Advanced Lewis Acid-Catalyzed Nitration for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
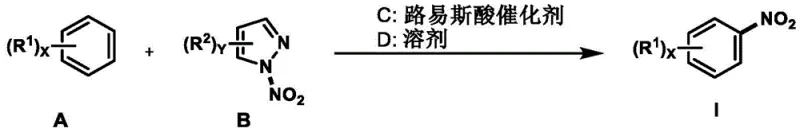
The landscape of aromatic nitration is undergoing a significant transformation driven by the urgent need for greener, more selective synthetic methodologies in the fine chemical and pharmaceutical sectors. Patent CN114276251A introduces a groundbreaking protocol for the synthesis of nitro(hetero)arenes that circumvents the harsh limitations of traditional mixed-acid systems. By leveraging a novel combination of substituted pyrazole-based nitrating reagents and catalytic amounts of Lewis acids, this technology enables the efficient introduction of nitro groups onto simple and heterocyclic aromatic scaffolds under remarkably mild conditions. The process operates effectively at temperatures ranging from 40°C to 100°C, utilizing commercially available solvents and inert gas protection to ensure safety and reproducibility. This innovation represents a pivotal shift towards sustainable manufacturing, offering a robust pathway for producing high-value intermediates essential for drug discovery and agrochemical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial standard for aromatic nitration has relied heavily on the use of mixed mineral acids, specifically concentrated sulfuric and nitric acid, to generate the active electrophilic nitronium ion. While effective for simple substrates, this legacy approach suffers from severe drawbacks that hinder modern pharmaceutical manufacturing. The highly corrosive nature of these acids necessitates specialized, expensive reactor materials and rigorous safety protocols to manage exothermic risks. Furthermore, the strong oxidizing environment often leads to poor chemoselectivity, resulting in the oxidation of sensitive functional groups such as hydroxyls or sulfides, which are common in complex drug intermediates. The generation of massive quantities of acidic waste streams also poses significant environmental compliance challenges and increases the cost of waste treatment, making the process economically and ecologically unsustainable for high-value fine chemical production.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a discrete, stable nitrating reagent, specifically derivatives like 5-methyl-1,3-dinitropyrazole, activated by Lewis acid catalysts. This system eliminates the need for bulk mineral acids, thereby drastically reducing corrosion risks and waste generation. The reaction proceeds through a controlled electrophilic substitution mechanism where the Lewis acid coordinates with the nitrating agent to facilitate the transfer of the nitro group with high precision. This approach allows for exceptional functional group tolerance, enabling the nitration of electron-deficient and electron-rich arenes alike without compromising sensitive moieties. The modularity of the system, compatible with various solvents like acetonitrile and hexafluoroisopropanol, provides process chemists with the flexibility to optimize solubility and reaction kinetics for diverse substrate classes, ensuring high yields and purity profiles suitable for GMP manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Electrophilic Nitration
The core of this technological advancement lies in the synergistic interaction between the Lewis acid catalyst and the nitrogen-heterocycle-derived nitrating reagent. Unlike traditional proton-catalyzed pathways, the Lewis acid (such as In(OTf)3 or Yb(OTf)3) acts as a potent activator by coordinating with the oxygen or nitrogen atoms of the nitrating reagent. This coordination polarizes the N-O or N-N bond, significantly lowering the energy barrier for the generation of the nitronium equivalent or a highly reactive nitro-complex. This activated species then attacks the aromatic ring in a classic electrophilic aromatic substitution manner. The choice of catalyst is critical; rare earth triflates are particularly effective due to their high oxophilicity and stability in organic media, allowing them to turnover efficiently without being deactivated by water or byproducts. This mechanistic pathway ensures that the nitration occurs regioselectively, often favoring the para- or ortho-positions depending on the electronic nature of the substrate substituents.
Furthermore, the mechanism inherently supports a clean impurity profile, which is a paramount concern for R&D directors overseeing API intermediate synthesis. The mild reaction conditions prevent side reactions such as over-nitration or oxidative degradation that plague mixed-acid methods. For instance, substrates containing phenolic hydroxyl groups, which are prone to oxidation under harsh acidic conditions, remain intact throughout the process. The use of protective gases like argon further suppresses radical-mediated side reactions, ensuring that the product distribution is dominated by the desired mono-nitrated species. This high level of control simplifies downstream purification, often allowing for straightforward filtration and crystallization rather than complex chromatographic separations, thereby enhancing the overall process mass intensity and reducing the environmental footprint of the synthesis.
How to Synthesize Nitro(Hetero)arenes Efficiently
Implementing this synthesis route requires precise control over stoichiometry and reaction parameters to maximize yield and minimize impurities. The process begins with the careful selection of the Lewis acid catalyst and solvent system tailored to the specific electronic properties of the starting arene. Operators must maintain an inert atmosphere to prevent moisture ingress, which could deactivate the Lewis acid catalyst. The reaction temperature should be ramped gradually to the target range of 80°C to ensure smooth initiation without thermal runaway. Following the reaction period, typically around 16 hours, the workup procedure involves simple filtration through celite to remove metal salts, followed by solvent removal and purification. For detailed operational parameters and specific stoichiometric ratios for various substrates, please refer to the standardized guide below.
- Combine simple (hetero)arene substrate, 5-methyl-1,3-dinitropyrazole nitrating reagent, and Lewis acid catalyst (e.g., In(OTf)3 or Yb(OTf)3) in an organic solvent like acetonitrile or HFIP under inert atmosphere.
- Stir the reaction mixture at a controlled temperature between 40°C and 100°C for 1 to 72 hours until conversion is complete.
- Upon completion, filter the mixture through Celite, wash with ethyl acetate, remove solvent under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers transformative benefits that directly impact the bottom line and operational resilience. The shift away from hazardous mineral acids to stable, solid nitrating reagents simplifies logistics and storage requirements, reducing the regulatory burden associated with transporting corrosive liquids. The use of catalytic amounts of Lewis acids, rather than stoichiometric quantities of reagents, significantly lowers the raw material cost per kilogram of product. Moreover, the high selectivity of the reaction reduces the formation of difficult-to-remove isomers and byproducts, leading to higher overall yields and reduced solvent consumption during purification. These factors collectively contribute to a more cost-effective manufacturing process that is less susceptible to supply chain disruptions caused by strict environmental regulations on acid waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive corrosion-resistant equipment and the reduction in waste treatment costs drive substantial savings. By using catalytic Lewis acids and avoiding bulk mineral acids, the process minimizes the consumption of auxiliary chemicals and reduces the load on effluent treatment plants. The high atom economy and selectivity mean less raw material is wasted on unwanted byproducts, directly lowering the cost of goods sold (COGS) for high-purity nitroarene intermediates.
- Enhanced Supply Chain Reliability: The reagents employed, such as 5-methyl-1,3-dinitropyrazole and common Lewis acid triflates, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors, which are widely available in CDMO networks, ensuring that production capacity can be scaled rapidly to meet demand without requiring specialized infrastructure investments.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exotherm management and lack of hazardous gas evolution. The simplified workup procedure, involving filtration and concentration, is easily adaptable to large-scale batch or continuous flow processing. Furthermore, the significant reduction in acidic waste aligns with increasingly stringent global environmental standards, facilitating smoother regulatory approvals and long-term sustainability goals for manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this nitration technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on catalyst selection, substrate compatibility, and operational safety. Understanding these nuances is critical for process development teams evaluating this route for potential technology transfer or scale-up activities.
Q: What are the optimal reaction conditions for this nitration method?
A: The process operates under mild conditions, typically requiring temperatures between 40°C and 100°C (preferably 80°C) for 1 to 72 hours (preferably 16 hours) under an argon or nitrogen protective atmosphere.
Q: Which Lewis acid catalysts are most effective for this transformation?
A: Rare earth metal triflates such as Indium(III) triflate (In(OTf)3) and Ytterbium(III) triflate (Yb(OTf)3) are preferred, though Copper, Zinc, and Scandium salts also demonstrate efficacy depending on the substrate.
Q: Does this method tolerate sensitive functional groups on the aromatic ring?
A: Yes, the method exhibits excellent functional group compatibility, successfully nitrating substrates containing hydroxyl, ester, halogen, trifluoromethyl, and sulfhydryl groups without significant degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitro(Hetero)arene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization. Our technical team has extensively evaluated the Lewis acid-catalyzed nitration pathway described in CN114276251A and confirmed its viability for producing complex pharmaceutical intermediates with exceptional purity. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of nitro(hetero)arene meets the highest international quality standards required by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this greener nitration method can optimize your budget. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us be your strategic partner in delivering high-quality chemical solutions that drive innovation and efficiency in your supply chain.
