Advanced One-Pot Synthesis of High-Performance Fluoroboron Fluorescent Dyes for Commercial Scale-Up
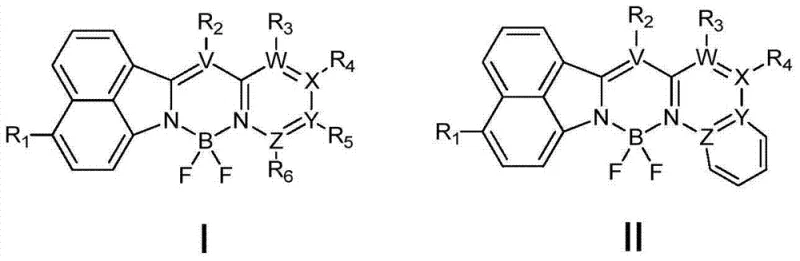
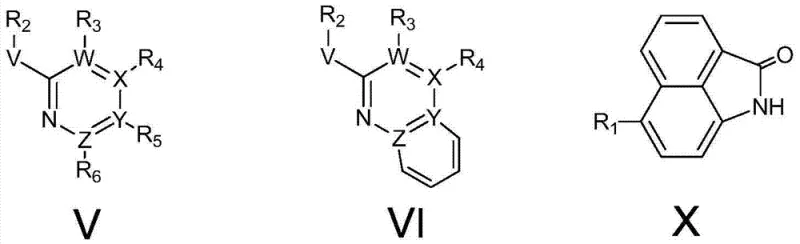
The landscape of fluorescent labeling and bioimaging is undergoing a significant transformation driven by the innovations disclosed in patent CN103865290A. This pivotal intellectual property introduces a novel class of fluoroboron fluorescent dyes that overcome the historical limitations of traditional BODIPY (boron-dipyrromethene) molecules, specifically addressing issues related to small Stokes shifts and fluorescence quenching. The core breakthrough lies in the unique molecular architecture defined by Formula (I) and Formula (II), which integrates a naphthalimide scaffold with a pyridine-based heterocyclic system coordinated to a boron fluoride center. This structural modification not only enhances photophysical stability but also extends the emission wavelength into the 518-600nm range, making these compounds exceptionally suitable for deep-tissue imaging and high-contrast diagnostic assays. For R&D directors seeking next-generation probes, this technology represents a critical leap forward in achieving high quantum yields without compromising chemical robustness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance fluorescent dyes has been plagued by inefficient multi-step protocols that hinder commercial viability. Traditional methods for constructing BODIPY analogs often require the preparation of delicate dipyrromethene intermediates under strictly anhydrous conditions, followed by a separate complexation step with boron trifluoride. These processes are not only labor-intensive but also suffer from low overall yields due to the instability of the intermediate species, which are prone to oxidation and hydrolysis. Furthermore, conventional routes frequently rely on expensive transition metal catalysts or exotic starting materials that are difficult to source in bulk quantities, creating bottlenecks for supply chain managers aiming for consistent production. The cumulative effect of these inefficiencies is a high cost of goods sold (COGS) and extended lead times, which makes scaling these materials for industrial bioanalysis applications economically challenging.
The Novel Approach
In stark contrast, the methodology outlined in CN103865290A employs a streamlined "one-pot" strategy that consolidates the condensation and complexation reactions into a single vessel operation. By utilizing readily available starting materials such as 1,8-naphthalimide derivatives and substituted aminopyridines, the process bypasses the need for isolating unstable intermediates. The reaction proceeds efficiently in common organic solvents like toluene under heating conditions of 100-130°C, utilizing accessible Lewis acids such as titanium tetrachloride or phosphorus oxychloride. This simplification drastically reduces the operational complexity and solvent waste associated with traditional purification steps. For procurement teams, this translates to a more resilient supply chain where raw material availability is guaranteed by the commodity status of the reagents, thereby ensuring continuity of supply for large-scale manufacturing campaigns.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The chemical elegance of this synthesis lies in the precise orchestration of Lewis acid catalysis to drive the formation of the rigid fluoroboron core. The mechanism initiates with the activation of the carbonyl group on the naphthalimide precursor by the Lewis acid, which increases its electrophilicity and facilitates nucleophilic attack by the amino group of the pyridine derivative. This condensation step is critical for establishing the conjugated pi-system responsible for the dye's fluorescence properties. Following the initial bond formation, the addition of boron trifluoride diethyl etherate serves to lock the molecular conformation through coordination with the nitrogen atoms, creating the characteristic BF2 chelate ring. This rigidification is essential for minimizing non-radiative decay pathways, thereby maximizing the fluorescence quantum yield. Understanding this mechanistic pathway allows process chemists to fine-tune reaction parameters, such as the molar ratio of Lewis acid to substrate (optimized at 2-8mol), to suppress side reactions and ensure high purity profiles.
Impurity control is another vital aspect of this mechanism, particularly for applications requiring stringent regulatory compliance. The planar nature of the resulting fluoroboron complexes, as evidenced by X-ray diffraction data in the patent, contributes to their exceptional photostability and resistance to aggregation-caused quenching. By maintaining the reaction temperature within the optimal 100-120°C window and employing acid-binding agents like triethylamine, the process effectively neutralizes acidic byproducts that could otherwise degrade the sensitive fluorophore structure. This results in a crude product with a significantly cleaner impurity profile, reducing the burden on downstream purification processes such as silica gel column chromatography. For quality assurance teams, this inherent process robustness means that batch-to-batch variability is minimized, ensuring that the final high-purity fluorescent dye meets the rigorous specifications demanded by the pharmaceutical and diagnostic industries.
How to Synthesize Fluoroboron Fluorescent Dyes Efficiently
The synthesis protocol described in the patent offers a reproducible framework for generating these advanced materials, balancing reaction kinetics with practical operability. The process begins by dissolving the naphthalimide and aminopyridine reactants in toluene, followed by the controlled addition of the Lewis acid catalyst under reflux conditions. After a reaction period of 8-12 hours to ensure complete conversion, the boron trifluoride complexing agent is introduced to finalize the dye structure. While the patent provides specific experimental examples yielding between 16% and 45% depending on the specific substituents, the general procedure is highly adaptable to various derivative targets. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining safety and efficiency.
- Condense 1,8-naphthalimide derivatives with aminopyridine reactants in toluene using a Lewis acid catalyst like TiCl4 at 100-120°C.
- Add an acid-binding agent such as triethylamine to neutralize byproducts and drive the equilibrium forward.
- Introduce boron trifluoride diethyl etherate to the reaction mixture to form the final stable fluoroboron complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers compelling advantages that directly address the pain points of cost and scalability in fine chemical manufacturing. The elimination of intermediate isolation steps inherently reduces the consumption of solvents and energy, leading to substantial operational expenditure savings. Moreover, the reliance on commodity-grade reagents rather than specialized custom synthons mitigates the risk of raw material price volatility. For supply chain heads, the simplicity of the one-pot process means that existing reactor infrastructure can be utilized without the need for significant capital investment in new equipment. This flexibility allows for rapid response to market demand fluctuations, ensuring that customers receive their orders with reduced lead times and greater reliability.
- Cost Reduction in Manufacturing: The streamlined one-pot methodology significantly lowers production costs by removing unit operations associated with intermediate workups and drying. By avoiding the use of precious metal catalysts in favor of inexpensive Lewis acids like TiCl4, the direct material costs are drastically optimized. Furthermore, the higher atom economy of the condensation reaction reduces waste disposal costs, contributing to a leaner and more profitable manufacturing model. These efficiencies compound at scale, making the commercial production of these high-value dyes economically viable for broad market adoption.
- Enhanced Supply Chain Reliability: The sourcing strategy for this synthesis is bolstered by the use of widely available industrial chemicals such as 1,8-naphthalimide and various aminopyridines. Unlike niche precursors that may have single-source suppliers, these building blocks are produced by multiple global vendors, ensuring a diversified and secure supply base. This redundancy protects against supply disruptions caused by geopolitical events or production outages at specific facilities. Consequently, manufacturers can guarantee consistent delivery schedules to their clients, fostering long-term partnerships based on trust and dependability.
- Scalability and Environmental Compliance: The robustness of the reaction conditions allows for seamless scale-up from laboratory benchtop to multi-ton annual production capacities. The process operates at moderate temperatures and pressures, reducing the safety risks associated with high-energy chemical transformations. Additionally, the reduced solvent usage and simplified waste stream facilitate easier compliance with increasingly stringent environmental regulations. This sustainability profile is increasingly important for corporate social responsibility goals and helps customers meet their own green chemistry mandates without compromising on performance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this fluoroboron dye technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the fit of this technology within their existing product portfolios and research pipelines.
Q: What are the primary advantages of this one-pot synthesis method over traditional BODIPY production?
A: The patented one-pot method eliminates the need for isolating unstable intermediates, significantly reducing processing time and solvent consumption compared to multi-step traditional routes.
Q: How does the Stokes shift of these new dyes compare to conventional fluorophores?
A: These novel fluoroboron dyes exhibit a significantly larger Stokes shift (up to 3964 cm-1), which minimizes self-quenching and background interference in sensitive bioimaging applications.
Q: Are the raw materials for this synthesis readily available for large-scale procurement?
A: Yes, the process utilizes commodity chemicals such as 1,8-naphthalimide, various aminopyridines, and standard Lewis acids, ensuring a robust and continuous supply chain for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroboron Fluorescent Dye Supplier
As the demand for advanced bioimaging tools continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver materials with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the photophysical properties of every batch, ensuring that the fluorescence quantum yield and Stokes shift performance match the theoretical potential of the patent. We are committed to being a strategic partner that bridges the gap between innovative academic research and industrial reality.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to accelerate your development timelines with confidence in a secure and high-quality supply chain.
