Advanced Hydrolysis Strategy for Acetophenone Acid: Scaling High-Purity Pharmaceutical Intermediates
The landscape of fine chemical synthesis is constantly evolving, driven by the dual imperatives of environmental sustainability and economic efficiency. A pivotal advancement in this domain is documented in patent CN112142587B, which discloses a novel preparation method for acetophenone acid and its derivatives. This compound serves as a critical building block in the manufacture of high-value agrochemicals, including the novel bactericide kresoxim-methyl and trifloxystrobin, as well as triazinone herbicides like metamitron. Furthermore, its utility extends into the pharmaceutical sector as a precursor for chiral mandelic acid and medical intermediates such as oxfenpyr-ammonium. The significance of this patent lies not merely in the molecule itself, but in the transformative process technology that enables its production. By shifting away from hazardous reagents and complex multi-step sequences, this innovation offers a streamlined pathway that addresses the longstanding pain points of toxicity and low atom economy associated with legacy manufacturing protocols.
For global supply chain leaders and R&D directors, the implications of adopting such a refined synthetic route are profound. The ability to produce high-purity intermediates with minimal environmental footprint is no longer just a regulatory requirement but a competitive advantage. This report analyzes the technical nuances of the inorganic acid-catalyzed hydrolysis method, demonstrating how it outperforms conventional strategies in terms of yield, operational simplicity, and scalability. As we delve into the mechanistic details and commercial advantages, it becomes evident that this technology represents a robust solution for the reliable supply of acetophenone acid derivatives in the modern chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acetophenone acid has been plagued by significant technical and environmental hurdles that hinder efficient large-scale production. Prior art methods, such as those described in US5221762, rely heavily on the use of thionyl chloride to convert o-methylbenzoic acid into acid chlorides, followed by reaction with sodium cyanide. This approach is inherently dangerous due to the involvement of cyanide, a highly toxic reagent that necessitates rigorous safety protocols and generates substantial hazardous waste streams. Similarly, other established routes utilizing benzoyl cyanide, as seen in US4596885, suffer from the high cost of raw materials and relatively low process yields, rendering them economically unviable for industrial applications. Furthermore, oxidative methods employing transition metal catalysts, like vanadium oxide, introduce the risk of heavy metal contamination in the final product, requiring expensive purification steps to meet stringent pharmaceutical grade specifications. These legacy processes collectively contribute to high operational expenditures and complex waste management challenges.
The Novel Approach
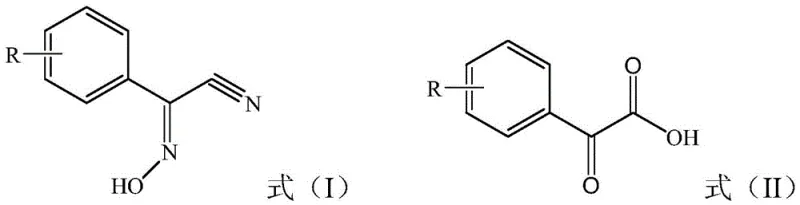
In stark contrast to these cumbersome traditional methods, the technology disclosed in CN112142587B introduces a direct and elegant hydrolysis strategy. The core innovation involves the catalytic hydrolysis of benzyl acetonitrile oxime, represented as Formula (I), using solely an inorganic acid to yield acetophenone acid, Formula (II). This single-step transformation eliminates the need for toxic cyanides, expensive acyl chlorides, or polluting transition metal catalysts. The process operates under relatively mild conditions compared to extreme high-pressure alternatives, utilizing common mineral acids like sulfuric acid or hydrochloric acid which are readily available and cost-effective. By simplifying the reaction sequence to a direct hydrolysis, the method drastically reduces the number of unit operations required, thereby minimizing potential points of failure and material loss. The result is a process that not only achieves exceptional yields, often exceeding 98%, but also delivers product purity levels upwards of 99%, setting a new benchmark for efficiency in fine chemical manufacturing.
Mechanistic Insights into Inorganic Acid-Catalyzed Hydrolysis
The success of this synthetic route hinges on the precise control of reaction parameters, particularly the concentration and stoichiometry of the inorganic acid catalyst. The mechanism proceeds through the protonation of the nitrile nitrogen and the oxime oxygen, facilitating nucleophilic attack by water molecules to cleave the carbon-nitrogen triple bond and convert the oxime functionality into a ketone and carboxylic acid group simultaneously. Critical to this process is the optimization of acid strength; experimental data reveals that while a 1:1 molar ratio theoretically suffices for hydrolysis, practical application requires a significant excess, such as 5 equivalents of sulfuric acid or 10 equivalents of hydrochloric acid, to drive the reaction to completion and suppress side reactions. The patent highlights that sulfuric acid generally outperforms hydrochloric acid in terms of final yield and purity, likely due to its non-volatile nature and higher boiling point which allows for sustained reaction temperatures without solvent loss. This careful balancing act ensures that the hydrolysis proceeds rapidly while minimizing the formation of polymeric byproducts or degradation species that could compromise the integrity of the final crystal lattice.
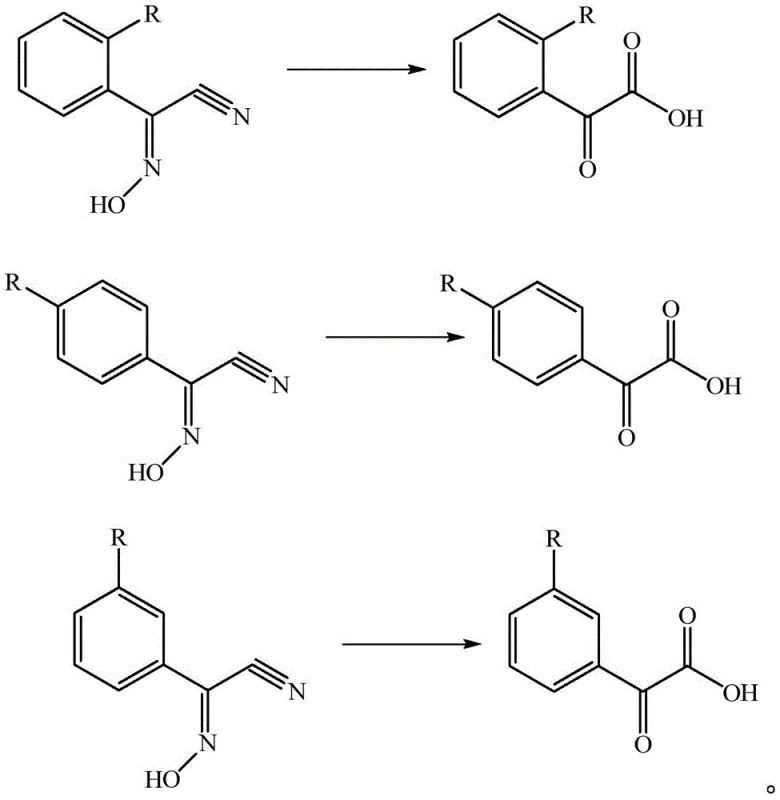
Furthermore, the versatility of this mechanism is demonstrated by its tolerance to various substituent groups on the aromatic ring, allowing for the synthesis of a broad spectrum of derivatives essential for diverse agricultural and pharmaceutical applications. Whether the substituent R is a hydrogen atom, a halogen like chlorine or fluorine, or an alkyl group such as methyl, ethyl, or tert-butyl, the hydrolysis conditions remain robust and effective. This structural flexibility is crucial for manufacturers who need to produce a portfolio of related intermediates without revalidating entirely new process chemistries for each analogue. The reaction accommodates ortho-, meta-, and para-substitutions with equal efficacy, as evidenced by the consistent high yields obtained across different isomers. This universality suggests a mechanism that is not sterically hindered by adjacent groups, making it a highly reliable platform technology for the production of complex substituted acetophenone acids required in advanced crop protection and drug discovery programs.
How to Synthesize Acetophenone Acid Efficiently
Implementing this synthesis route in a pilot or commercial plant requires strict adherence to the thermal and temporal parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the acid medium, where concentrated sulfuric acid is heated to an initial temperature range of 60 to 150°C, with a preferred window of 80 to 120°C to optimize reaction kinetics without inducing thermal degradation. The benzyl acetonitrile oxime starting material is then introduced gradually, typically over a one-hour period, to manage the exotherm and maintain homogeneous mixing. Following the addition, the reaction mixture is held at the target temperature for a duration ranging from 3 to 12 hours, depending on the specific substrate and acid concentration, until HPLC analysis confirms the complete consumption of the starting oxime. This controlled thermal profile is essential for maximizing the conversion rate while preventing the formation of tarry impurities that can occur under overly vigorous conditions.
- Prepare the reaction vessel by adding inorganic acid (preferably sulfuric acid with mass concentration 60-90%) and heating to an initial temperature of 60-150°C.
- Slowly add benzyl acetonitrile oxime to the heated acid over a period of 1 hour, then raise the temperature to 80-120°C and maintain for 3-12 hours to ensure complete hydrolysis.
- Upon completion, add an organic extractant such as dichloroethane or toluene, stir, cool the mixture in an ice bath to induce crystallization, and filter to obtain high-purity white crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this inorganic acid hydrolysis method offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of specialized and hazardous reagents such as sodium cyanide or thionyl chloride removes significant logistical burdens and regulatory compliance costs associated with the storage, handling, and disposal of toxic materials. This simplification of the raw material basket allows procurement teams to source inputs from a wider range of bulk chemical suppliers, thereby reducing dependency on single-source vendors and mitigating supply disruption risks. Moreover, the use of commodity inorganic acids like sulfuric acid, which are produced at massive global scales, ensures stable pricing and consistent availability, shielding the manufacturing process from the volatility often seen in the market for fine chemical reagents. These factors collectively contribute to a more predictable and secure supply chain for critical pharmaceutical and agrochemical intermediates.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the drastic simplification of the synthetic workflow and the removal of expensive catalytic systems. By avoiding the use of transition metal catalysts like vanadium oxide, manufacturers eliminate the costly downstream processing steps required to remove trace metal residues to meet ppm-level specifications. Additionally, the high atom economy of the direct hydrolysis reaction means that less raw material is wasted, leading to a significant reduction in the cost of goods sold per kilogram of finished product. The simplified workup procedure, which involves straightforward extraction and crystallization rather than complex distillation or chromatography, further lowers energy consumption and labor costs, resulting in substantial overall cost savings for large-scale production campaigns.
- Enhanced Supply Chain Reliability: The robustness of this chemistry translates directly into improved supply chain reliability and shorter lead times for customers. Because the reaction conditions are forgiving and the starting materials are stable and commercially abundant, the risk of batch failures due to reagent quality issues is minimized. This consistency allows for tighter production scheduling and more accurate delivery commitments, which is critical for just-in-time manufacturing environments in the pharmaceutical industry. Furthermore, the absence of highly toxic intermediates simplifies the transportation and warehousing requirements, enabling faster movement of materials through the logistics network without the delays associated with hazardous goods regulations. This agility ensures that downstream customers receive their essential intermediates without interruption, supporting continuous operation of their own formulation and synthesis lines.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is facilitated by its reliance on standard reactor equipment and common utilities, avoiding the need for specialized high-pressure or corrosion-resistant vessels often required for alternative routes. The environmental profile of the process is markedly superior, as it generates significantly less hazardous waste compared to cyanide-based methods, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. The primary waste stream consists of dilute acid which can be neutralized and treated using conventional wastewater infrastructure, reducing the burden on specialized waste disposal contractors. This eco-friendly characteristic not only lowers compliance costs but also enhances the brand reputation of the manufacturer as a responsible partner in the green chemistry initiative, appealing to environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrolysis technology, derived from the detailed experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages observed in side-by-side comparisons with prior art methods, highlighting the practical benefits of yield, purity, and operational safety that define this innovation.
Q: Why is the inorganic acid hydrolysis method superior to traditional cyanide-based routes?
A: Traditional methods often utilize toxic cyanides or expensive thionyl chloride, generating hazardous three-waste streams. The patented inorganic acid hydrolysis eliminates heavy metal catalysts and toxic cyanide reagents, significantly reducing environmental compliance costs and operational risks while achieving yields exceeding 98%.
Q: What represents the optimal acid concentration for maximizing yield and purity?
A: Experimental data indicates that sulfuric acid with a mass concentration between 60% and 90% provides the best balance. Lower concentrations lead to incomplete hydrolysis, while excessively high concentrations complicate waste acid treatment without proportional gains in product quality.
Q: Is this synthesis route applicable to substituted acetophenone acids?
A: Yes, the process demonstrates excellent substrate tolerance. It effectively synthesizes ortho-, meta-, and para-substituted derivatives, including methyl, ethyl, tert-butyl, and halogenated variants, maintaining high purity levels above 97% across diverse structural analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetophenone Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical and agrochemical supply chains. While the patent CN112142587B outlines a powerful methodology, translating such innovations into commercial reality requires deep process engineering expertise and state-of-the-art manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are fully realized in every batch we deliver. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every kilogram of acetophenone acid meets the exacting standards required for API and crop protection synthesis.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our optimized version of this hydrolysis technology, we can help you reduce total landed costs while securing a stable supply of high-quality intermediates. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Let us partner with you to engineer a more resilient and cost-effective supply chain for your critical chemical needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
