Revolutionizing Biotin Synthesis: Efficient Recovery of Dextroamine from Lactone Mother Liquor
The global demand for d-biotin, a vital vitamin for animal nutrition and human health applications, has surged in recent years, driving an intense search for efficient and sustainable production methodologies. However, the synthesis of d-biotin relies heavily on a key chiral intermediate known as dextroamine, the supply of which has become increasingly precarious due to the global decline in chloramphenicol production, its traditional source. Patent CN107382752B, published in March 2021, introduces a groundbreaking physicochemical separation technique designed to recover high-purity dextroamine directly from the mother liquor generated during lactone synthesis. This innovation addresses a critical bottleneck in the supply chain by transforming what was previously considered hazardous industrial waste into a valuable, reusable feedstock. By implementing this recovery protocol, manufacturers can mitigate the volatility of raw material pricing and reduce their dependency on external suppliers who are facing their own upstream constraints. The technology represents a significant leap forward in green chemistry principles applied to fine chemical manufacturing, turning a linear consumption model into a circular economy loop.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
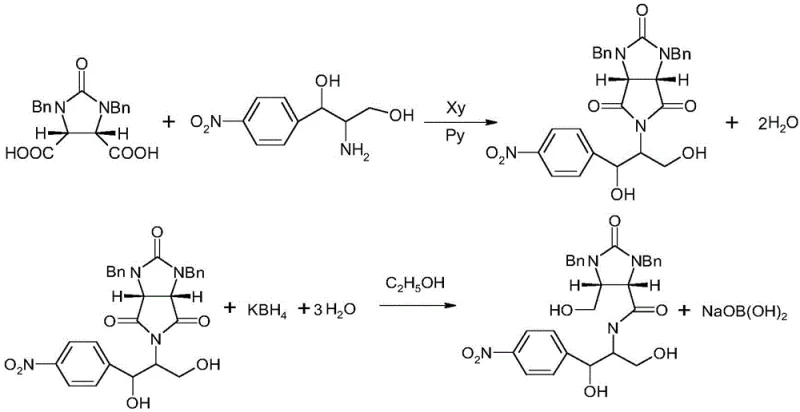
Traditionally, the production of the lactone intermediate required for d-biotin synthesis has been plagued by inefficiencies regarding raw material utilization and waste management. In the standard synthetic pathway, dextroamine reacts with specific dicarboxylic acid derivatives to form the lactone ring, a process that inevitably generates a complex mother liquor containing unreacted starting materials, inorganic salts like boric acid, and various organic byproducts. Historically, this mother liquor was treated as waste, leading to substantial economic losses given the high market value of dextroamine, which has seen price increases from tens of thousands to hundreds of thousands of currency units per ton. Furthermore, the disposal of this organic-rich waste stream imposes a heavy burden on environmental compliance teams, requiring extensive wastewater treatment protocols to handle the high chemical oxygen demand (COD) and potential toxicity. The reliance on purchasing fresh dextroamine from a shrinking market of chloramphenicol byproduct suppliers creates a fragile supply chain that is susceptible to sudden shortages and price spikes, threatening the continuity of biotin production lines globally.
The Novel Approach
The methodology disclosed in the patent offers a robust solution by leveraging differential solubility and crystallization thermodynamics to isolate dextroamine from the complex mixture. Instead of discarding the mother liquor, the process initiates with a controlled cooling phase to selectively precipitate boric acid, effectively removing a major inorganic contaminant without losing the valuable amine. Subsequent steps involve solvent exchange and reflux crystallization using common alcohols such as ethanol or isopropanol, which facilitate the separation of remaining inorganic salts from the organic amine salts. This multi-stage purification strategy ensures that the recovered dextroamine meets stringent quality specifications, with purity levels consistently achieving between 97.5% and 98.5%. By integrating this recovery loop, manufacturers can drastically reduce their net consumption of fresh raw materials, thereby insulating their operations from market fluctuations and enhancing the overall sustainability profile of their facility.
Mechanistic Insights into Crystallization and Separation Dynamics
The core of this recovery technology lies in the precise manipulation of solubility parameters across different temperature gradients and solvent systems. The initial cooling of the lactone mother liquor to a temperature range of 20-30°C is critical, as it exploits the lower solubility of boric acid in the aqueous-organic matrix at these temperatures, allowing it to crystallize out while keeping the dextroamine species in solution. Following the removal of boric acid, the filtrate undergoes vacuum evaporation to remove water, followed by the addition of an alcoholic solvent which changes the polarity of the medium. Heating this mixture to reflux ensures complete dissolution of the target compounds, and subsequent cooling to temperatures between 20-70°C induces the precipitation of residual inorganic salts, which are filtered off. The final isolation of the dextroamine hydrochloride is achieved by deep cooling the filtrate to approximately -5°C, a step that maximizes the yield of the crude salt by minimizing its solubility in the cold alcohol solvent. This systematic approach to fractional crystallization allows for the sequential removal of impurities based on their distinct physical properties.
Impurity control is further refined in the final liberation step, where the crude dextroamine hydrochloride is dissolved in water and treated with an alkali such as sodium hydroxide or potassium carbonate. Adjusting the pH to a range of 9-10 converts the water-soluble hydrochloride salt back into the free base form of dextroamine, which has significantly lower solubility in the aqueous medium at controlled temperatures of 20-25°C. This pH swing crystallization is highly effective at excluding organic byproducts that may remain soluble at alkaline pH or possess different ionization constants. The patent data indicates that maintaining the temperature during this precipitation phase for at least one hour is essential for the growth of well-defined crystals, which facilitates easier filtration and washing. This rigorous control over pH and temperature ensures that the final product is not only chemically pure but also physically suitable for direct reuse in the subsequent lactone synthesis reaction without requiring further recrystallization.
How to Synthesize Dextroamine Efficiently
The implementation of this recovery process requires careful attention to operational parameters to ensure consistent yields and purity. The procedure is designed to be scalable, moving seamlessly from laboratory verification to industrial production vessels. Operators must strictly adhere to the cooling rates and temperature setpoints defined in the patent to prevent the co-precipitation of impurities. The detailed standardized synthesis steps for recovering dextroamine from lactone mother liquor are outlined below.
- Cool the lactone mother liquor to 20-30°C to precipitate and filter out boric acid crystals.
- Evaporate the filtrate, reflux with alcohol solvent (e.g., ethanol), and cool to remove inorganic salts.
- Further cool the filtrate to -5°C to crystallize and isolate crude dextroamine hydrochloride.
- Dissolve the crude salt in water, adjust pH to 9-10 with alkali, and filter to obtain pure dextroamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this recovery technology translates into immediate and tangible strategic benefits. The primary advantage is the drastic reduction in raw material procurement costs, as the facility can internally generate a significant portion of its dextroamine demand rather than purchasing it at premium market rates. This internal sourcing capability acts as a hedge against the volatility of the global dextroamine market, which is currently constrained by regulatory pressures on chloramphenicol production. By closing the loop on material usage, companies can stabilize their production budgets and improve their gross margins on d-biotin sales. Furthermore, the simplicity of the equipment required—standard reactors, condensers, and filtration units—means that the capital expenditure for retrofitting existing plants is minimal compared to the long-term operational savings.
- Cost Reduction in Manufacturing: The elimination of waste disposal fees combined with the recovery of high-value raw materials creates a dual financial benefit. Since the process recovers between 80% and 85% of the dextroamine present in the mother liquor, the effective cost per kilogram of dextroamine utilized in the process is significantly lowered. This efficiency gain is achieved without the need for exotic catalysts or expensive reagents, relying instead on commodity chemicals like ethanol and sodium hydroxide. The ability to recycle solvents such as ethanol and isopropanol further enhances the economic viability of the process, reducing the overall variable cost of production.
- Enhanced Supply Chain Reliability: Dependence on external suppliers for critical chiral intermediates introduces risk, particularly when those suppliers are subject to their own regulatory or产能 constraints. By implementing this on-site recovery method, manufacturers secure a steady, predictable supply of dextroamine that is decoupled from the external chloramphenicol market. This self-sufficiency ensures continuous operation of the biotin synthesis line, preventing costly shutdowns due to raw material shortages. It also provides leverage in negotiations with external vendors, as the company is no longer solely dependent on their delivery schedules or pricing structures.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations that are standard in the fine chemical industry, making it easy to expand from pilot scale to multi-ton annual production. From an environmental perspective, the method significantly reduces the volume of hazardous organic waste discharged, lowering the burden on wastewater treatment facilities and ensuring compliance with increasingly strict environmental regulations. This reduction in environmental footprint enhances the company's corporate social responsibility profile and mitigates the risk of regulatory fines or production halts due to non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this dextroamine recovery technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide clarity for technical decision-makers.
Q: What is the expected purity and recovery rate of the recovered dextroamine?
A: According to patent CN107382752B, the method achieves a recovery rate between 80% and 85%, with a final product purity ranging from 97.5% to 98.5%, meeting industrial production standards.
Q: Which solvents are suitable for the reflux crystallization step?
A: The process allows for flexibility in solvent selection, including methanol, ethanol, 95% ethanol, n-propanol, and isopropanol, allowing manufacturers to optimize based on local availability and cost.
Q: How does this method address environmental concerns in biotin production?
A: By recovering valuable raw materials from mother liquor that would otherwise be discharged as hazardous organic waste, this method significantly reduces the load on wastewater treatment facilities and minimizes chemical waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dextroamine Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of securing high-quality intermediates for the uninterrupted production of vital nutrients like d-biotin. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require recovered dextroamine or fresh synthesis, your supply needs are met with precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of dextroamine we supply meets the exacting standards required for pharmaceutical and feed additive applications. Our commitment to quality ensures that your downstream synthesis of lactone and subsequent biotin production proceeds with maximum efficiency and yield.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through our advanced recovery solutions. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our dextroamine recovery technology can enhance your operational resilience and profitability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
