Advanced Catalytic Oxidation for High-Purity Agrochemical Intermediates and Commercial Scale-Up
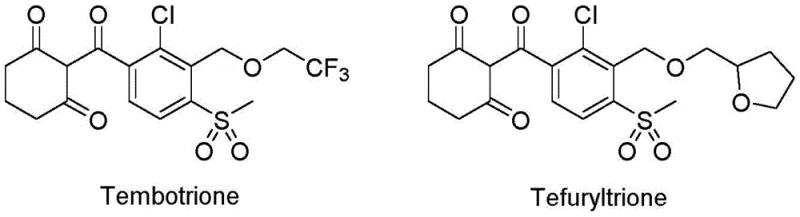
The global demand for high-efficiency herbicides targeting p-hydroxyphenylpyruvate dioxygenase (HPPD) continues to surge, driving the need for robust synthetic routes to critical intermediates like 2-chloro-3-methyl-4-methylsulfonylbenzoic acid. Patent CN112194603B introduces a transformative preparation method that replaces hazardous halogenated oxidants with molecular oxygen, addressing long-standing environmental and efficiency bottlenecks in agrochemical manufacturing. This innovation is particularly relevant for the production of next-generation triketone herbicides such as Tembotrione and Tefuryltrione, which dominate the corn and paddy field weed control markets. By leveraging a manganese and cobalt-catalyzed aerobic oxidation system, this technology ensures complete raw material conversion while eliminating the generation of massive saline wastewater streams associated with legacy processes. For R&D and procurement leaders, this represents a pivotal shift towards sustainable, cost-effective synthesis of complex aromatic acids.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
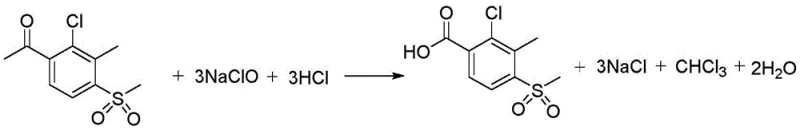
Historically, the synthesis of 2-chloro-3-methyl-4-methylsulfonylbenzoic acid has relied heavily on the haloform reaction mechanism utilizing sodium hypochlorite as the primary oxidant. This conventional approach suffers from severe thermodynamic and kinetic limitations, primarily due to the use of water-soluble solvents like dioxane or tetrahydrofuran which compromise the solubility of the hydrophobic acetophenone starting material. As the reaction progresses and sodium hypochlorite is added, the effective concentration of the organic solvent decreases, inducing premature crystallization of the raw material and resulting in incomplete conversion rates that plague production batches. Furthermore, the environmental footprint of this legacy method is staggering, generating approximately 8.3 tons of strong-acid wastewater containing 1.2 tons of sodium chloride for every single ton of target product manufactured. This immense waste burden creates prohibitive disposal costs and regulatory compliance challenges, making the traditional haloform route increasingly untenable for modern, eco-conscious chemical supply chains.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes molecular oxygen or air as the terminal oxidant within a carboxylic acid solvent system, fundamentally altering the reaction landscape to favor high efficiency and environmental compatibility. By employing acetic acid as the reaction medium, the process maintains a homogeneous phase throughout the oxidation, preventing the crystallization issues that hinder the older sodium hypochlorite method and ensuring that reactants remain fully accessible to the catalytic sites. The substitution of chlorine-based oxidants with oxygen not only eliminates the formation of inorganic salt byproducts but also drastically simplifies the downstream purification process, as the primary byproduct is formaldehyde which can be easily managed or recycled. This strategic shift allows for reaction temperatures between 50°C and 150°C and pressures up to 2.5 MPa, creating a versatile operational window that supports both laboratory optimization and large-scale industrial implementation without the baggage of toxic halogenated waste.
Mechanistic Insights into Mn/Co-Catalyzed Aerobic Oxidation
The core of this technological breakthrough lies in the synergistic catalytic cycle driven by manganese and cobalt species, which activate molecular oxygen to selectively oxidize the acetyl methyl group into a carboxylic acid functionality. In this mechanism, the metal catalysts, typically introduced as acetates, facilitate the formation of radical intermediates that abstract hydrogen atoms from the methyl group, allowing for sequential oxygen insertion without over-oxidation or ring degradation. The presence of the carboxylic acid solvent is critical not just for solubility, but also for stabilizing the high-valent metal-oxo species required for efficient turnover, thereby maintaining a rapid and smooth reaction profile even at moderate temperatures. This catalytic system demonstrates remarkable specificity, avoiding the chlorination side reactions common in hypochlorite-based methods, which ensures that the chlorine substituent on the aromatic ring remains intact while the aliphatic chain is transformed. The result is a clean reaction profile where the atom economy is maximized, and the need for extensive post-reaction neutralization steps is completely obviated.
Impurity control in this aerobic oxidation process is inherently superior due to the absence of halogenating agents that typically generate poly-chlorinated byproducts and tarry residues. The homogeneous nature of the reaction mixture ensures uniform heat and mass transfer, minimizing local hot spots that could lead to thermal decomposition or polymerization of the sensitive aromatic substrate. Furthermore, the ability to recover the acetic acid solvent under vacuum allows for the continuous removal of volatile impurities and water generated during the oxidation, driving the equilibrium towards the desired benzoic acid product. Post-reaction treatment involves a simple recrystallization from aqueous methanol, which effectively separates the target acid from any remaining catalyst traces or minor organic byproducts, yielding a product with purity levels consistently exceeding 96% and often reaching 98%. This high level of intrinsic purity reduces the burden on downstream coupling reactions, ensuring that the final herbicide active ingredients meet stringent regulatory specifications for residue and contaminant levels.
How to Synthesize 2-Chloro-3-Methyl-4-Methylsulfonylbenzoic Acid Efficiently
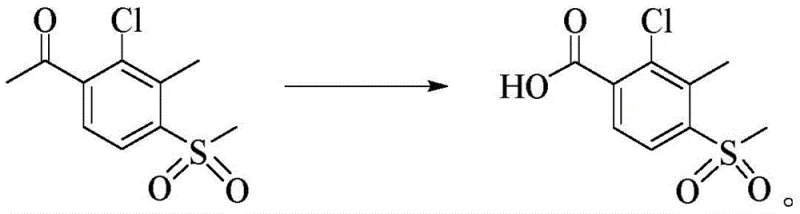
The optimized synthesis protocol outlined in the patent provides a clear roadmap for transitioning from batch to continuous processing, emphasizing the critical balance between catalyst loading, oxygen pressure, and temperature control. To achieve the reported yields of over 90%, operators must ensure that the oxygen supply is maintained dynamically to compensate for consumption, keeping the system pressure stable between 1.0 and 1.5 MPa throughout the 5 to 10-hour reaction window. The detailed standardized synthesis steps below outline the precise addition sequences and work-up procedures required to replicate the high-purity results demonstrated in the preferred embodiments, serving as a foundational guide for process engineers scaling this technology.
- Charge 2-chloro-3-methyl-4-methylsulfonyl acetophenone, acetic acid solvent, and manganese/cobalt acetate catalysts into an autoclave.
- Heat the mixture to 90-120°C and introduce oxygen to maintain a pressure of 1.0-1.5 MPa for 5-10 hours until pressure stabilizes.
- Recover the acetic acid solvent under vacuum, then recrystallize the crude product from aqueous methanol at 0°C to obtain high-purity acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aerobic oxidation technology translates directly into substantial operational expenditure reductions and enhanced supply security. By eliminating the need for sodium hypochlorite and the subsequent treatment of highly saline wastewater, manufacturers can bypass significant environmental compliance costs and avoid the logistical complexities associated with hazardous waste disposal. The simplified workflow, which removes multiple neutralization and extraction steps, shortens the overall production cycle time, allowing facilities to increase throughput without expanding physical infrastructure or capital investment in new reactor trains. Additionally, the use of readily available industrial gases like oxygen or air as reagents decouples production from the volatility of specialized chemical reagent markets, providing a more stable and predictable cost structure for long-term supply contracts.
- Cost Reduction in Manufacturing: The elimination of expensive halogenated oxidants and the drastic reduction in wastewater treatment requirements lead to significant savings in variable production costs. Since the acetic acid solvent is recoverable and recyclable, raw material consumption is minimized, and the avoidance of heavy metal catalysts or complex quenching agents further streamlines the bill of materials. This leaner manufacturing approach allows for competitive pricing strategies while maintaining healthy margins, making the supply of this key intermediate more resilient against market fluctuations.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or off-spec material that requires reprocessing. The reliance on commodity chemicals like oxygen and acetic acid, rather than specialized stoichiometric oxidants, mitigates supply chain risks associated with vendor shortages or transportation bottlenecks. This stability is crucial for meeting the rigid delivery schedules demanded by global agrochemical formulators who require uninterrupted feedstock for their own herbicide production lines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its homogeneous nature and manageable exotherm, facilitating a smooth transition from pilot plant to multi-ton commercial production without encountering the mixing or heat transfer limitations often seen in heterogeneous slurry reactions. Furthermore, the "green" credentials of the process, characterized by zero saline wastewater discharge and high atom economy, align perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against tightening emission standards and carbon taxes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced oxidation technology, drawing directly from the experimental data and comparative analysis provided in the patent documentation. These insights are designed to clarify the operational advantages and feasibility of adopting this route for large-scale intermediate production.
Q: How does the new aerobic oxidation method improve upon the traditional haloform reaction?
A: The traditional haloform reaction using sodium hypochlorite generates massive amounts of saline wastewater (approx. 8.3 tons per ton of product) and often causes raw material crystallization leading to incomplete reactions. The new aerobic oxidation method eliminates strong-acid salt wastewater entirely and maintains a homogeneous reaction phase for complete conversion.
Q: What catalysts are optimal for this oxidation process?
A: The patent specifies that manganese-containing compounds and/or cobalt-containing compounds, particularly manganese acetate and cobalt acetate, provide the highest efficiency. These catalysts enable high atom utilization and yields exceeding 90% under moderate temperatures.
Q: Is the solvent recoverable in this new process?
A: Yes, the carboxylic acid solvent, preferably acetic acid, can be recovered under vacuum after the reaction. This recyclability significantly reduces raw material consumption and waste generation compared to water-soluble solvents used in older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-Methyl-4-Methylsulfonylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the agrochemical value chain and possess the technical expertise to bring this patented aerobic oxidation process to commercial reality. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial volume is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-chloro-3-methyl-4-methylsulfonylbenzoic acid meets the exacting standards required for HPPD inhibitor synthesis.
We invite global partners to collaborate with us to leverage this greener, more cost-effective manufacturing route for their herbicide portfolios. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized supply chain can enhance your competitive position in the global agrochemical market.
