Advanced Enolization and Ethylidene Combined Reaction Technology for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Enolization and Ethylidene Combined Reaction Technology for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The landscape of synthetic chemistry for complex pharmaceutical intermediates is constantly evolving, driven by the need for higher purity, better selectivity, and more robust manufacturing processes. A significant breakthrough in this domain is documented in patent CN114181274B, which discloses a novel combined reaction method for enolization and ethylidene formation. This technology addresses critical challenges in the synthesis of alpha-alkyl ketone derivatives, particularly those used in the production of bile acid-based therapeutics like Obeticholic acid. By employing a unique mixed-base catalytic system, this invention stabilizes the notoriously unstable enolate intermediates, enabling a regio-specific ethylidenation reaction that was previously difficult to control with high fidelity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this patent is crucial for evaluating next-generation supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for generating ethylidene ketones often rely on the formation of enolates using single strong bases or metal reagents, which present significant drawbacks in an industrial setting. The primary issue lies in the instability of the enolization reaction product; these intermediates are highly sensitive to strong acids and bases, meaning that any mishandling during the subsequent reaction with Lewis acids can lead to immediate decomposition and total reaction failure. Furthermore, conventional silicon enolates tend to form linear intermediates rather than the desired six-membered ring structures due to the weak Lewis acidity of silicon, resulting in poor stereoselectivity. The lack of geometric control often leads to a mixture of syn-bodies and other isomers, complicating downstream purification and drastically reducing the overall yield of the target high-purity API intermediates. Additionally, prior art methods often require reaction times exceeding 48 hours and necessitate the中途 addition of reagents like trimethylbromosilane, which increases operational complexity and safety risks.
The Novel Approach
The innovative approach detailed in the patent overcomes these historical bottlenecks by introducing a synergistic mixed-base system that acts as both a catalyst and a protective group for the reaction intermediate. By utilizing a specific combination of bases, such as triethylamine and 2-methylmorpholine, in a precise mass ratio relative to the substrate, the method ensures the effective and sufficient progression of the enolization step without the degradation issues seen in single-base systems. This novel strategy allows the enol silyl ether to remain stable even in the presence of the Lewis acid catalyst required for the subsequent Mukaiyama-Aldol condensation. The result is a highly selective, regio-specific ethylidenation reaction that proceeds smoothly at moderate temperatures, significantly shortening the reaction time and eliminating the need for hazardous mid-process reagent additions. This streamlined workflow not only enhances the chemical efficiency but also aligns perfectly with the requirements for cost reduction in pharmaceutical intermediate manufacturing by minimizing waste and energy consumption.
Mechanistic Insights into Mixed-Base Catalyzed Enolization and Ethylidenation
To fully appreciate the value of this technology for a research and development team, one must delve into the specific mechanistic interactions that drive its success. The core of the innovation lies in the behavior of the silicon enolate intermediate. Unlike lithium or boron enolates, silicon enolates possess a high degree of freedom, which typically hinders high stereoselectivity. However, the patent demonstrates that by carefully selecting a mixed base with varying alkalinity—specifically a volume ratio of 2-5:1 between a strongly basic group and a weaker one—the reaction environment is optimized to favor the formation of the desired enol silyl ether. This mixed base system effectively shields the intermediate from the harsh conditions usually associated with Lewis acid catalysis. The silicon atom, while having weak Lewis acidity, forms a stable bond that prevents the premature collapse of the intermediate, allowing it to survive the transition to the ethylidenation step where it reacts with anhydrous acetaldehyde.
The subsequent ethylidenation step leverages the stability of this protected intermediate to achieve exceptional regio-specificity. In the presence of boron trifluoride and an organic solvent at controlled low temperatures ranging from -35°C to 15°C, the enol silyl ether undergoes a Mukaiyama-Aldol condensation. The mixed base continues to play a pivotal role here by modulating the electronic environment around the carbonyl group, ensuring that the acetaldehyde attacks at the correct position to form the ethylidene double bond rather than undergoing random polymerization or side reactions. This precise control over the reaction pathway results in a product profile with significantly fewer impurities, primarily limiting contaminants to unreacted starting materials which are easily removed. The ability to maintain such high selectivity while using relatively inexpensive and commercially available reagents makes this a transformative method for the commercial scale-up of complex pharmaceutical intermediates.
The versatility of this mechanism is further evidenced by its applicability to various alpha-alkyl ketone substrates beyond just bile acid derivatives. As illustrated in the reaction schemes for simpler cyclic ketones, the methodology consistently delivers high yields and purity across different molecular scaffolds. This broad substrate scope suggests that the technology is not limited to a single niche but represents a platform technology for introducing ethylidene groups into diverse chemical structures. For a procurement manager, this implies a standardized, robust process that can be adapted for multiple product lines, thereby reducing the need for bespoke, high-risk synthesis routes for new candidates.
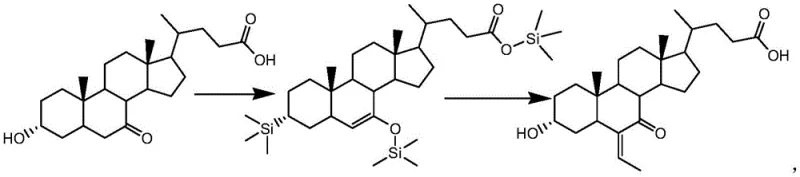
How to Synthesize 6-Ethylene-3α-Hydroxy-7-Keto-5β-Cholanic Acid Efficiently
The practical implementation of this patented method involves a straightforward two-stage sequence that is highly amenable to standard reactor setups found in modern chemical plants. The process begins with the enolization of the alpha-alkyl ketone substrate, such as 3α-hydroxy-7-keto-5β-cholanic acid, in an anhydrous organic solvent like dichloromethane or toluene. The critical innovation here is the addition of the mixed base system alongside the silicon base reagent, typically trimethylbromosilane, at a controlled temperature of 35-45°C. This step generates the stable enol silyl ether intermediate, which serves as the foundation for the subsequent transformation. Following the complete consumption of the starting material, confirmed via TLC or HPLC, the reaction mixture is cooled to sub-zero temperatures to prepare for the ethylidenation phase.
- Perform enolization of the alpha-alkyl ketone substrate with a silicon base using a novel mixed base system (triethylamine and 2-methylmorpholine) at 35-45°C to form a stable enol silyl ether.
- Conduct the ethylidenation reaction on the protected intermediate using boron trifluoride and acetaldehyde at low temperatures (-35 to 15°C) to ensure regio-specificity.
- Quench the reaction with alkaline water, separate the organic phase, and purify via crystallization to obtain high-purity ethylidene products suitable for API synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this enolization and ethylidene combined reaction method offers tangible strategic benefits that extend far beyond simple chemical yield. The primary advantage lies in the substantial cost savings achieved through process intensification and waste reduction. By eliminating the need for exotic or highly unstable reagents and replacing them with a robust mixed-base system, the raw material costs are significantly optimized. Furthermore, the enhanced stability of the intermediate means that the reaction is far less prone to batch failures, a common hidden cost in fine chemical manufacturing. The simplified post-treatment process, which involves standard aqueous quenching and crystallization rather than complex chromatographic separations, drastically reduces the consumption of solvents and silica gel, leading to a lower environmental footprint and reduced disposal costs.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by its ability to streamline the synthesis workflow. Traditional methods often require prolonged reaction times and the careful, timed addition of sensitive reagents, which demands high labor input and specialized equipment monitoring. In contrast, this mixed-base catalyzed process shortens the reaction timeline and operates under milder conditions, reducing energy consumption for heating and cooling. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which in turn reduces the number of recrystallization steps required to meet stringent purity specifications. This cumulative effect results in a leaner, more cost-effective manufacturing process that improves the overall margin for high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method translates directly into improved delivery reliability. The use of common, commercially available solvents like dichloromethane and toluene, along with standard bases like triethylamine, mitigates the risk of raw material shortages that can plague projects relying on specialty chemicals. Moreover, the tolerance of the process to slight variations in conditions, thanks to the protective nature of the mixed base, ensures consistent batch-to-batch quality. This consistency is vital for maintaining long-term contracts with pharmaceutical clients who require uninterrupted supply of critical intermediates for their own API production schedules, thereby strengthening the partnership between the supplier and the end-user.
- Scalability and Environmental Compliance: The design of this reaction inherently supports scalability, a key requirement for moving from laboratory grams to multi-ton commercial production. The avoidance of extremely volatile reagents and the use of a quenching protocol that safely neutralizes Lewis acids make the process safer to operate on a large scale. Additionally, the reduction in byproduct generation aligns with increasingly strict global environmental regulations regarding chemical waste. By producing a cleaner crude product, the facility can minimize the load on wastewater treatment systems and reduce the volume of hazardous solid waste, ensuring compliance with green chemistry principles and enhancing the corporate sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enolization and ethylidene technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what potential partners can expect when integrating this method into their supply chain. Understanding these details helps in assessing the feasibility of adopting this route for specific project needs.
Q: How does the mixed base system improve the stability of the enolization intermediate?
A: The patented mixed base system, typically comprising triethylamine and 2-methylmorpholine, effectively protects the intermediate enolate from decomposition by strong acids or bases. This stabilization prevents the destruction of the enolization product during the subsequent Lewis acid catalysis, ensuring high reaction selectivity and yield.
Q: What are the advantages of this method for synthesizing Obeticholic acid intermediates?
A: This method allows for the regio-specific ethylidenation of 3α-hydroxy-7-keto-5β-cholanic acid under mild conditions. It avoids the use of harsh reagents that degrade sensitive bile acid structures, resulting in fewer byproducts and simplified purification processes essential for high-purity API intermediates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial application. It utilizes common organic solvents like dichloromethane or toluene and avoids the need for continuous material supplementation at high temperatures, making the operation simple, safe, and highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Ethylene-3α-Hydroxy-7-Keto-5β-Cholanic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex bile acid derivatives like 6-ethylene-3α-hydroxy-7-keto-5β-cholanic acid requires not just chemical expertise but a deep commitment to quality and scalability. Our technical team has extensively analyzed the pathways described in patent CN114181274B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this advanced chemistry to life. We understand that achieving the high purity required for Obeticholic acid intermediates demands rigorous process control, which is why our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities that could affect downstream drug efficacy.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge synthesis technology for your next project. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, ensuring that you maximize the economic benefits of this efficient route. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our mastery of this enolization and ethylidene combined reaction can accelerate your development timeline and secure your supply chain for the future.
