Advanced Synthesis and Commercial Scalability of Potent Nitroimidazole-Oxadiazole Hybrids
Advanced Synthesis and Commercial Scalability of Potent Nitroimidazole-Oxadiazole Hybrids
The escalating crisis of bacterial resistance to conventional antibiotics necessitates the urgent development of novel chemical entities with distinct mechanisms of action. Patent CN103360370A discloses a groundbreaking class of nitroimidazole derivatives modified with an oxadiazole ring, demonstrating significant inhibitory effects against both Gram-positive and Gram-negative bacteria. This technological breakthrough addresses the critical limitation of existing nitroimidazole drugs, which are often restricted to anaerobic infections and suffer from notable adverse reactions. By strategically fusing the bioactive oxadiazole moiety to the nitroimidazole core, this innovation offers a promising pathway for creating next-generation antibacterial agents with improved safety profiles and broader spectra of activity.
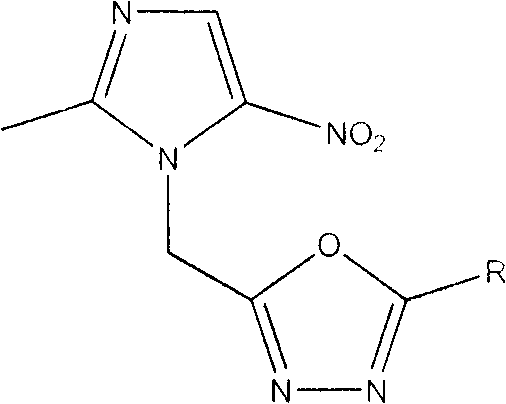
For pharmaceutical R&D directors and procurement specialists, understanding the structural versatility of these compounds is paramount. The general formula reveals a robust scaffold where the R group can be systematically varied to optimize pharmacokinetic properties and potency. The patent details a library of substituents ranging from halogens like fluorine and bromine to electron-donating groups like methoxy and methyl. This modularity allows for precise tuning of the molecule's lipophilicity and electronic characteristics, which are crucial factors in penetrating bacterial cell walls and evading efflux pumps. As a reliable supplier of pharmaceutical intermediates, recognizing the synthetic accessibility of these diverse analogs is key to securing a stable supply chain for future drug development projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying nitroimidazole often involve direct substitution on the imidazole ring, which can be chemically challenging due to the electron-deficient nature of the heterocycle. Furthermore, many existing nitroimidazole-based therapies are plagued by dose-limiting toxicities, including neurological and gastrointestinal side effects, which restrict their long-term use. Conventional synthesis routes for similar hybrids frequently rely on harsh reaction conditions, expensive coupling reagents, or multi-step protection-deprotection sequences that drastically increase the cost of goods sold (COGS). Additionally, purification often requires preparative HPLC or column chromatography, which creates significant solvent waste and bottlenecks in commercial scale-up, making these processes economically unviable for mass production of generic or novel antibiotics.
The Novel Approach
The methodology presented in the patent circumvents these issues through a streamlined three-step sequence that utilizes readily available starting materials. The strategy employs a nucleophilic substitution to attach an acetate linker, followed by hydrazinolysis to generate a reactive hydrazide intermediate. The final cyclization with substituted benzoic acids using phosphorus oxychloride is highly efficient, yielding the target oxadiazole ring in a single pot. This approach eliminates the need for precious metal catalysts and simplifies the workup procedure to basic filtration and recrystallization. By avoiding complex purification techniques, the process significantly reduces environmental impact and operational costs, aligning perfectly with the goals of cost reduction in pharmaceutical intermediates manufacturing while ensuring high chemical purity.
Mechanistic Insights into Phosphorus Oxychloride-Mediated Cyclization
The core of this synthetic innovation lies in the formation of the 1,3,4-oxadiazole ring, a bioisostere known for its metabolic stability and ability to enhance membrane permeability. The mechanism involves the activation of the carboxylic acid group of the substituted benzoic acid by phosphorus oxychloride (POCl3), generating a highly reactive acyl chloride or mixed anhydride species in situ. This electrophilic intermediate then undergoes nucleophilic attack by the terminal amino group of the nitroimidazole-hydrazide. Subsequent intramolecular cyclization and dehydration drive the formation of the five-membered oxadiazole ring. This transformation is thermodynamically favorable and proceeds cleanly at elevated temperatures, typically around 100°C, ensuring high conversion rates without the formation of complex byproduct mixtures that are difficult to separate.
From an impurity control perspective, this mechanism is advantageous because the byproducts are primarily inorganic salts or volatile phosphorous species that can be easily removed during the aqueous workup. The use of saturated sodium bicarbonate solution to quench the reaction effectively neutralizes excess acid and decomposes residual POCl3, preventing downstream corrosion or contamination. The final recrystallization from absolute ethanol further enhances the purity profile by excluding unreacted starting materials and isomeric impurities. For quality control teams, this predictable impurity profile simplifies the validation process and ensures that the final active pharmaceutical ingredient (API) intermediate meets stringent regulatory specifications for heavy metals and residual solvents.
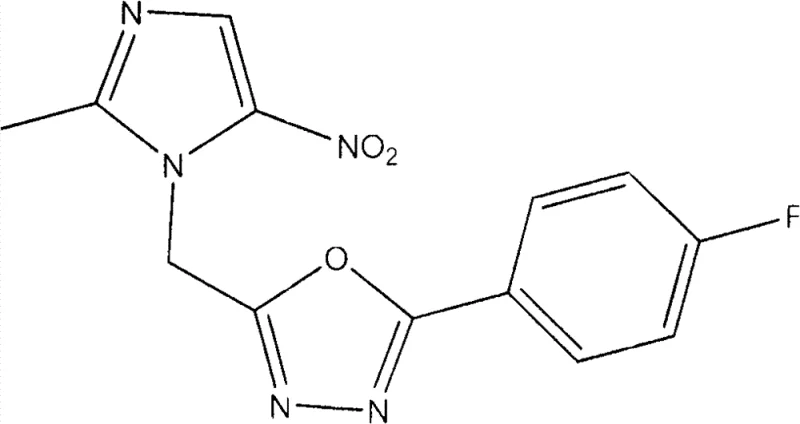
How to Synthesize 2-(4-Fluorophenyl)-5-((2-Methyl-5-Nitro-1H-Imidazol-1-Yl)Methyl)-1,3,4-Oxadiazole Efficiently
To illustrate the practical application of this technology, we examine the synthesis of Compound 1, a fluorinated derivative that serves as a representative example of the library. This specific compound is synthesized by reacting the nitroimidazole hydrazide intermediate with 4-fluorobenzoic acid under the optimized cyclization conditions. The choice of the para-fluoro substituent is strategic, as fluorine atoms often improve the metabolic stability and binding affinity of drug candidates. The following guide outlines the standardized operational parameters derived directly from the patent examples, ensuring reproducibility and safety during pilot plant operations.
- Perform N-alkylation of 2-methyl-5-nitroimidazole with ethyl chloroacetate using potassium carbonate in acetone at 50°C.
- Conduct hydrazinolysis of the ester intermediate with hydrazine hydrate in methanol at 70°C to form the hydrazide.
- Execute cyclodehydration using phosphorus oxychloride and substituted benzoic acid at 100°C to close the oxadiazole ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic viability of a new chemical entity is just as critical as its biological activity. The synthesis route described in CN103360370A offers substantial commercial advantages rooted in the simplicity of its unit operations and the commodity status of its raw materials. The primary starting materials, such as 2-methyl-5-nitroimidazole, ethyl chloroacetate, and various substituted benzoic acids, are bulk chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This abundance ensures price stability and consistent availability, which are essential for maintaining uninterrupted production schedules in a competitive market environment.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need for expensive transition metal catalysts or specialized ligands that often drive up the price of advanced intermediates. Furthermore, the reliance on recrystallization as the sole purification method drastically reduces solvent consumption and waste disposal costs compared to chromatographic techniques. The high yields reported in the patent examples, ranging from 51% to 62% across different derivatives, indicate a robust process with minimal material loss, directly contributing to a lower cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The synthetic pathway utilizes standard reaction conditions that do not require cryogenic temperatures or high-pressure equipment, allowing for execution in multipurpose chemical reactors commonly found in CDMO facilities. The reaction times are reasonable, with the longest step requiring approximately 50 hours but occurring at a mild 50°C, which facilitates flexible scheduling and batch planning. This operational flexibility ensures that production can be scaled up or down rapidly in response to fluctuating market demand without requiring significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process is favorable because it avoids the generation of persistent organic pollutants or toxic heavy metal waste. The solvents used, primarily acetone, methanol, and ethanol, are Class 3 solvents with low toxicity and can be easily recovered and recycled through distillation. This alignment with green chemistry principles not only reduces the environmental footprint but also simplifies regulatory compliance and permitting processes for manufacturing sites, thereby reducing lead time for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these nitroimidazole derivatives. These insights are derived from a detailed analysis of the patent specifications and are intended to assist decision-makers in evaluating the feasibility of integrating these intermediates into their development pipelines. Understanding these nuances is vital for establishing a successful partnership with a chemical supplier.
Q: What is the primary advantage of introducing an oxadiazole ring to the nitroimidazole scaffold?
A: The introduction of the oxadiazole ring serves as a prodrug modification strategy that can potentially reduce the adverse side effects associated with traditional nitroimidazoles, such as gastrointestinal irritation, while maintaining or enhancing antibacterial potency against both Gram-positive and Gram-negative strains.
Q: Which substituents on the phenyl ring demonstrated the highest antibacterial activity in the patent data?
A: According to the experimental data in patent CN103360370A, derivatives containing methoxy groups (specifically ortho- and meta-methoxy) and methyl groups exhibited superior minimum inhibitory concentrations (MIC) compared to halogenated or nitro-substituted analogs, particularly against E. coli and B. subtilis.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like acetone and methanol and avoids expensive transition metal catalysts. The purification relies on recrystallization rather than complex chromatography, making it highly amenable to scale-up from kilogram to multi-ton production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitroimidazole Derivatives Supplier
As the global demand for effective antibacterial agents continues to rise, the ability to reliably source high-quality intermediates is a strategic imperative. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of nitroimidazole chemistry, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that consistency is key in drug development, and our robust quality management systems guarantee that the material you receive is identical to the clinical grade standards required for regulatory filings.
We invite you to collaborate with us to optimize your supply chain for these critical antibacterial intermediates. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to deep process knowledge that can accelerate your timeline from bench to market. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with confidence and efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
