Scalable Synthesis of 3-Hydroxymethyl Tetrahydrofuran for High-Performance Agrochemical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to synthesize critical heterocyclic building blocks, and patent CN102276559B presents a significant breakthrough in the production of 3-hydroxymethyl tetrahydrofuran. This compound serves as a pivotal intermediate for the third-generation nicotinic insecticide MTI-446, known for its potent systemic activity and low toxicity profile. The disclosed method fundamentally shifts the synthetic paradigm by utilizing gamma-butyrolactone (GBL) as the primary feedstock, replacing older, less efficient routes that relied on diethyl malonate. By leveraging a robust alpha-substitution followed by a catalytic reduction strategy, this technology offers a streamlined approach that addresses both economic and environmental concerns inherent in fine chemical manufacturing. For R&D directors and supply chain managers, understanding this transition is crucial for securing reliable agrochemical intermediate suppliers capable of delivering high-purity materials at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-hydroxymethyl tetrahydrofuran has been plagued by inefficiencies and safety hazards associated with traditional malonate chemistry. As detailed in prior art such as WO2005065689, the conventional route involves the reaction of diethyl malonate with ethyl chloroacetate to form 2-ethoxy acyl-diethyl succinate, a step that typically suffers from mediocre yields around 64%. Furthermore, the subsequent reduction of this intermediate requires a massive excess of sodium borohydride (ratios up to 1:6), which not only drives up raw material costs but also generates significant amounts of hydrogen gas, posing serious operational safety risks in large-scale reactors. The process concludes with a dehydration step that often necessitates complex column chromatography for purification, further eroding the overall process mass intensity and increasing waste disposal burdens.
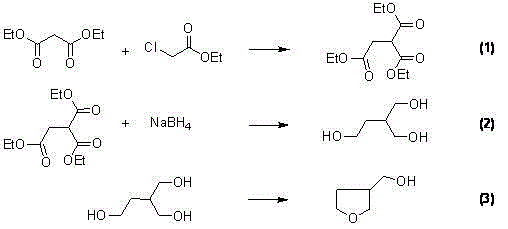
The Novel Approach
In stark contrast, the novel methodology outlined in CN102276559B utilizes gamma-butyrolactone, a commodity chemical, to construct the tetrahydrofuran core with greater atom economy. This approach bifurcates into two viable technical schemes: a direct reduction pathway and a reductive ring-opening followed by cyclization. The direct route allows for the conversion of alpha-substituted gamma-butyrolactone directly into the target product using metal borohydrides under Lewis acid catalysis, effectively collapsing multiple steps into a single pot operation. This consolidation not only reduces the total reaction time but also minimizes the handling of unstable intermediates, thereby enhancing the overall safety profile and throughput of the manufacturing process. The ability to tune the reaction conditions between -5°C and 50°C provides operators with flexible control over reaction kinetics, ensuring consistent quality across different batch sizes.
Mechanistic Insights into Gamma-Butyrolactone Functionalization and Reduction
The core of this innovation lies in the precise functionalization of the gamma-butyrolactone ring at the alpha-position. Under the influence of strong bases such as sodium methylate, sodium hydride, or lithium diisopropylamide, the acidic alpha-protons of the lactone are deprotonated to form a reactive enolate species. This nucleophile then attacks electrophiles such as diethyl carbonate, methyl formate, or formaldehyde, introducing the necessary carbon framework for the hydroxymethyl group. The choice of solvent plays a critical role here; aromatic hydrocarbons like toluene are preferred for the substitution step to facilitate the precipitation of salt by-products and drive the equilibrium forward. This step is highly efficient, with patent embodiments reporting yields exceeding 90% for the formation of alpha-ethoxy acetyl-gamma-butyrolactone, establishing a high-quality foundation for the subsequent transformation.
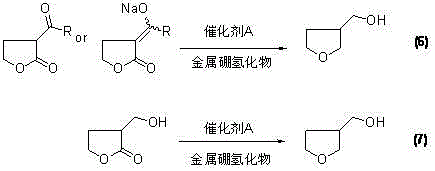
Following the substitution, the reduction mechanism is orchestrated by the synergistic action of metal borohydrides and Lewis acid catalysts. In the direct reduction scheme, catalysts such as boron trifluoride (BF3) or aluminum chloride (AlCl3) activate the carbonyl groups of the lactone and the ester side chain, making them susceptible to hydride attack from reagents like NaBH4 or LiBH4. This dual activation ensures that both the ring carbonyl and the side-chain functionality are reduced simultaneously or sequentially to form the cyclic ether structure directly. Alternatively, the second technical scheme employs a reductive ring-opening strategy where the lactone ring is cleaved to form a linear diol intermediate, specifically 2-hydroxymethyl-1,4-butanediol. This linear precursor is then subjected to acid-catalyzed cyclization using catalysts like polyphosphoric acid or p-toluenesulfonic acid, which promotes intramolecular dehydration to close the tetrahydrofuran ring. This alternative pathway has demonstrated exceptional efficacy, with certain embodiments achieving yields as high as 85% in the cyclization step, offering a robust fallback for optimizing overall process yield.
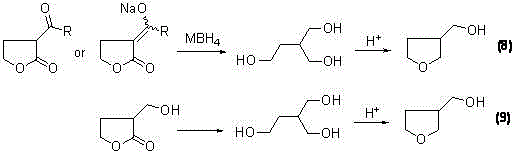
How to Synthesize 3-Hydroxymethyl Tetrahydrofuran Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize the formation of the desired regioisomer while minimizing oligomerization. The process begins with the generation of the alpha-substituted lactone, followed by the critical reduction phase where the choice of catalyst loading (typically 5-20% molar ratio) dictates the reaction rate. Operators must ensure rigorous exclusion of moisture during the borohydride addition to prevent premature decomposition of the reducing agent. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined below to guide process engineers in replicating these high-yield results.
- Perform alpha-substitution of gamma-butyrolactone using a strong base (e.g., sodium methylate) and an ester compound or formaldehyde in a solvent like toluene at reflux temperatures.
- Conduct a reduction reaction on the alpha-substituted intermediate using a metal borohydride (e.g., NaBH4) and a Lewis acid catalyst (e.g., BF3) in THF or alcohol solvents.
- Neutralize the reaction mixture with acid, filter, concentrate, and purify via rectification to obtain high-purity 3-hydroxymethyl tetrahydrofuran.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift to a gamma-butyrolactone-based synthesis offers substantial strategic advantages regarding cost stability and supply security. By moving away from multi-step malonate chemistries that require specialized precursors, manufacturers can leverage the abundant global supply of GBL and simple esters, insulating the supply chain from volatility in niche reagent markets. The reduction in unit operations—from three distinct stages in the old method to potentially two in the new method—translates directly into lower capital expenditure on equipment and reduced utility consumption per kilogram of product. Furthermore, the elimination of hazardous hydrogen evolution steps simplifies facility safety requirements, potentially lowering insurance and compliance costs associated with explosive atmosphere management.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom efficiency of the substitution step and the reduced consumption of expensive reducing agents. Unlike previous methods that required a six-fold excess of sodium borohydride, this optimized protocol operates with near-stoichiometric ratios (1:1 to 1:1.5), drastically cutting raw material expenses. Additionally, the ability to use common solvents like toluene and methanol, which are easily recovered and recycled, further diminishes the variable costs associated with solvent procurement and waste treatment, leading to a significantly leaner cost structure for the final active ingredient.
- Enhanced Supply Chain Reliability: Utilizing gamma-butyrolactone as a starting material ensures a robust supply chain, as GBL is a high-volume commodity chemical produced by multiple global vendors, unlike specialized malonate derivatives which may have limited sourcing options. The simplicity of the reaction conditions, which do not require extreme pressures or cryogenic temperatures beyond standard industrial cooling capabilities, means that production can be easily transferred between different manufacturing sites without extensive requalification. This flexibility is critical for maintaining continuity of supply for key agrochemical intermediates, especially during periods of market disruption or logistical constraints.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower due to the generation of less saline waste and the avoidance of heavy metal catalysts in the primary reduction steps. The process produces minimal wastewater compared to traditional aqueous workups, and the organic waste streams are composed largely of recyclable solvents. This alignment with green chemistry principles facilitates easier regulatory approval and permits for expansion, allowing manufacturers to scale production from pilot batches to multi-ton annual capacities with fewer environmental hurdles, ensuring long-term sustainability for high-purity agrochemical intermediate manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on yield expectations, safety protocols, and scalability factors for potential partners and licensees.
Q: What are the key advantages of the gamma-butyrolactone route over traditional malonate methods?
A: The gamma-butyrolactone route eliminates the need for hazardous hydrogen evolution associated with large excesses of reducing agents in malonate routes. It utilizes cheaper, readily available starting materials and achieves comparable or superior yields with fewer purification steps, significantly simplifying the operational workflow.
Q: Can this synthesis method be scaled for industrial production of agrochemical intermediates?
A: Yes, the patent explicitly designs the process for industrial suitability. It employs common solvents like toluene and THF, avoids exotic catalysts, and operates at moderate temperatures (-5°C to 50°C), making it highly adaptable for large-scale reactor setups without requiring specialized high-pressure equipment.
Q: How does the process control impurities during the reduction step?
A: Impurity control is managed through the precise selection of Lewis acid catalysts such as BF3 or AlCl3 and strict temperature regulation during the borohydride reduction. The subsequent acid neutralization and filtration steps effectively remove metal salts, while final rectification ensures the removal of organic by-products to meet stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxymethyl Tetrahydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that 3-hydroxymethyl tetrahydrofuran plays in the development of next-generation crop protection agents and pharmaceutical compounds. Our technical team has extensively analyzed the pathways described in CN102276559B and possesses the expertise to optimize these reactions for commercial viability. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to monitor every stage of the synthesis, from the initial alpha-substitution to the final rectification, guaranteeing that impurities are kept well below acceptable thresholds for sensitive downstream applications.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our mastery of this gamma-butyrolactone chemistry can drive value and efficiency in your manufacturing operations.
