Advanced Green Synthesis of Halogenated Benzamides for Scalable Agrochemical Production
The pharmaceutical and agrochemical industries are constantly seeking sustainable pathways to synthesize critical intermediates, and the preparation method disclosed in patent CN108623491B represents a significant leap forward in the production of halogenated benzamide compounds. These compounds serve as pivotal precursors for benzoylurea insecticides, a class of chitin synthesis inhibitors renowned for their high activity, broad spectrum, and environmental safety profile. Traditional synthetic routes have long struggled with the balance between yield, purity, and environmental impact, often relying on hazardous reagents or energy-intensive conditions. This novel approach utilizes a green oxidation hydrolysis strategy, employing hydrogen peroxide in an aqueous medium to convert halogenated benzonitriles directly into the corresponding amides. By shifting away from corrosive acids and expensive transition metal catalysts, this technology offers a compelling value proposition for a reliable agrochemical intermediate supplier looking to optimize their supply chain while adhering to stricter global environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydrolysis of cyanophenyl derivatives into amides has been plagued by significant technical and environmental drawbacks that hinder efficient cost reduction in agrochemical manufacturing. The traditional sulfuric acid hydrolysis method, while capable of delivering high yields, generates massive volumes of acidic wastewater that require expensive neutralization and treatment processes, creating a heavy burden on facility infrastructure. Alternatively, metal-catalyzed methods utilizing ruthenium, gold, or copper complexes introduce the risk of heavy metal contamination in the final product, necessitating complex purification steps to meet stringent pharmaceutical and agrochemical purity specifications. Furthermore, conventional alkaline hydrolysis often demands extreme conditions, such as high temperature and high pressure, which not only increase energy consumption but also promote undesirable side reactions like dehalogenation or over-hydrolysis to carboxylic acids, thereby compromising the overall yield and selectivity of the desired halogenated benzamide product.
The Novel Approach
In stark contrast to these legacy methods, the innovative process described in the patent data leverages a mild, aqueous-based system that fundamentally alters the reaction landscape for producing high-purity halogenated benzamides. By employing hydrogen peroxide as the oxidant in the presence of a catalytic amount of base within a water solvent system, the reaction proceeds smoothly at moderate temperatures ranging from 30°C to 50°C. This eliminates the need for volatile organic solvents like DMSO, which are not only costly but also pose significant disposal challenges. The method achieves exceptional conversion rates with minimal byproduct formation, ensuring that the halogen substituents remain intact throughout the transformation. This operational simplicity translates directly into enhanced process safety and reduced capital expenditure on specialized high-pressure equipment, making it an ideal candidate for the commercial scale-up of complex agrochemical intermediates in modern manufacturing facilities.
Mechanistic Insights into Aqueous Hydrogen Peroxide Oxidation
The core of this technological breakthrough lies in the precise control of the oxidative hydrolysis mechanism, where hydrogen peroxide acts as a nucleophilic oxidant under mildly alkaline conditions. The reaction initiates with the activation of the nitrile group by the base, followed by the attack of the hydroperoxide anion, leading to the formation of a peroxyimidic acid intermediate which subsequently rearranges to the primary amide. Crucially, the patent specifies a tightly controlled molar ratio of hydrogen peroxide to the nitrile substrate, typically between 1:1 and 1.6:1, which is sufficient to drive the reaction to completion without the excessive oxidant load seen in prior art. This stoichiometric precision prevents the over-oxidation of the amide to the corresponding carboxylic acid, a common failure mode in less optimized systems. The use of water as the sole reaction medium further facilitates heat dissipation, allowing for the safe management of the exothermic nature of peroxide reactions while maintaining a homogeneous environment that promotes consistent reaction kinetics across large batch sizes.
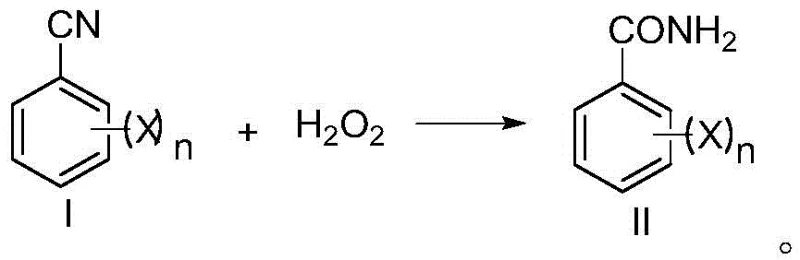
Impurity control is another critical aspect where this mechanism excels, particularly regarding the preservation of the halogen functionality which is essential for the biological activity of the downstream insecticide. In traditional high-temperature alkaline hydrolysis, the harsh conditions often trigger nucleophilic aromatic substitution where the halogen atom is displaced by a hydroxyl group, leading to difficult-to-separate phenolic impurities. However, by maintaining the reaction temperature strictly between 30°C and 50°C, preferably at 35°C, the kinetic energy of the system is kept below the threshold required for these deleterious side reactions. Additionally, the controlled addition time of hydrogen peroxide, spanning from 2 to 10 hours depending on the scale, ensures that the local concentration of the oxidant never spikes to levels that could induce radical-mediated degradation. This results in a crude product with purity exceeding 99.0% directly after filtration, significantly reducing the load on downstream purification units and ensuring a clean impurity profile for sensitive applications.
How to Synthesize Halogenated Benzamide Efficiently
Implementing this synthesis route requires careful attention to the addition rate of the oxidant and the maintenance of the alkaline environment to ensure reproducibility from laboratory to plant scale. The process begins by charging the reactor with the halogenated benzonitrile substrate, water, and a dilute aqueous solution of sodium hydroxide, establishing the necessary basic pH for activation. Once the mixture is stabilized at the target temperature, the hydrogen peroxide solution is introduced dropwise over several hours, a step that is critical for managing the reaction exotherm and maximizing selectivity. Detailed standardized operating procedures regarding the specific dosing profiles for gram, kilogram, and ton-scale batches are essential for maintaining the high yields reported in the patent examples. For a comprehensive guide on the exact parameters and safety protocols required for execution, please refer to the technical breakdown below.
- Prepare a mixed solution by combining the halogenated benzonitrile substrate (Formula I), water, and a catalytic amount of aqueous alkali (e.g., 30% NaOH) in a reaction vessel.
- Heat the mixture to a controlled temperature range of 30-50°C, preferably maintaining 35°C to ensure optimal reaction kinetics without thermal degradation.
- Slowly add hydrogen peroxide (27.5-30% aqueous solution) over a period of 2 to 10 hours, maintaining a molar ratio of 1: 1 to 1.6:1 relative to the nitrile substrate.
- Upon completion (monitored by HPLC/TLC until substrate <0.5%), filter the reaction mixture, wash the filter cake with water, and dry to obtain the high-purity amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this water-based oxidative hydrolysis technology offers transformative benefits that extend far beyond simple chemical conversion. The elimination of organic solvents removes a major variable from the raw material sourcing equation, insulating the production process from the volatility of petrochemical markets and reducing the logistical complexity associated with solvent recovery and recycling. Furthermore, the simplified workup procedure, which involves merely filtering and washing the solid product with water, drastically shortens the cycle time per batch and increases the throughput capacity of existing manufacturing assets without the need for major retrofitting. This operational efficiency directly contributes to substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing strategies in the global agrochemical market while maintaining healthy margins.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of expensive and hazardous reagents with commodity chemicals like water and hydrogen peroxide. By avoiding the use of precious metal catalysts such as ruthenium or gold, the method eliminates the capital tie-up associated with catalyst procurement and the operational costs of metal scavenging steps. Additionally, the reduction in hydrogen peroxide consumption to near-stoichiometric levels minimizes raw material waste, and the absence of organic solvents negates the need for energy-intensive distillation columns for solvent recovery. These factors combine to create a leaner cost structure that enhances the profitability of producing high-volume intermediates.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this chemistry ensures consistent delivery schedules and reduces the risk of production stoppages due to reagent shortages. The starting materials, including various halogenated benzonitriles and standard grade hydrogen peroxide, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The mild reaction conditions also reduce the wear and tear on reactor vessels and ancillary equipment, leading to lower maintenance downtime and higher asset availability. This reliability is crucial for meeting the just-in-time delivery requirements of downstream formulators who depend on a steady flow of key intermediates for their own production lines.
- Scalability and Environmental Compliance: The process has been rigorously validated across a wide range of scales, from small laboratory experiments to multi-ton industrial campaigns, demonstrating excellent linear scalability. The use of water as a solvent inherently aligns with green chemistry principles, significantly reducing the volume of hazardous waste generated per kilogram of product. This 'less three wastes' profile simplifies environmental permitting and compliance reporting, reducing the regulatory burden on the manufacturing site. As global regulations on industrial emissions tighten, adopting such an environmentally benign process future-proofs the supply chain against potential regulatory shocks and enhances the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogenated benzamide synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing production portfolios. We encourage stakeholders to review these points carefully to assess the alignment with their specific quality and operational requirements.
Q: Why is water preferred over organic solvents like DMSO for this hydrolysis?
A: Water eliminates the high cost and environmental burden associated with organic solvents like DMSO. It simplifies post-reaction workup to simple filtration and washing, drastically reducing three-waste discharge and improving the overall economic efficiency of the process.
Q: How does this method prevent dehalogenation side reactions?
A: By utilizing a mild alkaline environment combined with controlled hydrogen peroxide oxidation at moderate temperatures (30-50°C), the method avoids the harsh high-temperature and high-pressure conditions typical of traditional alkaline hydrolysis that often lead to unwanted dehalogenation.
Q: Is this process suitable for multi-ton industrial manufacturing?
A: Yes, the patent data explicitly demonstrates successful scale-up from gram levels to multi-ton batches (e.g., 1.25 tons), achieving consistent yields above 98% and purity exceeding 99.0%, confirming its robustness for commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Benzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of the global agrochemical industry, and we are uniquely positioned to support your needs through our advanced CDMO capabilities. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of halogenated benzamide meets or exceeds the >99.0% purity benchmarks established by the patent. Our commitment to quality assurance ensures that you receive a product that is consistent, reliable, and ready for immediate use in your downstream synthesis of benzoylurea insecticides.
We invite you to collaborate with us to leverage this green synthesis technology for your supply chain optimization initiatives. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We are prepared to provide specific COA data from our recent production runs and conduct detailed route feasibility assessments to demonstrate how this water-based process can enhance your operational efficiency. Contact us today to discuss how we can become your strategic partner in delivering high-performance agrochemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
