows for quantitative generation of the product, avoiding the low yields (around 30%) associated with traditional thiosuccinic acid cyclization methods.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of common solvents like water and ethanol, along with simple filtration and reflux steps, makes the process highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Hydroxy Thiosuccinimide Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality cross-linking reagents play in the development of next-generation biologics and diagnostic tools. Our technical team has thoroughly analyzed the synthetic route described in CN100480239C and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient process to the global market. We are committed to delivering N-hydroxy thiosuccinimide salt that meets stringent purity specifications, ensuring that your bioconjugation reactions proceed with maximum efficiency and reproducibility. Our rigorous QC labs employ advanced analytical techniques to verify the absence of isomeric impurities and confirm the structural integrity of every batch we produce.
We invite you to collaborate with us to optimize your supply chain for these essential chemical building blocks. By leveraging our manufacturing expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to evaluate how our superior manufacturing process can enhance your R&D productivity and reduce overall project costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
