Advanced Iridium-Catalyzed Synthesis of Chiral Bisallyl Compounds for Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for constructing complex molecular architectures, particularly those featuring multiple contiguous stereocenters. Patent CN113861075A introduces a groundbreaking approach to synthesizing chiral bisallyl substituted compounds possessing three consecutive chiral centers. This technology addresses a critical bottleneck in modern organic synthesis, where steric interference and spatial hindrance often compromise yield and selectivity. By leveraging a sophisticated dual iridium-catalyzed system, this method enables the precise assembly of highly functionalized scaffolds essential for the development of next-generation antidepressants, antitumor agents, and natural products. For R&D directors and procurement specialists, understanding this innovation is key to securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity building blocks with exceptional stereochemical control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of compounds with multiple consecutive chiral centers has been fraught with challenges. Conventional routes often rely on stoichiometric chiral auxiliaries or resolution techniques, which inherently limit atom economy and increase waste generation. Furthermore, establishing stereochemistry at adjacent positions typically suffers from poor diastereoselectivity due to the lack of effective catalysts that can differentiate between similar steric environments. The accumulation of intramolecular steric repulsion in such densely functionalized molecules frequently leads to sluggish reaction kinetics and the formation of complex mixtures of stereoisomers. This not only complicates downstream purification but also drastically inflates the cost of goods sold (COGS) by reducing overall process efficiency. For supply chain heads, these inefficiencies translate into longer lead times and higher risks of batch-to-batch variability, making scale-up a formidable obstacle.
The Novel Approach
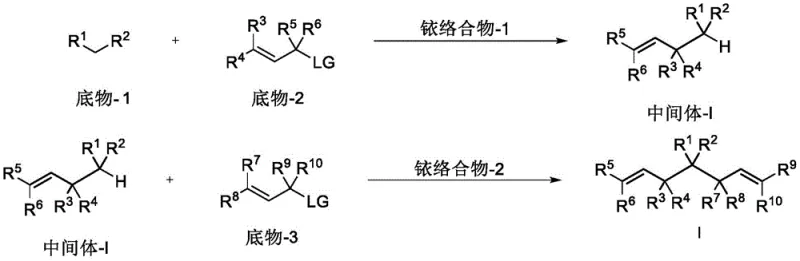
In stark contrast, the methodology disclosed in CN113861075A utilizes a sequential iridium-catalyzed asymmetric allylic alkylation strategy. This novel approach employs chiral phosphoramidite ligands coordinated with iridium salts to create a highly defined chiral pocket around the metal center. The process begins with the reaction of substrate-1 and substrate-2 to form a key intermediate, which then undergoes a second catalytic cycle with substrate-3. This tandem catalytic sequence allows for the independent control of stereocenters, effectively overcoming the steric barriers that plague traditional methods. The result is a streamlined pathway that achieves high yields and excellent diastereoselectivity without the need for cumbersome protection-deprotection sequences. This represents a significant leap forward in cost reduction in pharmaceutical intermediates manufacturing, offering a scalable solution for producing complex chiral motifs.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Allylic Alkylation
The core of this technology lies in the precise modulation of the iridium catalyst's coordination sphere. The patent details the use of specific chiral ligands, such as those depicted in the structural formulas provided, which dictate the facial selectivity of the allylic substitution. 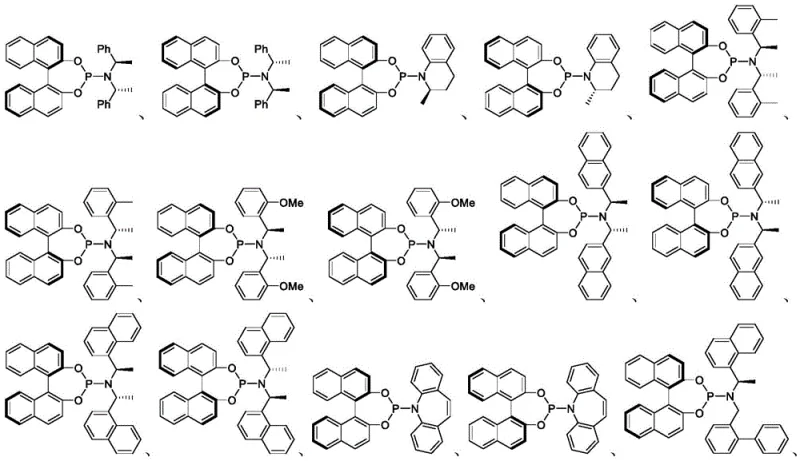 By switching between enantiomeric forms of the ligand (e.g., (S,S,S)-L1 versus (R,R,R)-L1) and varying the order of substrate addition, chemists can access a wide array of stereoisomers. The mechanism involves the oxidative addition of the allylic carbonate to the iridium center, forming a pi-allyl iridium complex. The nucleophile, typically a stabilized carbanion derived from cyanoacetates, then attacks this complex with high regio- and stereocontrol. This level of control is paramount for R&D teams aiming to explore structure-activity relationships (SAR) efficiently, as it allows for the rapid generation of stereodiverse libraries from a common set of starting materials.
By switching between enantiomeric forms of the ligand (e.g., (S,S,S)-L1 versus (R,R,R)-L1) and varying the order of substrate addition, chemists can access a wide array of stereoisomers. The mechanism involves the oxidative addition of the allylic carbonate to the iridium center, forming a pi-allyl iridium complex. The nucleophile, typically a stabilized carbanion derived from cyanoacetates, then attacks this complex with high regio- and stereocontrol. This level of control is paramount for R&D teams aiming to explore structure-activity relationships (SAR) efficiently, as it allows for the rapid generation of stereodiverse libraries from a common set of starting materials.
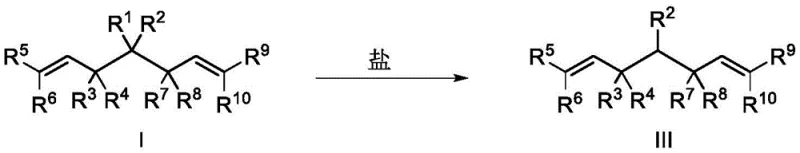
Furthermore, the versatility of the resulting bisallyl structure cannot be overstated. The presence of terminal alkene groups provides multiple handles for subsequent functionalization, such as cross-metathesis, hydrogenation, or oxidation. The patent further illustrates how the ester moiety in Formula I can be hydrolyzed under acidic conditions to yield carboxylic acids (Formula II) or subjected to decarboxylation in the presence of alkali metal salts to afford Formula III. 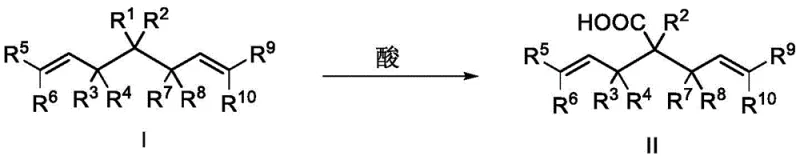
 These transformations expand the utility of the core scaffold, enabling the synthesis of chiral unnatural amino acids and other bioactive fragments. The ability to tolerate diverse functional groups, including halogens and heterocycles, ensures that this chemistry is compatible with late-stage functionalization strategies often required in medicinal chemistry campaigns.
These transformations expand the utility of the core scaffold, enabling the synthesis of chiral unnatural amino acids and other bioactive fragments. The ability to tolerate diverse functional groups, including halogens and heterocycles, ensures that this chemistry is compatible with late-stage functionalization strategies often required in medicinal chemistry campaigns.
How to Synthesize Chiral Bisallyl Substituted Compounds Efficiently
Implementing this synthesis requires careful attention to catalyst preparation and reaction conditions. The protocol involves generating the active iridium species in situ by mixing an iridium salt like [Ir(COD)Cl]2 with the chiral ligand in a degassed solvent. Subsequent addition of the substrates and a mild base initiates the coupling. The reaction proceeds under inert atmosphere at moderate temperatures, typically ranging from -20°C to 110°C depending on the specific step and substrate reactivity. Detailed standard operating procedures for scaling this reaction from milligram to kilogram quantities are critical for ensuring reproducibility and safety. The following guide outlines the fundamental steps required to execute this transformation effectively in a laboratory or pilot plant setting.
- Perform the first iridium-catalyzed allylation between substrate-1 and substrate-2 using chiral ligand L1 to generate Intermediate-I.
- Conduct the second catalytic reaction between Intermediate-I and substrate-3 using a specific iridium complex configuration to establish the third chiral center.
- Optionally hydrolyze the ester group with acid or perform decarboxylation with alkali metal salts to obtain diverse chiral derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers tangible strategic benefits beyond mere chemical elegance. The shift towards catalytic asymmetric synthesis eliminates the need for stoichiometric chiral reagents, which are often expensive and generate significant waste. This fundamentally alters the cost structure of the manufacturing process, leading to substantial cost savings in raw material procurement. Moreover, the high selectivity of the reaction minimizes the formation of unwanted diastereomers, thereby simplifying the purification workflow. Reduced purification complexity means less solvent consumption and shorter processing times, which directly contributes to enhanced supply chain reliability and faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The use of low-loading iridium catalysts significantly reduces the reliance on precious metals compared to stoichiometric methods. Since the catalyst loading is minimal (often less than 1 mol%), the overall metal cost per kilogram of product is drastically lowered. Additionally, the high atom economy of the allylic alkylation reaction ensures that most of the starting material mass is incorporated into the final product, minimizing waste disposal costs. This efficiency translates into a more competitive pricing structure for high-purity pharmaceutical intermediates, allowing partners to optimize their budget allocation for R&D activities.
- Enhanced Supply Chain Reliability: The robustness of this method across a broad range of substrates mitigates the risk of supply disruptions caused by specialized reagent shortages. Because the chemistry tolerates various functional groups and heterocycles, manufacturers are not locked into a single, fragile synthetic route. This flexibility allows for the sourcing of alternative starting materials if necessary, ensuring continuity of supply. Furthermore, the scalability of the process from small-scale discovery to commercial production ensures that demand surges can be met without compromising quality or delivery schedules, a critical factor for maintaining uninterrupted drug manufacturing pipelines.
- Scalability and Environmental Compliance: The reaction conditions described are amenable to large-scale operation, utilizing common organic solvents and standard reactor equipment. The elimination of harsh reagents and the reduction of chemical waste align with green chemistry principles, facilitating easier regulatory approval and environmental compliance. This is increasingly important as global regulations tighten around pharmaceutical manufacturing emissions. By adopting this cleaner technology, companies can future-proof their supply chains against evolving environmental standards while maintaining high production throughput and operational efficiency.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of this synthesis method. These insights are derived directly from the patent data to provide clarity on implementation and potential applications. Understanding these details helps stakeholders make informed decisions regarding technology transfer and partnership opportunities.
Q: What is the primary advantage of this iridium-catalyzed method over conventional synthesis?
A: The primary advantage is the ability to construct three consecutive chiral centers with high diastereoselectivity and stereodiversity, accessing up to six of eight possible stereoisomers, which is extremely difficult with traditional methods due to steric hindrance.
Q: Can this method be applied to heterocyclic substrates common in drug discovery?
A: Yes, the patent explicitly states that the method tolerates a wide variety of substrates, including important heterocyclic systems frequently found in medicinal chemistry, ensuring broad applicability for API intermediate synthesis.
Q: How does this process impact the production of unnatural amino acids?
A: The resulting chiral bisallyl compounds serve as versatile precursors that can be easily converted into chiral unnatural amino acids and derivatives, providing a streamlined route for developing novel peptide-based therapeutics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bisallyl Substituted Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN113861075A. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle sensitive organometallic reactions under strict inert conditions, ensuring that the integrity of the chiral centers is maintained throughout the manufacturing process. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral bisallyl compounds meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your timeline to market with reliable, high-quality intermediates produced through sustainable and efficient chemistry.
