Revolutionizing Boscalid Intermediate Purification: A Technical Analysis of Selective Nitro Reduction
Revolutionizing Boscalid Intermediate Purification: A Technical Analysis of Selective Nitro Reduction
The global agrochemical industry constantly seeks more efficient pathways to produce high-volume herbicides like Boscalid, where the purity and cost of intermediates dictate market competitiveness. A significant breakthrough in this domain is documented in Chinese Patent CN110143881B, which details a sophisticated method for the selective reduction of nitro compounds found in synthetic mother liquors. This technology specifically addresses the challenging separation of o-chloronitrobenzene and 4'-chloro-2-nitro-1,1'-biphenyl, two structurally similar yet distinct molecules that often coexist in the crude mixtures of Boscalid synthesis. By leveraging the unique chemical properties of thiourea dioxide, this patent offers a robust solution that transforms a traditional waste stream into a recoverable resource, presenting a compelling value proposition for any reliable agrochemical intermediate supplier aiming to optimize their production lines.
For R&D directors and process engineers, the implications of this patent extend beyond simple purification; it represents a fundamental shift in how we approach the downstream processing of complex aromatic systems. The ability to chemically differentiate between a monophenyl nitro compound and a biphenyl nitro compound within the same reaction vessel opens new avenues for process intensification. Instead of relying on energy-intensive distillation or ineffective recrystallization techniques that often result in significant product loss, this method utilizes controlled redox chemistry to alter the polarity and solubility of specific components. This strategic manipulation allows for the isolation of high-purity 4'-chloro-2-nitro-1,1'-biphenyl, a critical precursor, while simultaneously converting the contaminant o-chloronitrobenzene into a separable amine derivative, thereby enhancing the overall atom economy of the manufacturing process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the traditional industrial production of Boscalid, the synthesis of the key intermediate 4'-chloro-2-nitro-1,1'-biphenyl typically generates a crystallization mother liquor that is rich in both the desired product and the starting material, o-chloronitrobenzene. Historically, separating these two components has been a formidable technical bottleneck because their physical properties, such as boiling points and solubility profiles in common organic solvents, are remarkably similar. Conventional purification strategies like fractional distillation or simple recrystallization often fail to achieve the necessary degree of separation, leading to a buildup of impurities that can poison downstream catalysts or degrade the quality of the final herbicide. Furthermore, the inability to effectively recover the unreacted o-chloronitrobenzene results in substantial economic losses, as valuable raw materials are discarded along with the waste liquor. From an environmental compliance perspective, this inefficiency creates a heavy burden, as the discharge of nitro-containing organic waste requires extensive and costly treatment protocols to meet regulatory standards.
The Novel Approach
The innovative methodology described in the patent circumvents these physical separation limitations by introducing a chemical differentiation step. By employing thiourea dioxide as a selective reducing agent, the process exploits the subtle differences in reactivity between the single-ring o-chloronitrobenzene and the double-ring 4'-chloro-2-nitro-1,1'-biphenyl. Under optimized alkaline conditions, the reducing agent preferentially targets the nitro group on the smaller, less sterically hindered monophenyl molecule, converting it rapidly into o-chloroaniline. In contrast, the nitro group on the bulky biphenyl structure reacts much more slowly, allowing the operator to halt the reaction at a precise point where the contaminant is fully reduced while the desired intermediate remains largely untouched. This selective transformation drastically changes the chemical nature of the mixture, enabling the easy separation of the resulting amine from the unreacted nitro-biphenyl through standard acid-base extraction or filtration techniques, thus achieving a level of purity and recovery that physical methods alone cannot match.
Mechanistic Insights into Thiourea Dioxide Selective Reduction
To fully appreciate the efficacy of this process, one must understand the underlying chemical mechanism of thiourea dioxide, also known as formamidine sulfinic acid. In its solid state, thiourea dioxide exists in a stable tautomeric form, but upon dissolution in an alkaline medium, it undergoes a critical rearrangement. As illustrated in the structural conversion below, the increase in pH and temperature facilitates the isomerization of thiourea dioxide into its active sulfinate form, which serves as the potent reducing species capable of transferring electrons to the nitro group. This activation step is paramount; without sufficient alkalinity, typically maintained at a pH greater than 11 using bases like sodium hydroxide, the reducing potential is insufficient to drive the reaction efficiently, and unwanted side products such as azo compounds may form. The control of this equilibrium is the first lever process engineers must pull to ensure the reaction proceeds with the desired kinetics and selectivity.
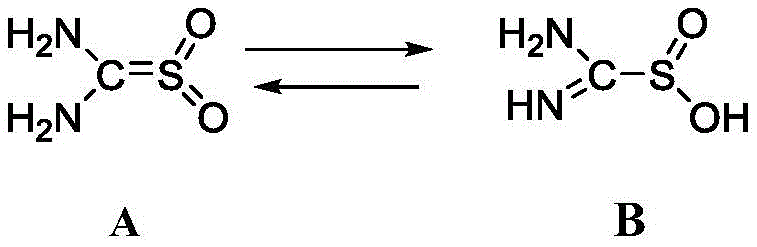
The second layer of mechanistic complexity involves the concept of chain-length selectivity, which is the cornerstone of this invention. The patent posits that the difference in the number of aromatic rings—specifically the steric bulk and electronic environment surrounding the nitro group—dictates the reaction rate. The monophenyl o-chloronitrobenzene presents a more accessible target for the nucleophilic attack or electron transfer initiated by the activated thiourea dioxide species. Conversely, the 4'-chloro-2-nitro-1,1'-biphenyl possesses a larger conjugated system and greater steric hindrance around the nitro functionality, which kinetically retards its reduction. This differential reactivity allows for a temporal window where the reduction of the impurity is nearly complete, while the conversion of the valuable biphenyl intermediate is minimized. The reaction scheme below clearly depicts the two competing pathways: Reaction One, the rapid reduction of o-chloronitrobenzene to o-chloroaniline, and Reaction Two, the slower, undesirable reduction of the biphenyl nitro compound. Mastering the balance between these two rates through precise control of temperature and stoichiometry is what enables the high selectivity reported in the examples.
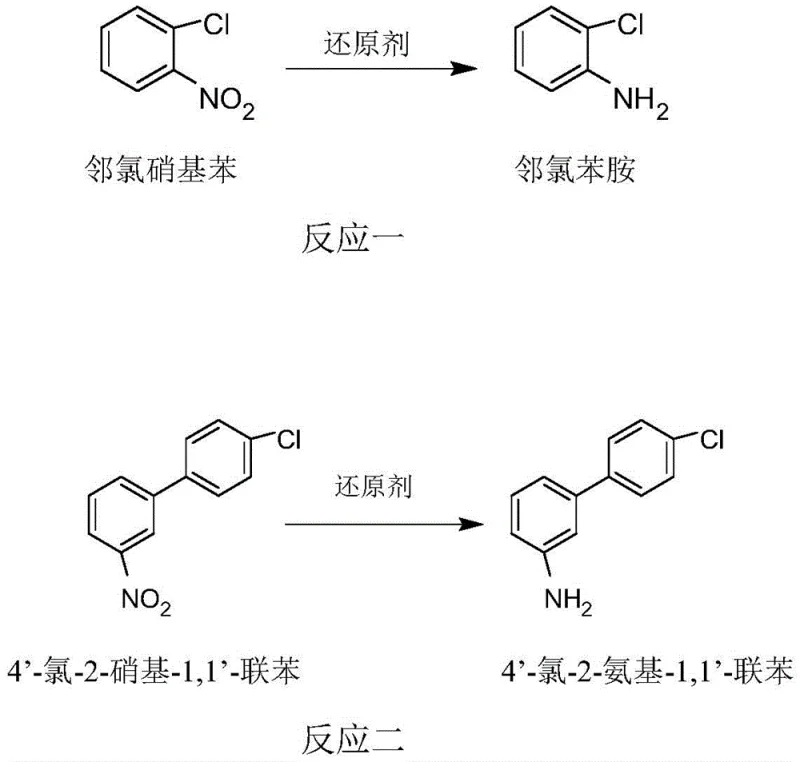
How to Synthesize High-Purity Boscalid Intermediate Efficiently
Implementing this selective reduction strategy requires a disciplined approach to reaction monitoring and parameter control to maximize the yield of the recovered intermediate. The process begins with the preparation of the mother liquor in a suitable solvent system, typically ethanol, which ensures good solubility for both the organic substrates and the inorganic salts generated during the reaction. The critical phase involves the gradual addition of the thiourea dioxide reducing agent in multiple portions rather than a single bolus. This portion-wise addition is essential for managing the exothermic nature of the reduction and maintaining the reaction temperature within the narrow optimal range of 10°C to 15°C. Deviating from this low-temperature window can accelerate the reduction of the biphenyl species, eroding the selectivity advantage. Detailed standardized synthesis steps for replicating this high-efficiency purification protocol are provided in the technical guide below.
- Dissolve the boscalid synthesis mother liquor in ethanol and adjust the pH to greater than 11 using a base like sodium hydroxide to activate the reducing agent.
- Add thiourea dioxide in multiple portions while maintaining the reaction temperature between 10°C and 15°C to ensure high selectivity for the monophenyl compound.
- Monitor the reaction progress via TLC or HPLC and stop the process once the o-chloronitrobenzene is reduced, allowing for the filtration and recovery of the unreacted biphenyl nitro compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this thiourea dioxide-based separation technology translates directly into tangible operational improvements and risk mitigation. The primary value driver is the dramatic enhancement in raw material utilization. By effectively recovering unreacted o-chloronitrobenzene and, more importantly, isolating high-purity 4'-chloro-2-nitro-1,1'-biphenyl from what was previously considered waste mother liquor, manufacturers can significantly reduce their net consumption of expensive starting materials. This circular approach to material flow not only lowers the direct cost of goods sold but also insulates the production line from volatility in the upstream supply of chloronitrobenzenes. Furthermore, the simplification of the purification train—replacing complex distillation columns or multi-stage recrystallizations with a single selective reduction and filtration step—reduces capital expenditure on equipment and lowers the energy intensity of the manufacturing process, contributing to a leaner and more agile supply chain.
- Cost Reduction in Manufacturing: The elimination of inefficient separation steps and the recovery of valuable intermediates lead to substantial cost savings without compromising product quality. By avoiding the loss of product in mother liquors, the overall yield of the synthesis pathway is improved, effectively lowering the unit cost of the final agrochemical active ingredient. Additionally, the use of thiourea dioxide, a stable and easily handled solid reagent, avoids the logistical complexities and safety costs associated with handling gaseous hydrogen or pyrophoric metal hydrides often used in traditional reductions.
- Enhanced Supply Chain Reliability: Implementing a robust recycling loop for key intermediates reduces dependency on external suppliers for make-up raw materials, thereby enhancing supply security. The ability to internally regenerate high-purity precursors from process streams ensures a more consistent feedstock quality for subsequent coupling reactions, minimizing batch-to-batch variability. This reliability is crucial for maintaining long-term contracts with major agrochemical companies who demand stringent consistency in their supply chains.
- Scalability and Environmental Compliance: The process operates under mild conditions (low temperature and atmospheric pressure) and uses common solvents like ethanol, making it inherently safer and easier to scale from pilot plant to commercial production volumes. From an environmental standpoint, converting nitro-compounds into amines facilitates easier wastewater treatment, and the reduction in organic waste volume helps facilities meet increasingly strict environmental regulations, avoiding potential fines and shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this selective reduction technology in an industrial setting. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this method for your specific production needs. Understanding these nuances is essential for process engineers tasked with integrating new purification technologies into existing infrastructure.
Q: Why is thiourea dioxide preferred for separating boscalid intermediates?
A: Thiourea dioxide offers unique selectivity based on chain length. Under controlled alkaline conditions, it rapidly reduces the shorter-chain o-chloronitrobenzene while leaving the longer-chain 4'-chloro-2-nitro-1,1'-biphenyl largely intact, enabling physical separation that recrystallization cannot achieve.
Q: What are the critical reaction parameters for maintaining selectivity?
A: The key parameters are maintaining a highly alkaline environment (pH > 11), controlling the temperature strictly between 10°C and 15°C, and optimizing the molar ratio of thiourea dioxide to the nitro compound (typically 1:3 to 1:10) to prevent over-reduction of the target biphenyl species.
Q: How does this process impact the environmental footprint of agrochemical production?
A: By enabling the recovery and recycling of valuable unreacted intermediates from mother liquor, this method significantly reduces chemical waste discharge. It transforms a disposal problem into a resource recovery opportunity, aligning with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boscalid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the path from patent to commercial reality requires more than just theoretical knowledge; it demands deep practical expertise in process engineering and scale-up. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative methods like the thiourea dioxide selective reduction are translated into robust, GMP-compliant manufacturing processes. Our rigorous QC labs and commitment to stringent purity specifications mean that we can deliver high-purity Boscalid intermediates that meet the exacting standards of the global agrochemical market, helping you secure your supply chain against disruptions.
We invite you to leverage our technical capabilities to optimize your current manufacturing routes. Whether you are looking to implement this specific recycling technology or explore other avenues for cost reduction in agrochemical manufacturing, our team is ready to assist. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis. By sharing your specific process challenges, we can provide detailed route feasibility assessments and specific COA data to demonstrate how our solutions can enhance your operational efficiency and profitability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
