Scalable Synthesis of 4-Chloro-7-Methoxyindole-2-Boric Acid for Advanced Pharmaceutical Applications
The pharmaceutical industry continuously demands efficient, scalable, and cost-effective routes for complex heterocyclic intermediates, particularly those serving as building blocks for kinase inhibitors and other targeted therapies. Patent CN102850384A introduces a robust synthetic methodology for producing 4-chloro-7-methoxyindole-2-boric acid, a valuable scaffold in medicinal chemistry. This technology addresses critical bottlenecks found in traditional syntheses, such as harsh reaction conditions, low overall yields, and the reliance on labor-intensive purification techniques like column chromatography. By leveraging a strategic sequence of Grignard cyclization, nitrogen protection, and directed lithiation-boronation, this process offers a streamlined pathway that significantly enhances operational simplicity. For R&D directors and process chemists, the ability to bypass chromatographic purification while maintaining high structural integrity represents a substantial advancement in process chemistry. Furthermore, the use of readily available starting materials, specifically 4-chloro-1-methoxy-2-nitrobenzene, ensures that the supply chain remains resilient and cost-efficient. This report analyzes the technical merits of this patented route, evaluating its potential for commercial adoption and its alignment with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted indole boronic acids has been plagued by significant technical challenges that hinder large-scale production. Conventional routes often involve multiple steps with incompatible functional groups, requiring extensive protecting group strategies that add both time and cost to the manufacturing process. A primary drawback in legacy methods is the reliance on column chromatography for purification, which is notoriously difficult to scale beyond the laboratory bench due to the massive volumes of solvents and silica gel required. This not only inflates the cost of goods sold (COGS) but also creates substantial environmental waste, conflicting with modern sustainability goals. Additionally, many traditional protocols utilize unstable intermediates that degrade rapidly under ambient conditions, leading to inconsistent yields and variable product quality. The harsh reaction conditions often cited in prior art, such as extreme temperatures or the use of toxic heavy metal catalysts, further complicate safety protocols and regulatory compliance. These factors collectively create a high barrier to entry for reliable suppliers attempting to bring these critical intermediates to the market at a competitive price point.
The Novel Approach
In stark contrast to these legacy issues, the methodology disclosed in CN102850384A presents a refined and industrial-friendly solution. The core innovation lies in a concise three-step sequence that transforms a simple nitrobenzene derivative into the target boronic acid with remarkable efficiency. The process initiates with a cyclization reaction using vinylmagnesium bromide, which constructs the indole core in a single operation. Following this, a Boc protection step stabilizes the indole nitrogen, preventing unwanted side reactions during the subsequent metalation. The final stage involves a lithiation-boronation sequence followed by a convenient one-pot hydrolysis, which directly yields the free boronic acid without the need for intermediate isolation or chromatographic purification. This approach drastically simplifies the workflow, reducing the number of unit operations and minimizing solvent consumption. By designing the synthesis to allow for purification via crystallization and filtration, the process becomes inherently scalable, addressing the needs of procurement managers looking for cost reduction in pharmaceutical intermediate manufacturing.
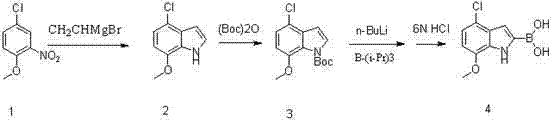
Mechanistic Insights into Grignard Cyclization and Lithiation-Boronation
The success of this synthetic route hinges on the precise control of organometallic reactions, beginning with the formation of the indole ring. The reaction of 4-chloro-1-methoxy-2-nitrobenzene with vinylmagnesium bromide is a sophisticated transformation where the Grignard reagent acts as both a nucleophile and a base. At cryogenic temperatures around -70°C, the vinyl magnesium species attacks the electron-deficient nitro group, initiating a cascade that eventually leads to ring closure and the formation of the indole system. Maintaining this low temperature is critical to suppress competing pathways, such as the polymerization of the vinyl Grignard reagent or non-selective addition to other sites on the aromatic ring. The choice of tetrahydrofuran (THF) as the solvent is paramount, as it effectively solvates the magnesium cation, enhancing the nucleophilicity of the reagent while ensuring the stability of the transition states involved in the cyclization. This step sets the foundation for the entire synthesis, establishing the core heterocyclic structure with the correct regiochemistry required for downstream functionalization.
Following the construction of the indole core, the introduction of the boronic acid moiety at the C2 position is achieved through a directed metalation strategy. The presence of the Boc protecting group on the indole nitrogen serves a dual purpose: it prevents the nitrogen from reacting with the strong base used in the next step, and it electronically activates the C2 position for deprotonation. When treated with n-butyllithium at low temperatures, the proton at the C2 position is abstracted to form a stable lithio-indole intermediate. This organolithium species is then quenched with triisopropyl borate, transferring the boron atom to the carbon framework. The subsequent acidic hydrolysis cleaves the borate ester and removes any remaining protecting groups if necessary, though in this specific route, the Boc group may remain or be removed depending on the specific hydrolysis conditions employed. This mechanism ensures high regioselectivity, minimizing the formation of isomeric impurities that are difficult to separate, thereby guaranteeing the high purity specifications required for API synthesis.
How to Synthesize 4-Chloro-7-Methoxyindole-2-Boric Acid Efficiently
To implement this synthesis effectively in a pilot or production plant, strict adherence to the patented operational parameters is essential. The process begins with the preparation of the Grignard reaction mixture under an inert atmosphere to prevent moisture ingress, which could deactivate the reagent. The addition of vinylmagnesium bromide must be controlled carefully to manage the exotherm, followed by a prolonged stirring period at -70°C to ensure complete conversion to the indole intermediate. Once the cyclization is complete, the reaction is quenched into ice water, and the product is isolated via extraction and crystallization, yielding a beige solid that serves as the substrate for the next step. The subsequent protection and lithiation steps follow similar protocols of temperature control and stoichiometric precision. For a comprehensive understanding of the specific reagent quantities, stirring times, and workup procedures, please refer to the standardized guide below.
- Cyclization of 4-chloro-1-methoxy-2-nitrobenzene with vinylmagnesium bromide in THF at -70°C to form the indole core.
- Protection of the indole nitrogen using di-tert-butyl dicarbonate ((Boc)2O) and DMAP catalyst to stabilize the intermediate.
- Lithiation at the C2 position using n-butyllithium followed by quenching with triisopropyl borate and acidic hydrolysis to yield the final boronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for supply chain stability and cost management. The elimination of column chromatography is perhaps the most significant economic driver, as it removes a major bottleneck in production throughput and drastically reduces the consumption of silica gel and organic solvents. This simplification translates directly into lower operating expenses and a smaller environmental footprint, aligning with the increasing regulatory pressure on chemical manufacturers to adopt greener processes. Furthermore, the use of commodity chemicals such as 4-chloro-1-methoxy-2-nitrobenzene and vinylmagnesium bromide ensures that raw material sourcing is straightforward and less susceptible to market volatility compared to exotic or custom-synthesized precursors. The robustness of the reaction conditions, particularly the tolerance for standard industrial solvents like THF and ether, means that existing manufacturing infrastructure can be utilized without the need for specialized equipment upgrades. These factors combine to create a supply chain that is both agile and resilient, capable of meeting fluctuating demand without compromising on delivery timelines.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of chromatographic purification steps. In traditional fine chemical synthesis, column chromatography can account for a substantial portion of production costs due to the high price of stationary phases and the energy required for solvent recovery. By engineering a route where the product precipitates or can be purified via crystallization, the process achieves significant cost savings. Additionally, the high atom economy of the cyclization step and the efficient use of reagents minimize waste generation, further reducing disposal costs. The overall yield profile, while typical for multi-step organometallic sequences, is balanced by the low cost of inputs and the high purity of the output, resulting in a favorable cost-per-kilogram metric for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on widely available starting materials. 4-chloro-1-methoxy-2-nitrobenzene is a bulk chemical produced by multiple vendors globally, reducing the risk of single-source dependency. The synthetic steps themselves are chemically robust, meaning that minor variations in raw material quality do not necessarily lead to batch failures, providing a buffer against supply fluctuations. The scalability of the process allows manufacturers to ramp up production quickly in response to clinical trial successes or market demand spikes. This reliability is crucial for pharmaceutical companies that require consistent quality and timely delivery to maintain their own production schedules and regulatory filings.
- Scalability and Environmental Compliance: The design of this synthesis inherently supports scale-up from gram to ton quantities. The reactions are performed in homogeneous solutions, which facilitates heat transfer and mixing in large reactors, mitigating the risks associated with exothermic events. The avoidance of heavy metal catalysts simplifies the regulatory landscape, as there is no need for extensive testing and validation of residual metal levels in the final product. Moreover, the reduced solvent usage and waste generation contribute to a lower E-factor (environmental factor), making the process more sustainable. This alignment with environmental, social, and governance (ESG) criteria is increasingly important for procurement teams evaluating potential suppliers, as it future-proofs the supply chain against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-chloro-7-methoxyindole-2-boric acid. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their development pipelines.
Q: How does this synthesis route avoid column chromatography?
A: The patented process utilizes specific crystallization techniques and one-pot hydrolysis methods that allow impurities to be removed via filtration and washing, eliminating the need for expensive and time-consuming silica gel column chromatography typically required for indole purification.
Q: What are the critical temperature controls for the Grignard step?
A: The cyclization step using vinylmagnesium bromide requires strict temperature control, specifically cooling to approximately -70°C using a dry ice/acetone bath. This prevents exothermic runaway and minimizes side reactions such as polymerization of the vinyl group or over-reaction with the nitro functionality.
Q: Is this process suitable for multi-kilogram production?
A: Yes, the patent explicitly states the route is suitable for industrialization. The use of common solvents like THF, standard reagents like n-BuLi and Boc anhydride, and the avoidance of complex purification equipment make it highly adaptable for commercial scale-up from 100 kgs to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-7-Methoxyindole-2-Boric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team of expert process chemists has extensively evaluated the route described in CN102850384A and possesses the technical capability to execute this synthesis with precision and efficiency. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from clinical trials to full-scale manufacturing. Our facilities are equipped with state-of-the-art reactors capable of handling cryogenic reactions and sensitive organometallic chemistry, supported by rigorous QC labs that enforce stringent purity specifications. We are committed to delivering products that meet the highest standards of quality, consistency, and regulatory compliance, providing you with a secure foundation for your drug development programs.
We invite you to collaborate with us to optimize your supply chain and accelerate your time to market. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with us, you gain access to our deep expertise in process optimization and our commitment to continuous improvement. Please contact us today to request specific COA data, discuss route feasibility assessments, or initiate a dialogue on how we can support your long-term strategic goals. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
