Advanced Asymmetric Catalysis for Commercial Production of (R)-Natural Jasmine Lactone
The global demand for optically pure aroma compounds has driven significant innovation in asymmetric synthesis, particularly for high-value ingredients like (R)-Natural Jasmine Lactone. Patent CN106946823B discloses a breakthrough methodology that addresses the longstanding challenges of producing this key fragrance molecule with high enantiomeric excess. Unlike traditional racemic syntheses that require costly and wasteful resolution steps, this novel approach utilizes a direct asymmetric alkynylation strategy to establish the critical stereogenic center early in the synthetic sequence. The target molecule, characterized by its distinct chiral carbon at the 5-position, is essential for replicating the authentic scent profile found in natural jasmine and tuberose essential oils. 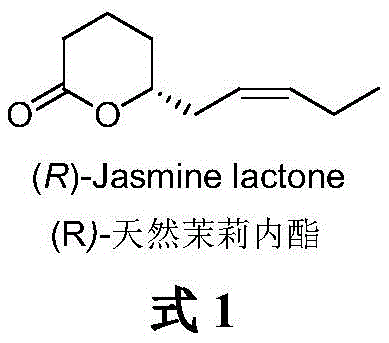 This structural fidelity is paramount for R&D directors in the flavor and fragrance industry who seek to replace expensive natural extracts with cost-effective, yet sensorially identical, synthetic alternatives. The patent outlines a robust four-step pathway that begins with readily available leaf alcohol derivatives, ensuring a secure supply chain foundation. By leveraging specific ligand-accelerated catalysis, the process achieves a total yield of 42 percent with an impressive optical purity of 95 percent, setting a new benchmark for efficiency in lactone synthesis.
This structural fidelity is paramount for R&D directors in the flavor and fragrance industry who seek to replace expensive natural extracts with cost-effective, yet sensorially identical, synthetic alternatives. The patent outlines a robust four-step pathway that begins with readily available leaf alcohol derivatives, ensuring a secure supply chain foundation. By leveraging specific ligand-accelerated catalysis, the process achieves a total yield of 42 percent with an impressive optical purity of 95 percent, setting a new benchmark for efficiency in lactone synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active jasmine lactone has been plagued by inherent inefficiencies associated with chiral resolution and multi-step enzymatic processes. Traditional chemical resolution methods typically involve hydrolyzing a racemic mixture and forming diastereomeric salts with chiral amines, a process that theoretically caps the maximum yield at 50 percent due to the discard of the unwanted enantiomer. Furthermore, these resolution protocols often require multiple recrystallization steps to achieve acceptable optical purity, leading to significant solvent consumption and extended processing times that inflate manufacturing costs. Enzymatic approaches, while greener in principle, often suffer from narrow substrate specificity and the need for complex protection-deprotection sequences to manage reactive functional groups during the synthesis. These legacy methods also frequently rely on precious metal catalysts for hydrogenation steps, introducing risks of heavy metal contamination that require rigorous and expensive purification protocols to meet stringent regulatory standards for food and cosmetic applications.
The Novel Approach
The methodology described in the patent represents a paradigm shift by employing a convergent synthetic strategy that constructs the carbon skeleton and the chiral center simultaneously. Instead of resolving a racemate, the process utilizes an asymmetric addition reaction between an aldehyde and an alkyne, catalyzed by a chiral ProPhenol ligand system. This direct construction of the propargylic alcohol intermediate ensures that the chirality is introduced with high fidelity from the outset, effectively doubling the theoretical material efficiency compared to resolution techniques. 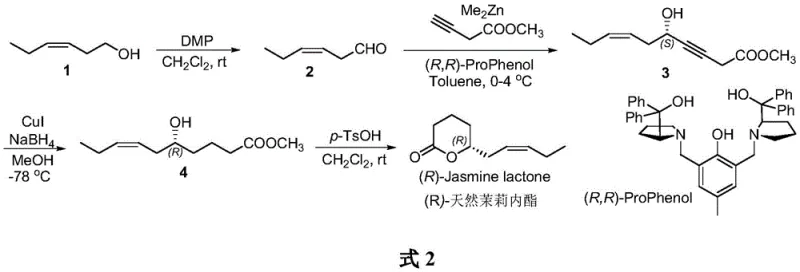 The subsequent steps involve a chemoselective reduction of the triple bond using a copper-mediated system, which avoids the over-reduction risks often seen with palladium catalysts, followed by a straightforward acid-catalyzed lactonization. This streamlined route not only simplifies the operational workflow by reducing the number of isolation steps but also utilizes reagents that are scalable and commercially accessible, making it an ideal candidate for industrial adoption by a reliable flavor and fragrance intermediate supplier.
The subsequent steps involve a chemoselective reduction of the triple bond using a copper-mediated system, which avoids the over-reduction risks often seen with palladium catalysts, followed by a straightforward acid-catalyzed lactonization. This streamlined route not only simplifies the operational workflow by reducing the number of isolation steps but also utilizes reagents that are scalable and commercially accessible, making it an ideal candidate for industrial adoption by a reliable flavor and fragrance intermediate supplier.
Mechanistic Insights into Asymmetric Alkynylation and Reduction
The core of this synthetic innovation lies in the enantioselective addition of methyl butynoate to (Z)-3-hexenal, mediated by a dimethyl zinc and (R,R)-ProPhenol complex. In this catalytic cycle, the chiral ligand coordinates with the zinc species to create a rigid chiral environment that directs the nucleophilic attack of the alkyne to a specific face of the aldehyde carbonyl group. This precise stereocontrol is critical for generating the (S)-configured propargylic alcohol intermediate, which subsequently translates into the desired (R)-configuration in the final lactone product after the necessary functional group transformations. The use of the ProPhenol ligand is particularly advantageous as it facilitates the formation of the active zinc acetylide species under mild conditions, minimizing side reactions such as aldol condensation that could compromise the purity of the intermediate. For R&D teams, understanding this mechanism highlights the importance of ligand purity and stoichiometry in maintaining the high 95 percent ee observed in the patent examples.
Following the establishment of chirality, the conversion of the alkyne moiety to the cis-alkene is achieved through a specialized reduction protocol using copper iodide and sodium borohydride in methanol at cryogenic temperatures. This step functions as a chemical alternative to Lindlar hydrogenation, offering superior control over the stereochemistry of the resulting double bond to ensure the (Z)-geometry is preserved, which is vital for the biological activity and scent profile of the molecule. The mechanism likely involves the formation of a copper hydride species that adds across the triple bond in a syn-fashion, preventing isomerization to the thermodynamically more stable trans-isomer. Finally, the intramolecular transesterification catalyzed by p-toluenesulfonic acid closes the six-membered ring, driven by the entropic favorability of lactone formation. This mechanistic understanding assures procurement managers that the process is robust and less sensitive to the variability often encountered in biocatalytic systems.
How to Synthesize (R)-Natural Jasmine Lactone Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing strict temperature control and inert atmosphere conditions to maximize yield and selectivity. The process begins with the oxidation of the starting alcohol using DMP, followed by the crucial asymmetric addition which requires careful maintenance of temperatures between 0 and 4 degrees Celsius to prevent racemization. The subsequent reduction step demands even lower temperatures of -78 degrees Celsius to ensure the chemoselectivity of the copper-mediated reduction, highlighting the need for precise thermal management in reactor design. For a comprehensive guide on the specific molar ratios, workup procedures, and purification techniques required to replicate these results, please refer to the standardized synthesis instructions below.
- Oxidize (Z)-3-hexen-1-ol to (Z)-3-hexenal using DMP reagent in dichloromethane at room temperature.
- Perform asymmetric addition with methyl butynoate using (R,R)-ProPhenol ligand and dimethyl zinc at 0-4°C to establish chirality.
- Reduce the triple bond to a double bond using CuI and NaBH4 in methanol at -78°C, followed by acid-catalyzed cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented route offers substantial advantages in terms of cost structure and supply chain resilience for manufacturers of fine chemicals. By eliminating the need for chiral resolution, the process inherently improves the overall mass balance of the synthesis, meaning that less raw material is required to produce the same amount of active ingredient compared to traditional methods. This improvement in atom economy directly translates to significant cost reduction in synthetic flavor manufacturing, as the expense associated with purchasing and disposing of the unwanted enantiomer is completely removed from the cost model. Furthermore, the avoidance of high-pressure hydrogenation equipment reduces capital expenditure requirements for production facilities, allowing for more flexible manufacturing arrangements that can be scaled up rapidly to meet market demand without extensive infrastructure upgrades.
- Cost Reduction in Manufacturing: The elimination of chiral resolving agents and the associated recrystallization solvents drastically lowers the variable costs per kilogram of the final product. Since the synthesis builds chirality directly rather than separating it, the theoretical yield ceiling is effectively doubled, providing a massive buffer against raw material price fluctuations. Additionally, the use of base metals like copper instead of precious metals like palladium or platinum for the reduction step further decreases the catalyst cost burden and simplifies the recovery of valuable metals from waste streams. These factors combine to create a highly competitive cost profile that allows suppliers to offer high-purity intermediates at more attractive price points without sacrificing margin.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as (Z)-3-hexen-1-ol and methyl butynoate, are commodity chemicals with well-established global supply chains, reducing the risk of bottlenecks associated with exotic or proprietary reagents. The robustness of the chemical steps, which do not rely on sensitive biological enzymes that can vary in activity between batches, ensures consistent production output and reliable lead times for downstream customers. This stability is crucial for supply chain heads who need to guarantee continuity of supply for long-term contracts in the personal care and food industries, where formulation changes due to ingredient shortages are highly disruptive and costly.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize common organic solvents like dichloromethane and toluene, which are easily recovered and recycled in modern chemical plants, aligning with increasingly strict environmental regulations. The absence of heavy metal catalysts that are difficult to remove simplifies the purification process, reducing the generation of hazardous waste and lowering the environmental footprint of the manufacturing process. This green chemistry profile not only facilitates easier regulatory approval for use in food and cosmetics but also enhances the brand value of the final product by appealing to eco-conscious consumers and retailers who prioritize sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of (R)-Natural Jasmine Lactone using this advanced catalytic method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's viability. Understanding these details helps stakeholders make informed decisions about integrating this new supply source into their existing procurement strategies and product formulations.
Q: What is the optical purity achieved in this new synthesis method?
A: The patented method achieves an optical purity of 95% ee for the final (R)-natural jasmine lactone product, ensuring high sensory quality for premium fragrance applications.
Q: How does this route compare to traditional chiral resolution methods?
A: Unlike traditional resolution which theoretically wastes 50% of the material, this asymmetric catalytic route builds chirality directly, offering better atom economy and eliminating the need for diastereomer separation.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the process utilizes mild temperatures ranging from -78°C to room temperature and avoids high-pressure hydrogenation equipment, making it highly adaptable for standard chemical reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Natural Jasmine Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality chiral intermediates that meet the exacting standards of the global flavor and fragrance industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of (R)-Natural Jasmine Lactone delivers the consistent olfactory profile required for premium perfume and food applications. Our capability to implement complex asymmetric synthesis routes like the one described in CN106946823B demonstrates our technical depth and dedication to innovation.
We invite procurement leaders and R&D directors to contact our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this more efficient manufacturing method. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a partnership that drives both quality and value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
