Scaling High-Purity Oxo-Ionone Production via Green Copper-Catalyzed Oxidation
Introduction to Advanced Oxo-Ionone Manufacturing
The global demand for high-purity fragrance and pharmaceutical intermediates continues to drive innovation in synthetic methodology, particularly for complex terpenoid derivatives. Patent CN101624336A introduces a transformative approach to the preparation of oxo-α,β-ionone, a critical structural motif found in valuable aroma compounds and bioactive carotenoid precursors. This technology leverages a copper-catalyzed oxidation system that replaces hazardous stoichiometric oxidants with a catalytic cycle driven by alkyl hydrogen peroxide. The significance of this development lies in its ability to produce key isomers, specifically 4-oxo-beta-ionone and 3-oxo-alpha-ionone, through a streamlined one-step process that operates under mild thermal conditions. By shifting away from traditional heavy metal oxidants, this method addresses both the economic and environmental pressures facing modern fine chemical manufacturing.
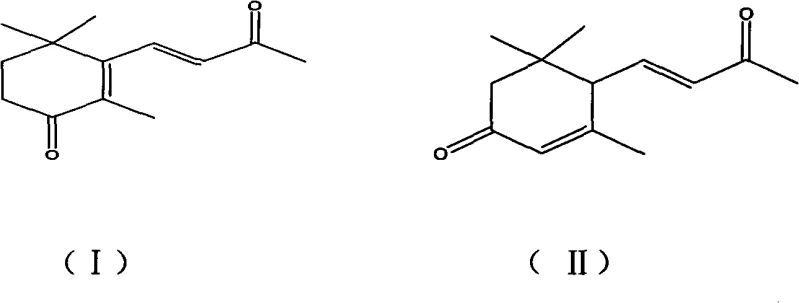
The versatility of this synthetic route is evident in its applicability across multiple high-value sectors, including tobacco flavorings, food additives, and the synthesis of complex heterocyclic compounds for medicinal chemistry. The patent details a robust protocol where α,β-ionone serves as the starting material, undergoing selective oxidation at the allylic position. This transformation is crucial because the resulting ketone functionality serves as a versatile handle for further derivatization, such as the synthesis of Megastigmatrienone or various glucoside derivatives used in the nutraceutical industry. For procurement specialists and R&D directors, understanding the mechanistic underpinnings and operational simplicity of this patent is essential for evaluating its potential to reduce costs and improve supply chain reliability for these high-demand intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of oxo-ionones has relied heavily on stoichiometric oxidation reagents that pose significant safety and environmental liabilities. Traditional protocols frequently employ chromium-based oxidants such as chromium trioxide, chromic acid tert-butyl ester, or pyridinium chlorochromate (PCC). While chemically effective, these reagents generate substantial quantities of toxic hexavalent chromium waste, which requires expensive and rigorous disposal procedures to meet increasingly stringent environmental regulations. Furthermore, the presence of residual heavy metals in the final product is a critical quality attribute failure for applications in food, cosmetics, and pharmaceuticals, necessitating complex and yield-reducing purification steps to ensure compliance with safety standards. Additionally, some older methods utilizing sodium chlorate or molecular oxygen often suffer from unsatisfactory yields or require tediously long reaction times and complex multi-step build-up processes that hinder efficient mass production.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101624336A utilizes a catalytic system comprising a metal copper compound and a hydrocarbyl hydrogen peroxide, preferably tert-butyl hydroperoxide (TBHP). This novel approach fundamentally changes the economic and operational landscape of oxo-ionone production by enabling a direct, one-step synthesis that proceeds efficiently at temperatures below 80°C. The use of copper salts, such as copper(II) chloride or copper(II) acetate, allows for the activation of the peroxide oxidant in a catalytic cycle, drastically reducing the amount of metal waste generated compared to stoichiometric chromium methods. This shift not only simplifies the reaction workup—often requiring only filtration and solvent removal—but also inherently improves the environmental profile of the manufacturing process. The result is a cleaner reaction mixture with fewer by-products, facilitating easier isolation of the target oxo-ionone isomers with high purity levels exceeding 98% without the need for exhaustive metal scavenging treatments.
Mechanistic Insights into Copper-Catalyzed Allylic Oxidation
The core of this technological advancement lies in the redox cycling of the copper catalyst, which facilitates the homolytic cleavage of the peroxide bond in tert-butyl hydroperoxide. In this mechanistic pathway, the copper species, oscillating between Cu(I) and Cu(II) oxidation states, generates tert-butoxyl radicals that selectively abstract an allylic hydrogen atom from the ionone substrate. This radical abstraction creates a resonance-stabilized allylic radical intermediate, which subsequently reacts with oxygen species derived from the peroxide to form the desired ketone functionality. The elegance of this mechanism is its selectivity; by carefully controlling the reaction temperature (optimized between 10°C and 60°C) and the specific copper ligand environment provided by the solvent, the process can be tuned to favor oxidation at specific positions on the ionone ring or side chain. This level of control is paramount for distinguishing between the formation of 4-oxo-beta-ionone and 3-oxo-alpha-ionone, ensuring that the production output aligns with the specific isomeric requirements of the downstream application.
From an impurity control perspective, this catalytic system offers distinct advantages over non-selective oxidants. Traditional strong oxidants often lead to over-oxidation, epoxide formation, or cleavage of the sensitive conjugated double bond system inherent in ionones. However, the copper-catalyzed radical mechanism described in the patent demonstrates a high degree of chemoselectivity, preserving the integrity of the enone system while introducing the ketone group. The patent data indicates that byproducts such as epoxy-ionones are formed in manageable quantities and can be effectively separated during the standard aqueous workup involving sodium sulfite washing. This inherent selectivity reduces the burden on downstream purification units, such as distillation columns or chromatography systems, thereby increasing the overall throughput and yield of the manufacturing campaign. For R&D teams, this implies a more predictable impurity profile, which simplifies the validation process for regulatory submissions in the pharmaceutical and food sectors.
How to Synthesize Oxo-Alpha-Beta-Ionone Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it highly attractive for scale-up operations. The general procedure involves dissolving the starting α,β-ionone and the chosen copper catalyst in a polar organic solvent such as acetonitrile or ethanol. Once the mixture is homogenized under magnetic stirring, the oxidant (TBHP) is added dropwise while strictly maintaining the reaction temperature within the specified range to prevent thermal runaway or non-selective decomposition. Following the reaction period, typically lasting between 2 to 10 hours depending on the specific isomer target, the catalyst is removed via simple filtration. The subsequent workup involves solvent evaporation, liquid-liquid extraction to remove organic impurities, and washing with reducing agents like sodium sulfite to quench any residual peroxides. For a detailed, step-by-step breakdown of the exact molar ratios, specific solvent choices for different isomers, and purification parameters, please refer to the standardized synthesis guide below.
- Dissolve alpha/beta-ionone and a divalent or monovalent copper compound (e.g., CuCl2) in a polar organic solvent like acetonitrile.
- Slowly add alkyl hydrogen peroxide (preferably TBHP) while maintaining the reaction temperature between 10°C and 80°C under magnetic stirring.
- After reaction completion, filter the catalyst, remove solvents via rotary evaporation, and purify the crude product through extraction and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed oxidation technology represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with regulated reagents. The elimination of chromium-based oxidants removes a significant variable cost related to hazardous waste disposal and environmental compliance fees, which have been rising globally. Furthermore, the reliance on commodity chemicals like copper salts and tert-butyl hydroperoxide ensures a stable and diversified supply base, reducing the risk of bottlenecks that often occur with specialized or highly regulated oxidizing agents. The simplified one-step nature of the reaction also translates to reduced processing time and lower energy consumption, as the reaction proceeds efficiently at moderate temperatures without the need for extended heating or cryogenic cooling. These factors collectively contribute to a more resilient and cost-effective supply chain for high-value fragrance and pharma intermediates.
- Cost Reduction in Manufacturing: The transition from stoichiometric chromium reagents to a catalytic copper system fundamentally alters the cost equation by eliminating the need for expensive heavy metal waste treatment and complex purification protocols. Since the copper catalyst is used in relatively small amounts and the byproduct of the oxidant (tert-butanol) is volatile and easily removed, the downstream processing costs are significantly lowered. This efficiency gain allows manufacturers to offer competitive pricing for high-purity oxo-ionones while maintaining healthy margins, providing a clear economic advantage over legacy production methods that suffer from low atom economy and high waste generation.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including various copper salts and organic peroxides, are widely produced commodities with robust global supply networks. This contrasts sharply with specialized chromium reagents, which may face supply constraints due to tightening environmental regulations on their manufacture and transport. By utilizing a chemistry based on readily available inputs, suppliers can guarantee consistent production schedules and shorter lead times, ensuring that downstream customers in the flavor and pharmaceutical industries receive their materials without interruption. The robustness of the reaction conditions further supports this reliability, as the process is less sensitive to minor fluctuations in operating parameters compared to more finicky biological or enzymatic oxidation methods.
- Scalability and Environmental Compliance: The mild reaction conditions (below 80°C) and the absence of highly toxic reagents make this process exceptionally well-suited for large-scale commercial production. Scaling from pilot plant to multi-ton reactors does not require exotic materials of construction or specialized safety infrastructure beyond standard peroxide handling protocols. Moreover, the environmental footprint of the process is drastically reduced, aligning with the sustainability goals of major multinational corporations. The ease of waste management and the potential for solvent recycling further enhance the green credentials of this manufacturing route, facilitating smoother regulatory approvals and fostering long-term partnerships with environmentally conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of oxo-ionones using the patented copper-catalyzed method. These insights are derived directly from the experimental data and technical specifications outlined in patent CN101624336A, providing clarity on the feasibility and advantages of this synthetic route. Understanding these details is crucial for stakeholders evaluating the integration of this technology into their existing supply chains or R&D pipelines.
Q: Why is the copper-catalyzed method preferred over traditional chromium oxidation?
A: Traditional methods using chromium trioxide or pyridinium chlorochromate generate toxic heavy metal waste that is difficult and costly to dispose of, especially for food and pharmaceutical grades. The copper-catalyzed route described in CN101624336A eliminates hexavalent chromium, significantly simplifying downstream purification and environmental compliance.
Q: Can this process selectively produce specific isomers like 3-oxo-alpha-ionone?
A: Yes, the patent demonstrates that by adjusting reaction parameters such as the specific copper salt used and reaction duration, the process can favor the formation of either 4-oxo-beta-ionone or 3-oxo-alpha-ionone, allowing manufacturers to tailor production to specific market demands.
Q: What are the scalability advantages of this oxidation protocol?
A: The reaction operates at mild temperatures (below 80°C) and uses commercially available reagents like tert-butyl hydroperoxide. This avoids the need for extreme cryogenic conditions or high-pressure equipment, making the transition from laboratory bench scale to multi-ton commercial production straightforward and safe.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxo-Alpha-Beta-Ionone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products, whether they are premium fragrances or life-saving pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering oxo-alpha-beta-ionone and related derivatives with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the highest international standards for residual metals and organic impurities. Our facility is equipped to handle the specific requirements of copper-catalyzed processes, ensuring safe and efficient manufacturing that aligns with the green chemistry principles highlighted in recent patent literature.
We invite you to collaborate with us to leverage these advanced synthetic capabilities for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener oxidation route for your supply chain. We encourage you to reach out today to obtain specific COA data for our current inventory and to discuss route feasibility assessments tailored to your volume requirements. Let us be your partner in driving efficiency and quality in the production of complex fine chemical intermediates.
