Revolutionizing 2-Methoxy-6-Methylbenzoic Acid Production: A Catalytic Breakthrough for Industrial Scale-Up
The global demand for high-performance agrochemicals continues to drive the need for efficient, cost-effective synthesis routes for key intermediates such as 2-methoxy-6-methylbenzoic acid, a critical building block for the fungicide metrafenone. Recent advancements detailed in patent CN114105755A introduce a transformative preparation method that addresses long-standing inefficiencies in traditional manufacturing protocols. This novel approach leverages a catalytic copper system combined with inexpensive oxidants to achieve high conversion rates while significantly reducing environmental impact and operational costs. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for optimizing supply chains and ensuring the consistent availability of high-purity agrochemical intermediates. The shift from stoichiometric heavy metal usage to a catalytic regime represents a paradigm shift in fine chemical manufacturing, offering a sustainable pathway that aligns with modern green chemistry principles without compromising on yield or product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methoxy-6-methylbenzoic acid has relied on multi-step processes that are both economically burdensome and environmentally taxing. One prominent traditional route involves the methylation of 2-hydroxy-6-methylbenzaldehyde followed by oxidation using silver oxide, a reagent that is prohibitively expensive for large-scale industrial application. As illustrated in the reaction scheme below, this legacy method necessitates harsh conditions and generates significant metallic waste, creating substantial disposal challenges for manufacturing facilities.
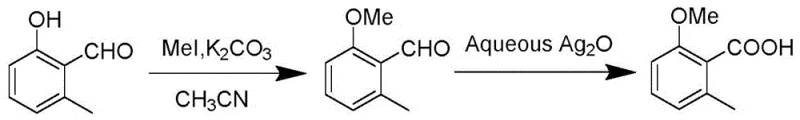
Furthermore, alternative prior art methods utilizing stoichiometric amounts of copper salts coupled with oxidants like tert-butyl hydroperoxide or sodium chlorite suffer from similar drawbacks regarding reagent cost and atom economy. These conventional pathways often require rigorous purification steps to remove residual heavy metals, adding complexity to the downstream processing and increasing the overall lead time for production. The reliance on stoichiometric metal loads not only inflates the raw material costs but also complicates the regulatory compliance landscape due to strict limits on heavy metal residues in final agrochemical products. Consequently, there has been a persistent industry need for a more streamlined, catalytic approach that can deliver the same high-quality intermediate with a reduced ecological footprint and lower operational expenditure.
The Novel Approach
The methodology disclosed in the referenced patent offers a compelling solution by employing a catalytic amount of copper salt in conjunction with a specific ligand system and potassium persulfate as the primary oxidant. This innovative strategy allows for the direct oxidation of 2,3-dimethyl anisole under mild thermal conditions, effectively bypassing the need for expensive noble metals or hazardous peroxide oxidants. The process is designed to be telescoped, where the initial oxidation is followed by a secondary oxidation step using sodium hypochlorite in the presence of sulfamic acid, ensuring complete conversion to the carboxylic acid functionality. By drastically reducing the copper loading to catalytic levels, the new method minimizes metal waste and simplifies the purification workflow, thereby enhancing the overall efficiency of the manufacturing process. This approach not only lowers the direct material costs but also improves the safety profile of the operation by avoiding the handling of large quantities of unstable oxidizing agents.
Mechanistic Insights into Copper-Catalyzed Oxidative Functionalization
The core of this technological advancement lies in the synergistic interaction between the copper catalyst, the nitrogen-based ligand, and the persulfate oxidant, which facilitates the selective activation of the benzylic C-H bond. The reaction mechanism likely proceeds through a radical pathway where the copper species mediates the generation of sulfate radicals from potassium persulfate, which subsequently abstract a hydrogen atom from the methyl group of the substrate. The presence of the ligand is crucial as it stabilizes the copper center, preventing premature precipitation or deactivation, and enhances the regioselectivity of the oxidation to favor the formation of the aldehyde intermediate which is further oxidized in situ. This controlled radical generation ensures that over-oxidation or non-selective degradation of the aromatic ring is minimized, leading to a cleaner reaction profile with fewer side products. Understanding this mechanistic nuance is vital for process chemists aiming to replicate these results on a pilot or commercial scale, as slight variations in ligand structure or oxidant addition rates can significantly influence the reaction kinetics and final yield.
Following the initial benzylic oxidation, the introduction of sodium hypochlorite and sulfamic acid triggers a Pinnick-type oxidation or a similar hypochlorite-mediated conversion of the intermediate aldehyde to the corresponding carboxylic acid. This second stage is critical for achieving the final product specification, and the use of sulfamic acid serves to buffer the reaction medium and quench excess oxidant, thereby preventing chlorination of the aromatic ring which is a common side reaction in hypochlorite oxidations. The careful control of pH and temperature during this phase ensures that the delicate methoxy group remains intact while the aldehyde is efficiently converted to the acid. This two-stage oxidative sequence within a single pot exemplifies a sophisticated understanding of reaction engineering, allowing for the transformation of a simple hydrocarbon feedstock into a valuable functionalized intermediate with high atom economy. The result is a robust process capable of delivering product purities exceeding 96%, as demonstrated in the experimental examples, making it highly suitable for downstream coupling reactions in the synthesis of complex agrochemical active ingredients.
How to Synthesize 2-Methoxy-6-Methylbenzoic Acid Efficiently
Implementing this novel synthesis route requires precise adherence to the reaction parameters outlined in the patent to maximize yield and minimize impurity formation. The process begins with the charging of 2,3-dimethyl anisole, a catalytic quantity of copper salt such as copper sulfate pentahydrate, a nitrogen-containing ligand like phenanthroline or bipyridine, and potassium persulfate into a suitable reaction vessel containing a mixed solvent system of water and an organic co-solvent. The detailed standardized synthetic steps for this procedure are provided in the guide below.
- Charge 2,3-dimethyl anisole, catalytic copper salt, ligand, and potassium persulfate into a reactor with solvent and heat to 50-100°C.
- Cool the reaction mixture to room temperature and add sulfamic acid followed by the dropwise addition of aqueous sodium hypochlorite.
- Perform standard workup including filtration, extraction, pH adjustment, and recrystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic methodology presents a strategic opportunity to optimize the cost structure and reliability of the supply chain for agrochemical intermediates. By shifting away from stoichiometric metal reagents and expensive oxidants, manufacturers can achieve substantial cost savings in raw material procurement, which directly translates to improved margins and competitive pricing for the final active ingredient. The use of commodity chemicals like potassium persulfate and sodium hypochlorite, which are widely available and inexpensive, mitigates the risk of supply disruptions associated with specialized or imported reagents. Furthermore, the simplified workup procedure reduces the consumption of solvents and auxiliary materials during purification, contributing to a leaner and more sustainable manufacturing operation that aligns with corporate sustainability goals.
- Cost Reduction in Manufacturing: The transition to a catalytic copper system eliminates the need for costly stoichiometric amounts of metal salts and expensive oxidants like silver oxide, leading to a drastic reduction in direct material costs. Additionally, the simplified purification process reduces the consumption of solvents and energy required for extensive washing and metal scavenging steps. This cumulative effect results in a significantly lower cost of goods sold (COGS), allowing for more flexible pricing strategies in a competitive market. The economic benefit is further amplified by the high yield and purity achieved, which minimizes waste and maximizes the output per batch.
- Enhanced Supply Chain Reliability: The reliance on readily available, bulk commodity chemicals such as sodium hypochlorite and potassium persulfate ensures a stable and resilient supply chain that is less susceptible to geopolitical or logistical disruptions. Unlike specialized reagents that may have long lead times or single-source dependencies, the key inputs for this process are produced globally in large volumes. This accessibility allows for better inventory management and reduces the risk of production stoppages due to raw material shortages. Consequently, suppliers can offer more reliable delivery schedules and maintain consistent stock levels to meet the fluctuating demands of the agrochemical sector.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous-compatible solvent system make this process highly scalable from laboratory to multi-ton commercial production without significant engineering hurdles. The reduction in heavy metal waste and the use of less hazardous oxidants simplify wastewater treatment and waste disposal, ensuring compliance with increasingly stringent environmental regulations. This environmental friendliness not only reduces the liability associated with waste management but also enhances the brand reputation of the manufacturer as a responsible producer. The robustness of the process supports continuous improvement initiatives and facilitates the rapid scale-up required to meet growing market demand for metrafenone and related derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method, providing clarity for stakeholders evaluating its potential for integration into their existing manufacturing portfolios. These insights are derived directly from the technical specifications and experimental data presented in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of technology transfer and the potential return on investment for adopting this greener, more efficient pathway.
Q: What are the primary cost advantages of this new synthesis method compared to traditional routes?
A: The primary advantage lies in the transition from stoichiometric to catalytic copper usage and the replacement of expensive oxidants like silver oxide or tert-butyl hydroperoxide with cost-effective potassium persulfate and sodium hypochlorite.
Q: How does this process ensure high purity for agrochemical applications?
A: The process utilizes a specific ligand system that enhances regioselectivity during oxidation, minimizing byproduct formation, followed by a robust purification protocol involving pH-controlled extraction and recrystallization.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the method operates under mild temperatures (50-100°C), uses readily available industrial reagents, and features a simple workup procedure, making it highly adaptable for multi-ton production scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methoxy-6-Methylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the broader context of agrochemical development and production. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the catalytic oxidation method described in CN114105755A can be seamlessly translated into robust industrial processes. We are committed to delivering high-purity 2-methoxy-6-methylbenzoic acid that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality and consistency makes us a trusted partner for multinational agrochemical companies seeking to secure their supply chains with reliable, cost-effective intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this route offers compared to your current supply sources. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your long-term production goals with superior quality and service.
