Advanced Catalytic Oxidation Strategy for Commercial Scale Production of High-Purity Oxo-Ionones
Advanced Catalytic Oxidation Strategy for Commercial Scale Production of High-Purity Oxo-Ionones
The fine chemical industry is constantly seeking more efficient and environmentally benign pathways for producing high-value intermediates, particularly in the fragrance and pharmaceutical sectors. A pivotal advancement in this domain is detailed in patent CN101805249B, which discloses a novel method for synthesizing oxo-alpha-ionone and oxo-beta-ionone. These compounds serve as critical building blocks for tobacco flavorings, food additives, and complex carotenoid synthesis used in medicine and material science. The patented technology replaces hazardous traditional oxidants with a sodium bromate system in a hydrophilic organic solvent, achieving high yields under mild conditions. This innovation not only addresses the growing regulatory pressure against heavy metal usage but also streamlines the manufacturing workflow, offering a robust solution for reliable oxo-ionone supplier networks globally.
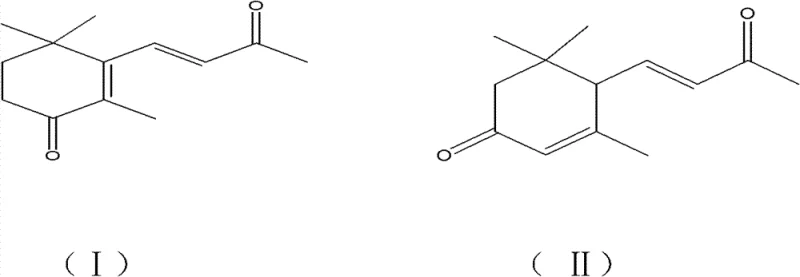
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxo-ionones has relied heavily on oxidation protocols that pose significant safety and environmental challenges. Conventional techniques often employ vanadium-based oxidation styles or molecular oxygen oxidation methods, which frequently suffer from unsatisfactory productivity and tediously long processing times. More critically, many established routes utilize chromium-based reagents such as chromium trioxide, chromic acid tert-butyl, or pyridinium chlorochromate (PCC). These substances are notoriously toxic and generate hazardous waste streams that require complex and costly disposal procedures. Furthermore, prior art methods, such as those utilizing chloroform solvents with vanadium-potassium iodide systems, have demonstrated reaction times extending up to 24 hours with yields often falling below 40%, creating a bottleneck for cost reduction in flavor and fragrance manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN101805249B introduces a paradigm shift by utilizing sodium bromate as a single, effective oxidant within a hydrophilic organic solvent system. This approach operates at temperatures below 80°C, typically optimized between 35°C and 60°C, drastically reducing energy consumption compared to harsher thermal conditions. The reaction completes within a mere 0.5 to 12 hours, with optimal results achieved in just 1 to 3 hours, representing a substantial improvement in throughput efficiency. By avoiding virulent solvents like chloroform and eliminating heavy metal catalysts, this process ensures that the byproduct is merely sodium bromide, an environmentally safe salt that can be easily managed or repurposed, thereby aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Sodium Bromate Mediated Oxidation
The core of this synthetic breakthrough lies in the precise control of oxidative potential through acid catalysis. The mechanism relies on the generation of active bromine species from sodium bromate in the presence of a mineral or organic acid, such as sulfuric acid. The oxidizing power of the bromate ion is intrinsically linked to the acidity of the reaction medium; by maintaining the pH value of the reaction system between 1 and 3, the process ensures a high concentration of the active oxidizing species necessary to attack the allylic positions of the ionone ring. This controlled acidity prevents over-oxidation or degradation of the sensitive conjugated double bond systems present in the ionone structure, thereby preserving the integrity of the molecule while selectively introducing the ketone functionality at the desired position.
Furthermore, the choice of solvent plays a pivotal role in the mechanistic pathway and impurity profile. The use of hydrophilic organic solvents like tetrahydrofuran (THF), acetone, or acetonitrile facilitates the homogeneity of the reaction mixture, ensuring efficient mass transfer between the organic substrate and the aqueous acid phase. This homogeneous environment minimizes the formation of side products such as epoxy derivatives, which are often observed in less optimized systems. For instance, experimental data indicates that using THF as the solvent yields significantly higher purity profiles compared to ethanol, demonstrating that solvent polarity and coordination ability are critical parameters for maximizing the selectivity of the oxidation and achieving the reported product purity of not less than 98%.
How to Synthesize Oxo-Ionones Efficiently
The operational simplicity of this synthesis route makes it highly attractive for immediate adoption in pilot and commercial plants. The process begins by preparing a mixed solvent system where the hydrophilic organic solvent accounts for approximately 60% of the total volume, combined with an acid solution to establish the requisite acidic environment. The starting material, either alpha or beta-ionone, is introduced under magnetic agitation, followed by the slow, controlled addition of sodium bromate to manage the exotherm and reaction rate. Detailed standardized synthetic steps, including specific molar ratios and workup procedures like separation and rotary evaporation, are essential for replicating the high yields described in the patent documentation.
- Prepare a mixed solvent system comprising a hydrophilic organic solvent (such as THF or acetone) and an acid solution (e.g., 7% sulfuric acid) to maintain a reaction pH between 1 and 3.
- Introduce the starting material, alpha or beta-ionone, into the solvent mixture under magnetic agitation, ensuring complete dissolution before initiating the oxidation.
- Slowly add sodium bromate as the oxidant while controlling the temperature between 35°C and 60°C, allowing the reaction to proceed for 1 to 3 hours until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this sodium bromate-based protocol offers compelling economic and logistical benefits. The elimination of expensive and regulated heavy metal catalysts removes the need for specialized purification steps, such as metal scavenging columns or complex extraction sequences, which traditionally inflate the cost of goods sold. Additionally, the use of commodity chemicals like sodium bromate and sulfuric acid ensures a stable and resilient supply chain, as these reagents are widely available globally and are not subject to the same geopolitical restrictions or scarcity issues as rare transition metals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the downstream processing. Since the oxidation byproduct is sodium bromide, a water-soluble salt, the workup involves simple washing and phase separation rather than complex chromatographic purifications. This reduction in unit operations translates directly to lower labor costs, reduced solvent consumption, and decreased utility usage, resulting in substantial cost savings for high-purity oxo-ionones production without compromising on quality standards.
- Enhanced Supply Chain Reliability: By relying on a robust chemical platform that avoids niche or hazardous reagents, manufacturers can significantly mitigate supply risks. The raw materials required for this synthesis are bulk commodities with established logistics networks, ensuring consistent availability even during market fluctuations. This reliability allows for better production planning and inventory management, effectively reducing lead time for high-purity oxo-ionones and enabling suppliers to meet tight delivery schedules demanded by major flavor and fragrance houses.
- Scalability and Environmental Compliance: From a regulatory standpoint, this method offers a clear path to sustainable manufacturing. The absence of chromium and other toxic metals simplifies environmental compliance and waste disposal, lowering the overhead associated with environmental health and safety (EHS) protocols. The mild reaction conditions and short cycle times further enhance the scalability of the process, allowing for seamless commercial scale-up of complex flavor intermediates from kilogram to multi-ton scales while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational parameters and quality outcomes expected from this synthesis route.
Q: What are the primary advantages of the sodium bromate oxidation method over traditional chromium-based processes?
A: The sodium bromate method eliminates the use of toxic heavy metal oxidants like chromium trioxide or pyridinium chlorochromate (PCC), resulting in a significantly cleaner environmental profile and simplifying waste treatment protocols.
Q: How does the acidity of the reaction system influence the oxidation efficiency?
A: The oxidizing power of bromate is directly correlated with the acidity of the medium; maintaining a pH between 1 and 3 ensures sufficient oxidative strength to drive the reaction to completion within a shortened timeframe of 1 to 3 hours.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes common, inexpensive reagents and mild reaction conditions below 80°C, making it highly scalable with simplified post-reaction workup procedures that facilitate commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxo-Ionone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in maintaining a competitive edge in the fine chemical market. Our technical team has extensively analyzed the sodium bromate oxidation pathway and possesses the expertise to optimize this chemistry for your specific production needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of oxo-ionone meets the exacting standards required for food, cosmetic, and pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can drive value and efficiency for your organization.
