Advanced One-Step Synthesis of 4-Oxo-Beta-Damascenone for Industrial Flavor Applications
Introduction to High-Efficiency Aroma Compound Manufacturing
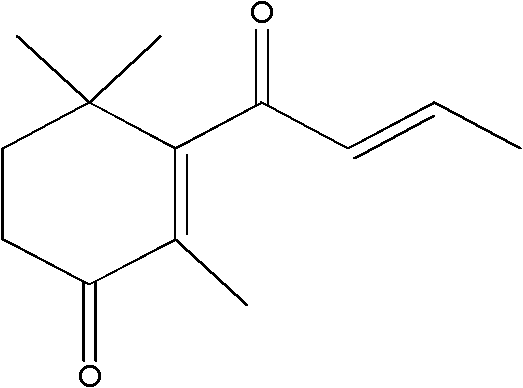
The disclosed technology in patent CN102329203A represents a paradigm shift in the manufacturing of high-value aroma compounds, specifically targeting the efficient production of 4-oxo-beta-damascenone. This molecule, a critical oxidative breakdown product of carotenoids, serves as a cornerstone in the formulation of premium tobacco aromatics and fine fragrances due to its unique fruity and smooth olfactory profile. The innovation lies in a streamlined one-step synthetic protocol that utilizes sodium bromate and sulfuric acid within a hydrophilic organic solvent system, effectively bypassing the complex multi-stage sequences traditionally associated with ionone derivatives. By maintaining reaction temperatures below 80°C and strictly controlling the pH between 1 and 3, the process achieves a remarkable balance between reaction kinetics and product stability, ensuring high purity levels suitable for sensitive applications in food additives and cosmetics. This technical breakthrough addresses the longstanding industry demand for reliable flavor & fragrance intermediate suppliers who can deliver consistent quality without the volatility of extraction-based supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of damascenone derivatives has been plagued by inefficient methodologies that struggle to balance yield with selectivity. Traditional approaches often rely on the thermal degradation of carotenoids at temperatures around 700°C, a violent process that generates a chaotic mixture of more than 50 different split products, including various trimethyl-cyclohexenones and ketones. This lack of specificity necessitates arduous downstream purification steps to isolate the desired aroma active, drastically inflating production costs and reducing overall material throughput. Furthermore, alternative synthetic routes involving multi-step functional group transformations often require expensive transition metal catalysts or hazardous reagents that pose significant environmental and safety liabilities. The cumulative effect of these inefficiencies is a supply chain prone to bottlenecks, where the cost reduction in fine chemical manufacturing is stifled by low atom economy and excessive waste generation.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data introduces a direct oxidative transformation that converts beta-damascenone (dorinone) into the target 4-oxo derivative in a single operational step. By leveraging the tunable oxidation potential of sodium bromate in an acidic medium, the reaction proceeds smoothly at mild temperatures between 45°C and 60°C, eliminating the need for extreme thermal conditions. This method not only simplifies the operational workflow but also enhances the selectivity of the oxidation, achieving a chromatographic peak area ratio of up to 93.5% for the desired product in optimized embodiments. The use of common hydrophilic solvents such as THF, acetone, or ethanol further democratizes the process, allowing for easy solvent recovery and recycling. Consequently, this route offers a robust solution for the commercial scale-up of complex flavor intermediates, providing a clear pathway to higher yields and reduced environmental impact compared to legacy technologies.
Mechanistic Insights into Acidic Bromate Oxidation
The core of this synthetic strategy relies on the precise modulation of electrophilic oxidation power through acid catalysis. In this system, sodium bromate serves as the primary oxidant, but its reactivity is intrinsically linked to the proton concentration of the reaction medium. Under the specified acidic conditions where the pH is maintained between 1 and 3, the bromate ion is activated to a species capable of selectively attacking the electron-rich positions on the beta-damascenone scaffold. This controlled activation prevents the runaway oxidation that typically leads to ring cleavage or the formation of carboxylic acid byproducts. The mechanism likely involves the formation of an enol or enolate intermediate which is subsequently oxidized to introduce the ketone functionality at the 4-position of the cyclohexenone ring. Understanding this pH-dependency is crucial for R&D directors aiming to replicate the high purity specifications, as deviations in acidity can lead to incomplete conversion or the generation of polymeric tars.
Furthermore, the choice of solvent plays a pivotal role in stabilizing the transition states and managing the solubility of both the organic substrate and the inorganic oxidant. The patent specifies the use of hydrophilic organic solvents which create a quasi-homogeneous environment, facilitating better mass transfer between the aqueous acid phase and the organic substrate. This miscibility ensures that the oxidation occurs uniformly throughout the reaction vessel, minimizing local hotspots of high oxidant concentration that could degrade the sensitive conjugated enone system. The result is a clean reaction profile where the primary impurity is simply the unreacted starting material or minor over-oxidation products, both of which are easily removed during the subsequent recrystallization step. This mechanistic clarity provides a solid foundation for process optimization, allowing manufacturers to fine-tune parameters for maximum efficiency.
How to Synthesize 4-Oxo-Beta-Damascenone Efficiently
Implementing this synthesis requires careful attention to the order of addition and thermal management to ensure safety and reproducibility. The process begins with the dissolution of the beta-damascenone starting material in a selected hydrophilic solvent, followed by the gradual introduction of the acid solution to establish the necessary acidic environment before the oxidant is added. This sequence is vital to prevent the premature decomposition of the bromate or the formation of explosive bromine species. Once the pH is stabilized, the sodium bromate is introduced slowly to manage the exotherm, keeping the temperature strictly within the 45°C to 60°C window for a duration of 2 to 5 hours. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below.
- Prepare the reaction mixture by dissolving beta-damascenone in a hydrophilic organic solvent such as THF or acetone, then add dilute sulfuric acid to establish a pH between 1 and 3.
- Slowly introduce sodium bromate to the stirred mixture while maintaining the temperature between 45°C and 60°C to control the oxidation rate and prevent side reactions.
- After reacting for 2 to 5 hours, neutralize the mixture, separate the organic phase, wash to remove inorganic salts, and purify the crude product via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-step oxidative method presents compelling economic and logistical benefits that directly impact the bottom line. The elimination of multi-step sequences and high-temperature thermal cracking units significantly lowers the capital expenditure required for production facilities, while the use of commodity chemicals like sulfuric acid and sodium bromate ensures a stable and cost-effective raw material supply. The drastic reduction in reaction time from over 10 hours in conventional methods to merely 2-5 hours in this process translates to higher asset utilization rates, allowing manufacturers to respond more agilely to market fluctuations in the tobacco and flavor industries. Moreover, the simplified workup procedure, which involves basic phase separation and recrystallization rather than complex chromatography, reduces labor costs and solvent consumption, contributing to substantial cost savings in overall manufacturing operations.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of expensive catalysts and energy-intensive thermal steps with inexpensive inorganic oxidants and mild heating requirements. By avoiding the need for precious metal catalysts, the process eliminates the costly and technically challenging step of heavy metal removal, which is often a regulatory hurdle in pharmaceutical and food-grade applications. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting material to side products, thereby improving the effective yield and reducing the cost per kilogram of the final active ingredient. These factors combine to create a leaner production model that maximizes margin potential for suppliers of high-purity flavor intermediates.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on widely available bulk chemicals mitigates the risk of supply disruptions that often plague specialty reagent markets. The robustness of the reaction conditions means that production can be scaled up or down with minimal requalification, ensuring a continuous flow of goods to downstream formulators. The shorter cycle time also means that inventory turnover is accelerated, reducing the working capital tied up in work-in-progress materials. This reliability is essential for maintaining the continuity of supply for major FMCG clients who depend on consistent batches of aroma compounds for their global product lines.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to traditional methods, as the primary byproduct is sodium bromide, a benign salt that can be easily managed or utilized in other industrial processes. The absence of toxic heavy metals and the use of recyclable hydrophilic solvents align with increasingly stringent global environmental regulations, reducing the burden of waste treatment and disposal. This 'green' credential not only future-proofs the manufacturing site against regulatory changes but also enhances the brand value for customers seeking sustainable sourcing options. The simplicity of the scale-up process ensures that moving from pilot plant to commercial production involves minimal technical risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-oxo-beta-damascenone, based on the specific capabilities demonstrated in the patent literature. These insights are designed to clarify the feasibility of adopting this technology for large-scale industrial use and to highlight the quality standards achievable through this method. Understanding these nuances helps stakeholders make informed decisions about integrating this intermediate into their supply chains.
Q: What are the primary advantages of the sodium bromate oxidation method over traditional thermal degradation?
A: The sodium bromate method offers a controlled one-step pathway that significantly reduces reaction time from over 10 hours to just 2-5 hours, while avoiding the complex mixture of 50+ split products associated with high-temperature thermal cleavage.
Q: How does pH control impact the purity of 4-oxo-beta-damascenone?
A: Maintaining a strict pH range of 1-3 is critical because the oxidation potential of bromate is acidity-dependent; this precise control ensures complete conversion of the starting material while minimizing the formation of over-oxidized impurities.
Q: Is this synthesis method scalable for industrial production of tobacco aromatics?
A: Yes, the process utilizes common industrial reagents like sulfuric acid and sodium bromate in standard hydrophilic solvents, and the workup involves simple phase separation and recrystallization, making it highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Oxo-Beta-Damascenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of premium flavor and fragrance formulations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4-oxo-beta-damascenone performs consistently in your final applications. Whether you require custom synthesis or established catalog items, our infrastructure is designed to support your growth with reliability and technical excellence.
We invite you to collaborate with us to optimize your supply chain and reduce your overall material costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can become a strategic asset for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
