Advanced One-Step Synthesis of Dicyclopropylmethylamine for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with operational safety, particularly for critical antihypertensive intermediates. Patent CN102351710B introduces a transformative preparation method for dicyclopropylmethylamine, a key building block in the synthesis of rilmenidine. This technical disclosure marks a significant departure from legacy methodologies by replacing hazardous metal reductions with a streamlined reductive amination protocol using ammonium formate. For R&D directors and procurement strategists, this innovation represents a pivotal opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. The patent details a direct conversion of dicyclopropyl ketone to the target amine, bypassing the unstable oxime intermediates typical of older routes. By leveraging this technology, manufacturers can achieve substantial improvements in both process safety and total yield, addressing the growing demand for reliable pharmaceutical intermediate suppliers who can deliver consistent quality without compromising on environmental or safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of dicyclopropylmethylamine relied on a cumbersome two-step sequence that introduced significant inefficiencies and safety liabilities into the manufacturing workflow. The traditional pathway begins with the condensation of dicyclopropyl ketone with hydroxylamine to form an oxime derivative, followed by a rigorous reduction step utilizing metallic sodium in an ethanol solvent system. This reliance on alkali metals creates severe operational challenges, including the necessity for strictly anhydrous conditions and the management of pyrophoric materials that pose inherent fire and explosion risks. Furthermore, the multi-step nature of this conventional synthesis inherently compounds yield losses at each stage, resulting in a dismal total reaction yield of approximately 25%, which is economically unsustainable for modern cost-sensitive API manufacturing. The generation of hazardous waste streams associated with quenching excess sodium and disposing of solvent residues further complicates the environmental compliance profile of this outdated method.
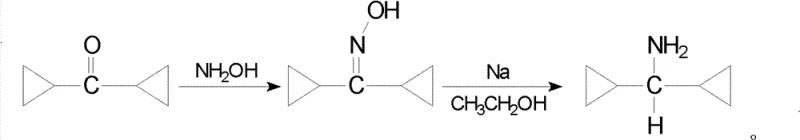
The Novel Approach
In stark contrast to the perilous conventional routes, the novel approach disclosed in the patent utilizes a direct reductive amination strategy driven by ammonium formate as a safe and effective hydrogen donor. This methodology collapses the synthesis into a single operational unit where dicyclopropyl ketone reacts directly with ammonium formate under reflux conditions, effectively eliminating the isolation of the oxime intermediate. The reaction proceeds smoothly at temperatures between 165-170°C, generating carbon dioxide, ammonia, and water as benign byproducts that are easily managed through standard separation techniques. This simplification not only removes the need for dangerous metallic sodium but also dramatically shortens the production cycle, allowing for a total yield improvement that exceeds 80% in optimized examples. The transition to this chemistry exemplifies a strategic shift towards greener synthesis principles, offering a viable solution for cost reduction in pharmaceutical intermediate manufacturing while ensuring a safer working environment for plant personnel.
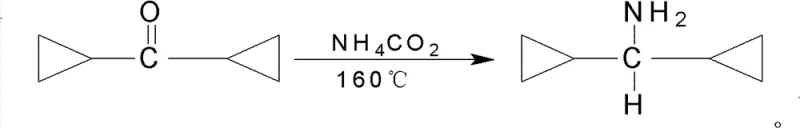
Mechanistic Insights into Ammonium Formate Mediated Reductive Amination
The core chemical transformation driving this process is a variation of the Leuckart-Wallach reaction, where the carbonyl group of the dicyclopropyl ketone undergoes nucleophilic attack by ammonia generated in situ from the thermal decomposition of ammonium formate. This initial condensation forms an imine intermediate, which is subsequently reduced by the formate ion acting as a hydride source. The thermal energy provided at 165-170°C facilitates the decarboxylation of the formate species, releasing the necessary reducing equivalents to convert the C=N double bond into the stable C-N single bond of the primary amine. Understanding this mechanism is crucial for R&D teams aiming to replicate the process, as the continuous removal of water via azeotropic distillation is essential to drive the equilibrium towards the imine formation, preventing the hydrolysis of the intermediate back to the starting ketone. The precise control of temperature ensures that the decomposition of ammonium formate occurs at a rate that matches the reduction kinetics, minimizing the formation of secondary amine byproducts or N-formyl derivatives that could complicate downstream purification.
Impurity control in this synthesis is achieved through a sophisticated workup procedure that leverages the basicity of the target amine to separate it from neutral organic impurities and acidic byproducts. Following the reaction, the crude mixture is subjected to a series of pH-adjusted extractions, initially washing with water to remove unreacted ammonium salts and then acidifying to pH 4-5 to convert the amine into its water-soluble hydrochloride salt. This acid extraction step effectively leaves neutral organic impurities in the organic phase, which is discarded, thereby purifying the amine salt in the aqueous layer. Subsequent basification to pH 10 regenerates the free amine, which is then extracted into dichloromethane, dried, and distilled under reduced pressure. This rigorous purification protocol ensures that the final dicyclopropylmethylamine meets stringent purity specifications of 98-99.5%, making it suitable for direct use in the synthesis of sensitive pharmaceutical active ingredients without requiring further chromatographic purification.
How to Synthesize Dicyclopropylmethylamine Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the reagents and the efficiency of the water separation apparatus to maximize conversion. The patent specifies a molar ratio of ammonium formate to ketone ranging from 2:1 to 4:1, with a preferred ratio of 2.5:1 to ensure complete consumption of the valuable ketone starting material. Operators must utilize a reactor equipped with a Dean-Stark trap or similar water separator to continuously remove the water generated during the reaction, as the presence of water can inhibit the formation of the critical imine intermediate and lower the overall yield. The detailed standardized synthesis steps below outline the precise heating profiles, workup sequences, and distillation parameters required to achieve the reported high yields and purity levels consistently.
- Mix dicyclopropyl ketone with ammonium formate (2-4 molar equivalents) in a reactor equipped with a water separator and condenser.
- Heat the mixture to 165-170°C under normal pressure, maintaining reflux for 10-12 hours while continuously removing generated water.
- Cool the reaction, wash with water, adjust pH to 4-5 with acid, extract impurities, basify to pH 10, extract product with dichloromethane, and distill under reduced pressure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ammonium formate-based synthesis offers profound logistical and economic benefits that extend far beyond simple yield improvements. The elimination of metallic sodium from the process inventory removes a major bottleneck related to the storage, handling, and disposal of hazardous chemicals, thereby reducing insurance costs and regulatory burdens associated with dangerous goods. Additionally, the use of ammonium formate, a commodity chemical with widespread availability, mitigates supply chain risks associated with specialized reagents, ensuring a more resilient production schedule that is less susceptible to market volatility. The simplification of the process from two distinct reaction stages to a single pot operation also translates to reduced equipment occupancy time and lower utility consumption, contributing to significant operational expenditure savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic reduction in raw material costs and waste treatment expenses. By avoiding the use of expensive and hazardous metallic sodium, manufacturers eliminate the need for specialized quenching protocols and the associated costs of neutralizing reactive metal waste. Furthermore, the substantial increase in yield from roughly 25% to over 80% means that significantly less starting ketone is required to produce the same amount of final product, directly lowering the cost of goods sold. The simplified workup also reduces solvent consumption and labor hours, creating a leaner manufacturing process that enhances overall profit margins for high-volume production runs.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method ensures consistent output and shorter lead times for high-purity pharmaceutical intermediates. The reliance on stable, non-hazardous reagents like ammonium formate allows for uninterrupted production cycles without the safety shutdowns often necessitated by the handling of pyrophoric materials. This stability is critical for maintaining just-in-time delivery schedules for downstream API manufacturers, reducing the risk of stockouts that can disrupt the broader pharmaceutical supply network. The ability to source raw materials from multiple general chemical suppliers further diversifies the supply base, protecting against single-source failures.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards associated with sodium additions. The reaction operates under atmospheric pressure with manageable thermal profiles, making it compatible with standard glass-lined or stainless steel reactors used in fine chemical plants. Environmentally, the process generates benign byproducts such as carbon dioxide and water, simplifying effluent treatment and helping facilities meet increasingly strict environmental regulations. This ease of scale-up supports the commercialization of complex pharmaceutical intermediates without requiring massive capital investment in specialized safety infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process safety, yield expectations, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement specialists assessing the long-term viability of suppliers utilizing this technology.
Q: What are the safety advantages of the ammonium formate method over the traditional sodium reduction route?
A: The traditional method utilizes metallic sodium in ethanol, which poses significant fire and explosion risks due to the pyrophoric nature of sodium and hydrogen gas evolution. The novel ammonium formate method operates under standard atmospheric pressure without reactive metals, drastically reducing operational hazards and simplifying safety protocols for large-scale manufacturing.
Q: How does the new process impact the overall yield and purity of dicyclopropylmethylamine?
A: By eliminating the multi-step oxime formation and subsequent reduction, the new one-step reductive amination process significantly boosts total yield from approximately 25% in conventional routes to over 80%. Furthermore, the simplified workup involving pH-controlled extractions allows for the isolation of the final amine with purity levels reaching 98-99.5%.
Q: Is this synthesis route suitable for commercial scale-up of complex pharmaceutical intermediates?
A: Yes, the process is highly scalable because it avoids hazardous reagents like metallic sodium that require specialized handling equipment. The use of commercially available ammonium formate and standard reflux conditions (165-170°C) makes it ideal for cost reduction in API manufacturing and ensures consistent supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicyclopropylmethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the modern pharmaceutical landscape. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ammonium formate reduction method are translated into reliable industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch of dicyclopropylmethylamine against the highest industry standards. We understand that for antihypertensive drug manufacturers, the consistency of the intermediate is paramount, and our advanced manufacturing capabilities are designed to deliver exactly that level of precision and reliability.
We invite global partners to collaborate with us to leverage these technological advancements for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this safer, high-yield process. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your production of rilmenidine and related therapeutics remains competitive, compliant, and secure.
