Advanced Surfactant-Assisted Oxidation for Commercial Nopinone Production
The global demand for high-purity terpene derivatives continues to surge, driven by their critical applications in the fragrance, flavor, and pharmaceutical sectors. Within this landscape, the efficient synthesis of Nopinone stands out as a pivotal challenge for process chemists seeking to optimize yield and minimize waste. A significant technological breakthrough in this domain is detailed in patent CN110256215B, which discloses a novel preparation method utilizing acidic potassium permanganate oxidation facilitated by surfactant catalysts. This approach represents a paradigm shift from traditional oxidative protocols, offering a robust pathway that achieves a beta-pinene conversion rate exceeding 99% and a Nopinone selectivity greater than 90%. For R&D directors and procurement strategists, this patent data underscores a viable route to secure a reliable nopinone supplier capable of delivering consistent quality while adhering to stringent environmental and economic constraints. The integration of surfactant technology not only accelerates reaction kinetics but also fundamentally alters the interfacial dynamics of the oxidation process, ensuring that industrial partners can achieve superior outcomes without compromising on safety or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of beta-pinene to Nopinone has been plagued by significant inefficiencies that hinder cost-effective manufacturing. Traditional methods often rely on stoichiometric amounts of strong oxidants like potassium permanganate without adequate phase-transfer mediation, leading to heterogeneous reaction mixtures where mass transfer becomes the rate-limiting step. In these conventional setups, the hydrophobic nature of beta-pinene restricts its interaction with the aqueous oxidant phase, necessitating prolonged reaction times often exceeding 5 hours to reach acceptable conversion levels. Furthermore, the lack of precise control over the oxidation environment frequently results in over-oxidation, generating undesirable byproducts such as nopinic acid and various diols, which drastically complicate the purification process and reduce overall yield. These inefficiencies translate directly into higher operational expenditures due to increased energy consumption for extended stirring and heating, as well as the substantial costs associated with separating complex impurity profiles. Consequently, manufacturers relying on these legacy processes struggle to maintain competitive pricing margins in the volatile market for fragrance intermediates.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110256215B introduces a sophisticated surfactant-assisted strategy that effectively resolves the mass transfer limitations inherent in previous techniques. By incorporating specific surfactants such as cetyltrimethylammonium bromide or 2-bromoethyltrimethylammonium bromide, the process creates a micro-emulsion or improved interfacial area that allows the oxidant to access the substrate with unprecedented efficiency. This innovation enables the reaction to proceed rapidly within a温和 window of 1 to 4 hours at ambient temperatures ranging from 20 to 30°C, eliminating the need for energy-intensive heating or cryogenic cooling systems. The result is a dramatic improvement in process metrics, with beta-pinene conversion consistently surpassing 99% and product selectivity maintained above 90%. This novel approach not only streamlines the synthetic route but also significantly reduces the burden on downstream processing units, as the cleaner reaction profile minimizes the formation of stubborn byproducts. For supply chain managers, this translates to a more predictable and agile production schedule, essential for meeting the rigorous delivery timelines demanded by multinational clients.
Mechanistic Insights into Surfactant-Assisted Permanganate Oxidation
The core mechanistic advantage of this patented process lies in the dual role played by the surfactant catalyst within the reaction matrix. In a standard biphasic system involving organic beta-pinene and aqueous potassium permanganate, the reaction is limited by the surface area available at the interface. The introduction of amphiphilic surfactant molecules lowers the interfacial tension, effectively dispersing the organic substrate into the aqueous phase or creating micellar structures that solubilize the reactants. This enhanced proximity facilitates the electrophilic attack of the permanganate ion on the electron-rich double bond of the beta-pinene, initiating the oxidative cleavage sequence with much lower activation energy. Furthermore, the acidic environment, maintained by the addition of sulfuric or hydrochloric acid, ensures that the permanganate species remains in its most reactive protonated forms, further accelerating the kinetics. This synergistic effect between the surfactant and the acidic medium allows for the precise tuning of reaction selectivity, favoring the formation of the ketone functionality in Nopinone while suppressing deeper oxidation pathways that would lead to carboxylic acids.
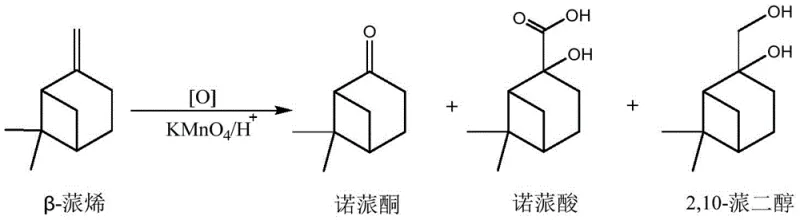
Controlling the impurity profile is paramount for producing high-purity nopinone suitable for sensitive applications in fine fragrances and pharmaceuticals. The mechanistic design of this process inherently limits the generation of over-oxidized species such as nopinic acid, which typically arises from the further oxidation of the initial ketone product or the glycol intermediate. By strictly regulating the reaction temperature to remain between 20 and 30°C, the thermal energy available for secondary oxidation reactions is kept below the threshold required for significant byproduct formation. Additionally, the rapid consumption of the starting material due to the catalytic effect of the surfactant means that the exposure time of the newly formed Nopinone to the oxidizing environment is minimized. Post-reaction, the quenching step using sodium sulfite effectively neutralizes any residual permanganate, preventing post-process degradation during workup. This rigorous control over the reaction trajectory ensures that the crude product requires minimal purification, thereby preserving the integrity of the molecular structure and maximizing the recovery of the target compound.
How to Synthesize Nopinone Efficiently
The practical implementation of this synthesis route offers a straightforward yet highly effective protocol for laboratory and pilot-scale operations. The process begins with the dissolution of the surfactant catalyst in acetone, followed by the addition of beta-pinene to form a homogeneous or semi-homogeneous mixture. The oxidant, acidic potassium permanganate, is then introduced gradually to manage the exotherm, although the mild conditions generally keep temperature spikes manageable. Detailed standard operating procedures regarding specific molar ratios, agitation speeds, and workup parameters are critical for reproducing the high yields reported in the patent literature. For technical teams looking to adopt this methodology, understanding the nuances of the extraction and washing steps is equally important to ensure the removal of manganese dioxide sludge and inorganic salts. The comprehensive guide below outlines the standardized steps required to achieve optimal results, serving as a foundational reference for scaling this technology to commercial production levels.
- Mix beta-pinene with a surfactant catalyst (e.g., CTAB) in acetone solvent, then add acidic potassium permanganate solution while maintaining temperature between 20-30°C.
- Stir the reaction mixture for 1 to 4 hours to ensure complete conversion of the starting material, monitoring for the disappearance of the purple permanganate color.
- Quench excess oxidant with sodium sulfite, filter off manganese dioxide precipitate, extract with ethyl acetate, and purify via washing and drying to obtain high-purity nopinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this surfactant-assisted oxidation technology presents a compelling value proposition centered on cost optimization and operational reliability. The primary economic driver is the substantial reduction in reaction time, which directly correlates to increased reactor throughput and lower utility costs per kilogram of product. Unlike traditional methods that may require days to complete or necessitate expensive cryogenic equipment, this process operates efficiently at near-ambient temperatures, drastically simplifying the infrastructure requirements for manufacturing facilities. Moreover, the high selectivity of the reaction minimizes the loss of valuable raw materials into waste streams, ensuring that the input cost of beta-pinene is maximized in the final output. This efficiency is crucial for maintaining margin stability in a market where raw material prices can fluctuate unpredictably. By partnering with a manufacturer utilizing this advanced protocol, buyers can secure a more stable supply of intermediates with reduced risk of production bottlenecks.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the reduction in processing time lead to significant operational savings. The use of inexpensive and readily available surfactants replaces the need for costly noble metal catalysts or specialized reagents, further driving down the bill of materials. Additionally, the simplified workup procedure, which involves basic filtration and extraction rather than complex chromatographic separations, reduces labor and solvent consumption costs. These cumulative efficiencies allow for a more competitive pricing structure without sacrificing product quality, providing a distinct advantage in cost reduction in fragrance intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is essential for long-term supply agreements. The high conversion rates mean that there is less variability in yield, allowing production planners to forecast output with greater accuracy. Furthermore, the reliance on commodity chemicals like potassium permanganate and acetone mitigates the risk of supply disruptions associated with exotic or single-source reagents. This resilience is vital for reducing lead time for high-purity nopinones, enabling suppliers to respond swiftly to urgent customer demands and maintain inventory levels that buffer against market volatility.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates manganese dioxide as the primary solid waste, which can be filtered and potentially recycled or disposed of safely, avoiding the release of toxic heavy metals often associated with other oxidation methods. The mild temperature profile reduces the carbon footprint of the manufacturing process by lowering energy consumption for heating and cooling. These factors facilitate easier regulatory compliance and support sustainability goals, making the commercial scale-up of complex terpene derivatives more feasible and environmentally responsible. The ability to scale this process from grams to tons without losing efficiency makes it an ideal candidate for industrial expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Nopinone synthesized via this advanced oxidation method. These insights are derived directly from the experimental data and process descriptions found in the relevant patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding specification requirements and process integration. Whether you are concerned about impurity profiles, scalability limits, or raw material sourcing, the answers below provide a solid foundation for technical discussions.
Q: What is the primary advantage of using surfactants in Nopinone synthesis?
A: Surfactants act as phase transfer catalysts or emulsifiers, significantly improving the contact between the hydrophobic beta-pinene and the aqueous permanganate oxidant. This leads to higher conversion rates (>99%) and better selectivity (>90%) compared to traditional non-catalyzed methods.
Q: How does this method control over-oxidation byproducts?
A: By strictly controlling the reaction temperature (20-30°C) and reaction time (1-4h), the process minimizes the formation of over-oxidized byproducts like nopinic acid, ensuring a cleaner crude product profile that simplifies downstream purification.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the mild reaction conditions eliminate the need for extreme heating or cooling, reducing energy consumption and safety risks associated with exothermic runaway. The simple workup involving filtration and extraction is highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nopinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent concept to commercial reality requires not just chemical expertise, but deep engineering proficiency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the surfactant-assisted oxidation process are fully realized in our manufacturing plants. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality assurance means that every kilogram of Nopinone we deliver meets the exacting requirements of global pharmaceutical and fragrance houses, providing you with a partner who understands the critical nature of supply chain continuity.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to our optimized supply chain. We encourage potential partners to contact us directly to索取 specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal benchmarks. Let us demonstrate how our technical prowess and commercial dedication can drive value for your organization, securing your position as a leader in the fine chemicals market.
