Advanced Three-Step Synthesis of Alpha-Glucosidase Inhibitor Intermediates for Commercial Scale
Advanced Three-Step Synthesis of Alpha-Glucosidase Inhibitor Intermediates for Commercial Scale
The global pharmaceutical landscape is increasingly demanding efficient, green, and scalable synthetic routes for complex heterocyclic intermediates, particularly those targeting metabolic disorders such as diabetes. Patent CN113831330A introduces a groundbreaking three-step methodology for the synthesis of 3-(2-thiophene-2-methylene)hydrazinoquinoxaline-2-one, a potent alpha-glucosidase inhibitor. This novel approach leverages quinoxaline-2-one as a foundational building block, subjecting it to a sequence of oxidation, hydrazinolysis, and condensation reactions. For R&D directors and procurement specialists, this patent represents a significant shift away from traditional, hazard-heavy synthesis towards a more sustainable and economically viable manufacturing paradigm. The strategic implementation of ammonium persulfate as an oxidant and water-miscible solvent systems underscores a commitment to reducing environmental impact while maintaining high chemical fidelity. As we analyze the technical nuances of this disclosure, it becomes evident that this route offers substantial advantages in terms of operational simplicity and raw material accessibility, positioning it as a prime candidate for commercial scale-up in the competitive API intermediate market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex quinoxaline derivatives bearing hydrazino and thiophene moieties has been plagued by synthetic inefficiencies and environmental concerns. Traditional pathways often rely on the use of stoichiometric amounts of toxic heavy metal oxidants or harsh halogenated solvents, which not only escalate the cost of goods sold (COGS) due to expensive waste treatment protocols but also introduce significant safety risks in a manufacturing environment. Furthermore, multi-step sequences involving protecting group strategies or cryogenic conditions frequently result in cumulative yield losses and extended production cycles, creating bottlenecks in the supply chain. The reliance on specialized reagents that are not commodity chemicals further exacerbates supply volatility, making it difficult for procurement managers to secure long-term contracts at stable prices. These legacy methods often fail to meet the rigorous standards of modern green chemistry, leading to regulatory hurdles and increased scrutiny from environmental agencies, which can delay time-to-market for critical antidiabetic therapies.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113831330A offers a streamlined, three-step trajectory that elegantly bypasses these historical impediments. By initiating the synthesis with the direct oxidation of quinoxaline-2-one using ammonium persulfate, the process eliminates the need for transition metal catalysts, thereby simplifying the downstream purification landscape and removing the risk of heavy metal contamination in the final API. The subsequent hydrazinolysis and condensation steps are conducted in benign solvent systems such as water and ethanol, which are not only cost-effective but also significantly reduce the ecological footprint of the manufacturing process. This approach ensures that the reaction conditions remain mild, typically operating between 60°C and 100°C, which minimizes energy consumption and enhances operational safety. The robustness of this synthetic design allows for a more predictable production schedule, directly addressing the supply chain head's need for reliability and continuity in the sourcing of high-purity pharmaceutical intermediates.
Mechanistic Insights into Oxidative Functionalization and Hydrazone Formation
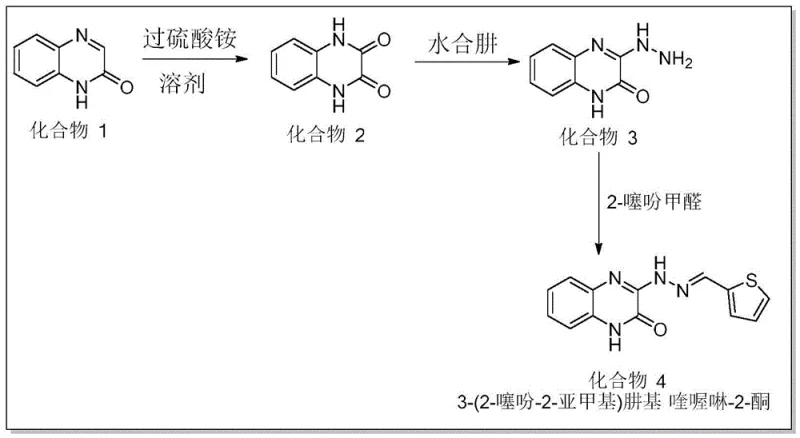
The core of this synthetic innovation lies in the initial oxidative transformation, where quinoxaline-2-one is converted into a reactive dione intermediate via a radical-mediated mechanism facilitated by ammonium persulfate. In this critical first step, the persulfate anion decomposes thermally to generate sulfate radical anions, which abstract hydrogen atoms from the substrate to initiate the oxidation cascade. This metal-free oxidation is particularly advantageous for pharmaceutical applications as it precludes the formation of organometallic impurities that are notoriously difficult to remove to ppm levels. The reaction proceeds efficiently in a mixed solvent system of acetonitrile and water, where the polar environment stabilizes the charged intermediates and facilitates the dissolution of the inorganic oxidant. Following this, the hydrazinolysis step involves the nucleophilic attack of hydrazine hydrate on the electrophilic carbonyl carbon, resulting in the formation of a hydrazone linkage with the concomitant release of water. This transformation is highly selective and proceeds under reflux conditions in pure water, demonstrating the remarkable chemoselectivity of the process which prevents side reactions on the sensitive heterocyclic core.
The final condensation step serves to install the thiophene moiety, a crucial pharmacophore for alpha-glucosidase inhibitory activity, through a classic Schiff base formation mechanism. The reaction between the hydrazino-quinoxaline intermediate and 2-thiophenecarboxaldehyde is driven by the removal of water, typically facilitated by azeotropic distillation or the use of molecular sieves, although the patent describes a simple reflux in ethanol which suffices for high conversion. The resulting azomethine bond (C=N) is stabilized by conjugation with both the quinoxaline and thiophene rings, enhancing the thermal and chemical stability of the final drug molecule. From an impurity control perspective, the linearity of this three-step sequence minimizes the generation of complex byproducts, allowing for straightforward purification via crystallization or standard column chromatography. This mechanistic clarity provides R&D teams with a clear roadmap for process optimization, ensuring that critical quality attributes (CQAs) such as purity and particle size distribution can be tightly controlled throughout the manufacturing lifecycle.
How to Synthesize 3-(2-thiophene-2-methylene)hydrazinoquinoxaline-2-one Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity profiles, particularly during the oxidation and condensation phases. The protocol outlined in the patent provides a robust framework for laboratory-scale preparation, which serves as a foundation for scaling up to pilot and commercial volumes. Operators must ensure strict adherence to temperature profiles, maintaining the oxidation step at approximately 60°C to prevent over-oxidation or decomposition of the persulfate reagent. Similarly, the reflux conditions in the subsequent steps must be monitored to ensure complete conversion without degrading the sensitive hydrazino functionality. For detailed operational procedures, including specific workup techniques and purification protocols, please refer to the standardized synthesis guide below which encapsulates the critical process parameters derived from the patent examples.
- Oxidation of quinoxaline-2-one using ammonium persulfate in acetonitrile/water at 60°C to form the dione intermediate.
- Hydrazinolysis of the intermediate with hydrazine hydrate in water under reflux at 100°C to generate the hydrazino derivative.
- Condensation of the hydrazino derivative with 2-thiophenecarboxaldehyde in ethanol under reflux at 80°C to yield the final drug molecule.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible economic and logistical benefits that extend far beyond the laboratory bench. The elimination of expensive transition metal catalysts and the substitution of hazardous chlorinated solvents with water and ethanol drastically reduces the raw material costs and waste disposal fees associated with production. This shift not only lowers the direct manufacturing expenses but also mitigates the regulatory risks associated with handling and transporting dangerous chemicals, thereby streamlining the logistics of the supply chain. Furthermore, the use of commodity chemicals like ammonium persulfate and hydrazine hydrate ensures a stable and diversified supplier base, reducing the vulnerability of the production schedule to single-source bottlenecks or geopolitical supply disruptions. The inherent simplicity of the three-step process also implies a shorter cycle time, enabling manufacturers to respond more agilely to fluctuations in market demand for antidiabetic intermediates.
- Cost Reduction in Manufacturing: The strategic replacement of noble metal catalysts with ammonium persulfate represents a fundamental shift in cost structure, removing the need for costly metal scavenging resins and extensive purification steps required to meet residual metal specifications. By utilizing water as a primary solvent in the hydrazinolysis step, the process significantly cuts down on the volume of organic solvents required, leading to substantial savings in solvent purchase, recovery, and incineration costs. Additionally, the mild reaction temperatures reduce the energy load on the manufacturing facility, contributing to lower utility bills and a smaller carbon footprint. These cumulative efficiencies result in a significantly reduced cost of goods sold, allowing for more competitive pricing strategies in the global API market without compromising on quality margins.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade starting materials such as quinoxaline-2-one and 2-thiophenecarboxaldehyde ensures that the supply chain is resilient against shortages that often plague specialty reagents. Since the reagents are not proprietary or sourced from limited geographic regions, procurement teams can negotiate better terms and secure long-term supply agreements with multiple vendors. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, further stabilizing the production output. This reliability is critical for maintaining continuous manufacturing operations and meeting the just-in-time delivery requirements of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The green chemistry credentials of this process, characterized by the use of benign solvents and the absence of heavy metals, facilitate a smoother regulatory approval process for new drug filings. The simplified waste stream, primarily consisting of aqueous salts and recoverable alcohols, is easier to treat and dispose of in compliance with stringent environmental regulations. This ease of waste management lowers the barrier for scaling up from kilogram to tonne quantities, as the engineering controls required are less complex than those for hazardous processes. Consequently, the pathway supports rapid commercial scale-up, enabling the swift transition from clinical trial material to full-scale commercial production to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this quinoxaline derivative, based on the detailed disclosures within the patent literature. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their existing drug development pipelines. The answers provided reflect the consensus on the chemical behavior and process advantages observed during the validation of this novel synthetic route.
Q: What is the primary therapeutic application of 3-(2-thiophene-2-methylene)hydrazinoquinoxaline-2-one?
A: This compound acts as a potent alpha-glucosidase inhibitor, making it a valuable intermediate for developing antidiabetic medications that regulate postprandial blood glucose levels.
Q: How does this synthesis method improve upon traditional manufacturing processes?
A: The method utilizes ammonium persulfate as a benign oxidant and water-based solvent systems, eliminating the need for toxic heavy metal catalysts and hazardous organic solvents, thereby aligning with green chemistry principles.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (40-150°C) and utilize readily available raw materials like quinoxaline-2-one and hydrazine hydrate, ensuring high scalability and supply chain stability for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2-thiophene-2-methylene)hydrazinoquinoxaline-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate complex patent methodologies into robust, commercial-grade manufacturing processes. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to green chemistry aligns perfectly with the sustainable synthesis route described in CN113831330A, allowing us to offer a product that is not only chemically superior but also environmentally responsible.
We invite potential partners to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized process can reduce your overall project costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for this vital alpha-glucosidase inhibitor intermediate is secure, compliant, and cost-effective for the long term.
