Scalable Manufacturing of High-Performance Bidentate Oxazoline Chiral Ligands for Asymmetric Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and fine chemical sectors. Patent CN111606868B represents a significant technological breakthrough in this domain, disclosing a robust and highly efficient preparation method for a novel class of bidentate oxazoline chiral ligands. These ligands are critical components in modern asymmetric synthesis, serving as the chiral environment that dictates the stereochemical outcome of complex transformations. The invention addresses a longstanding gap in the literature where, despite the known efficacy of such ligands in specific catalytic cycles, a practical and scalable synthetic route was previously unavailable. By leveraging simple, economical starting materials and optimizing reaction conditions, this patent provides a viable pathway for the industrial production of these high-value compounds, ensuring consistent quality and supply continuity for downstream applications in drug discovery and process chemistry.
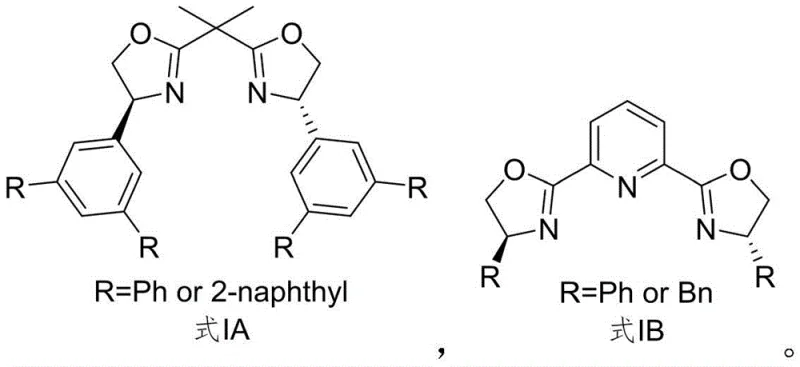
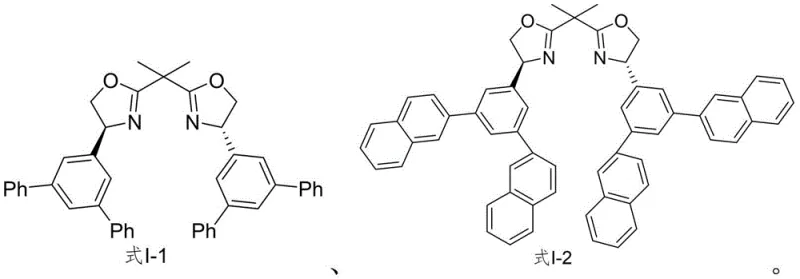
The development of these specific ligand architectures, such as those depicted in the structural formulas I-1 and I-2, underscores a strategic shift towards designing catalysts that offer superior steric control. The ability to synthesize these molecules reliably allows research and development teams to explore new chemical spaces with greater confidence in stereochemical fidelity. For procurement and supply chain leaders, the existence of a defined patent-protected synthesis route translates directly into reduced supply risk. No longer dependent on obscure or custom-only sources, manufacturers can now establish secure supply lines for these essential chiral auxiliaries. This technological advancement not only supports the immediate needs of asymmetric catalysis but also lays the groundwork for future innovations in chiral pool synthesis, making it a pivotal development for any organization focused on high-purity pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex bidentate oxazoline ligands has been fraught with challenges that hinder their widespread adoption in industrial settings. Traditional methods often rely on multi-step sequences involving expensive chiral pool starting materials or resolution processes that inherently limit the maximum theoretical yield to fifty percent. Furthermore, many existing protocols utilize harsh reaction conditions or sensitive reagents that are difficult to manage on a large scale, leading to inconsistent batch-to-batch reproducibility. The lack of a generalized synthetic strategy means that minor structural modifications to the ligand often require a complete redesign of the synthetic route, resulting in significant time and resource expenditures. In the context of the specific ligands discussed in this patent, prior art failed to provide a method to access the sterically demanding architectures necessary for high-performance catalysis, leaving a critical void in the toolkit available to process chemists seeking to optimize enantioselective transformations.
The Novel Approach
The methodology disclosed in patent CN111606868B offers a transformative solution to these historical bottlenecks by introducing a rational, convergent synthetic design. This novel approach utilizes a modular strategy that allows for the facile introduction of diverse aryl groups, such as phenyl or 2-naphthyl moieties, through robust cross-coupling chemistry. By starting from readily available 3,5-dibromobenzaldehyde, the process bypasses the need for rare natural products, significantly lowering the barrier to entry for production. The route is characterized by high-yielding transformations and the use of standard organic synthesis reagents, which simplifies purification and workup procedures. This streamlined process not only enhances the overall efficiency of ligand production but also ensures that the critical chiral information is installed and preserved with high fidelity throughout the synthesis. The result is a versatile platform technology capable of delivering high-purity chiral ligands that outperform conventional options like PyBOX in specific asymmetric reactions.
Mechanistic Insights into the Multi-Step Synthesis of Bidentate Oxazoline Ligands
The synthetic pathway detailed in the patent is a masterclass in modern organic synthesis, integrating several distinct mechanistic manifolds to construct the complex target molecule. The sequence initiates with a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction, which serves as the cornerstone for building the biaryl backbone. This step is crucial as it establishes the steric environment around the central core, utilizing arylboronic acids to introduce the desired substituents with high precision. Following this, the aldehyde functionality undergoes a Corey-Chaykovsky epoxidation using trimethylsulfonium iodide, generating an epoxide intermediate that is subsequently opened under acidic conditions to yield a vicinal diol. This diol is then selectively protected and oxidized to a ketone, setting the stage for the installation of chirality. The introduction of the chiral center is achieved through the condensation with (S)-tert-butylsulfinamide, a powerful chiral auxiliary that directs the stereochemistry of subsequent reductions. The use of diisobutylaluminum hydride (DIBAL-H) for the reduction of the sulfinylimine is a critical step, proceeding with high diastereoselectivity to establish the correct absolute configuration required for the final ligand activity.
The final stages of the synthesis involve the construction of the oxazoline rings themselves, which coordinate to the metal center during catalysis. After deprotection of the silyl ether and removal of the sulfinyl group, the resulting amino alcohol is coupled with dimethylmalonyl dichloride to form a bis-amide intermediate. The cyclization to form the oxazoline rings is effected using p-toluenesulfonyl chloride and a base, a transformation that closes the heterocyclic rings and locks the conformation of the ligand. This specific cyclization mechanism is vital for creating the rigid, C2-symmetric (or pseudo-symmetric) environment that defines the ligand's performance. Throughout this ten-step sequence, the careful selection of reagents and conditions minimizes side reactions and impurity formation, ensuring that the final product meets the stringent purity specifications required for catalytic applications. The mechanistic robustness of this route allows for the scalable production of ligands that exhibit superior steric bulk compared to simpler analogues.
How to Synthesize Bidentate Oxazoline Chiral Ligands Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and optical purity. The process begins with the coupling of 3,5-dibromobenzaldehyde and proceeds through oxidation, chiral induction, and cyclization steps. Each stage has been optimized to balance reaction rate with selectivity, utilizing common solvents like THF, DCM, and toluene. The detailed standardized synthesis steps, including specific molar ratios, temperatures, and workup procedures, are outlined in the structured guide below to ensure reproducibility for process development teams.
- Perform Suzuki-Miyaura coupling of 3,5-dibromobenzaldehyde with arylboronic acid to form the biaryl aldehyde intermediate.
- Execute epoxidation using trimethylsulfonium iodide followed by acid-catalyzed ring opening to generate the diol precursor.
- Condense the oxidized intermediate with (S)-tert-butylsulfinamide, reduce with DIBAL-H, and cyclize with dimethylmalonyl dichloride to form the final oxazoline rings.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the implementation of this patented synthesis route offers substantial strategic advantages for organizations managing the supply of chiral intermediates. The primary benefit lies in the drastic simplification of the supply chain for raw materials. By relying on commodity chemicals such as 3,5-dibromobenzaldehyde and common boronic acids, the process eliminates the volatility associated with sourcing specialized chiral building blocks. This shift to widely available feedstocks ensures a stable and continuous supply, mitigating the risk of production delays caused by raw material shortages. Furthermore, the avoidance of precious metal catalysts in the later stages of the synthesis, relying instead on standard organic reagents, contributes to a more predictable cost structure. The process is designed to be robust, meaning it can tolerate minor variations in input quality without compromising the final product, which is a key factor in maintaining consistent manufacturing operations.
- Cost Reduction in Manufacturing: The economic profile of this synthesis is significantly improved by the elimination of expensive resolution steps and the use of high-yielding reactions that minimize waste. The reliance on inexpensive starting materials and the ability to perform reactions under relatively mild conditions reduces the energy consumption and equipment requirements associated with production. Additionally, the high stereoselectivity achieved during the chiral induction step means that less material is lost to unwanted isomers, effectively increasing the overall process mass intensity. This efficiency translates directly into lower cost of goods sold, allowing for more competitive pricing in the market for high-purity pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: The modular nature of the synthetic route allows for flexible manufacturing strategies that can adapt to fluctuating demand. Because the key intermediates are stable and can be stored or produced in advance, manufacturers can build inventory buffers to ensure uninterrupted delivery to clients. The use of standard chemical transformations also means that the process can be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without the need for specialized equipment or proprietary technology transfers. This flexibility enhances the resilience of the supply chain, ensuring that critical chiral ligands are available when needed for time-sensitive drug development projects.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, having been designed with commercial production in mind from the outset. The reactions utilize solvents and reagents that are manageable on a large scale, and the workup procedures involve standard extraction and crystallization techniques that are easily implemented in multipurpose reactors. From an environmental standpoint, the process avoids the generation of heavy metal waste streams associated with some alternative catalytic methods, simplifying waste treatment and disposal. The high atom economy of the key coupling and cyclization steps further reduces the environmental footprint, aligning with the growing industry emphasis on green chemistry and sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bidentate oxazoline chiral ligands. The answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on the capabilities and limitations of this synthetic technology. Understanding these details is essential for making informed decisions about integrating these ligands into your catalytic processes.
Q: What distinguishes this bidentate oxazoline ligand from traditional PyBOX ligands?
A: The novel ligand features a unique structural framework with significant steric hindrance derived from the specific aryl substitution pattern, which has been demonstrated to significantly improve enantioselectivity in zinc-catalyzed [4+3] asymmetric cyclization reactions compared to standard PyBOX systems.
Q: Is the synthetic route suitable for large-scale commercial production?
A: Yes, the process utilizes robust and scalable reactions such as Suzuki coupling and standard acylation, avoiding exotic catalysts or extreme conditions, which facilitates commercial scale-up from kilogram to metric ton quantities.
Q: What are the key starting materials for this synthesis?
A: The synthesis begins with economically viable and widely available commodity chemicals, specifically 3,5-dibromobenzaldehyde and various arylboronic acids, ensuring a stable and cost-effective supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bidentate Oxazoline Chiral Ligand Supplier
The successful translation of this patented laboratory synthesis into a commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering comprehensive CDMO services tailored to the complex needs of chiral intermediate production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to ton-scale manufacturing is seamless and efficient. We understand that the integrity of the chiral center is paramount, which is why our facilities are equipped with rigorous QC labs and analytical instrumentation capable of verifying stringent purity specifications and enantiomeric excess values. By partnering with us, you gain access to a supply chain that is not only reliable but also technically sophisticated enough to handle the nuances of asymmetric synthesis.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these critical chiral ligands. Whether you require a Customized Cost-Saving Analysis for your current sourcing strategy or need specific COA data to validate our material against your internal standards, we are prepared to provide the support you need. We encourage you to request route feasibility assessments to explore how this patented technology can be integrated into your broader manufacturing portfolio. Let us help you secure a competitive advantage through superior quality and supply reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
