Scalable One-Step Synthesis of Benzofuran β-Dehydrogenated Unnatural Amino Acids for Advanced Drug Discovery
Introduction to Patent CN108047177B: A Breakthrough in Unnatural Amino Acid Synthesis
The landscape of molecular biology and drug discovery is constantly evolving, driven by the need for novel building blocks that can expand the functional diversity of proteins. Patent CN108047177B, published in May 2021, introduces a transformative methodology for the one-step synthesis of benzofuran β-dehydrogenated unnatural amino acids. These specific compounds, characterized by a 2-amino and 3-carboxyl substitution pattern on the benzofuran core, represent a critical class of intermediates for probing protein structures and developing new therapeutic agents. Traditional approaches to constructing such complex heterocyclic scaffolds often involve multi-step sequences with harsh conditions, limiting their accessibility for high-throughput screening and commercial applications. This patent discloses a streamlined catalytic protocol that operates under mild conditions, utilizing inexpensive reagents to achieve high atom economy. For research directors and procurement specialists alike, this innovation signals a shift towards more sustainable and cost-effective manufacturing pathways for high-purity pharmaceutical intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of benzofuran derivatives has relied heavily on transition metal-catalyzed coupling reactions, such as those involving palladium or copper complexes. While effective for certain substrates, these conventional methodologies suffer from significant drawbacks that hinder their industrial viability. Firstly, the starting materials often require pre-functionalization, adding extra synthetic steps and increasing the overall cost of goods. Secondly, the reliance on precious metal catalysts introduces severe challenges in downstream processing, particularly regarding the removal of trace metal impurities to meet stringent regulatory standards for active pharmaceutical ingredients (APIs). Furthermore, many of these traditional reactions necessitate elevated temperatures and inert atmospheres, which escalate energy consumption and complicate reactor design. The limited substrate scope of these metal-catalyzed systems also restricts the diversity of accessible analogs, often failing to accommodate sensitive functional groups required for biological activity.
The Novel Approach
In stark contrast, the method described in CN108047177B offers a paradigm shift by employing a base-catalyzed cyclization strategy that bypasses the need for transition metals entirely. By reacting phenoxyamide compounds with bromopropiolates in an organic solvent environment, specifically ethyl acetate, the process achieves the formation of the benzofuran ring in a single operational step. This novel approach not only simplifies the synthetic route but also drastically reduces the environmental footprint by generating minimal waste, primarily sodium bromide, which is easily separated. The reaction proceeds efficiently at room temperature and atmospheric pressure, eliminating the need for specialized heating equipment or rigorous exclusion of moisture and oxygen. This simplicity translates directly into operational excellence, allowing for easier handling and a significant reduction in production lead times for complex unnatural amino acids.
Mechanistic Insights into Sodium Methoxide-Catalyzed Cyclization
The core of this technological advancement lies in the elegant mechanism facilitated by sodium methoxide (NaOMe) as the catalyst. The reaction initiates with the deprotonation of the phenoxyamide substrate, generating a nucleophilic species that attacks the electron-deficient triple bond of the bromopropiolate. This intramolecular cyclization is highly favored under the mild basic conditions provided by the methoxide ion, leading to the rapid formation of the furan ring system. Unlike radical-based metal catalysis which can lead to unpredictable side reactions, this ionic pathway offers superior control over regioselectivity and stereochemistry. The use of ethyl acetate as the solvent further stabilizes the transition states and ensures good solubility for both the organic substrates and the inorganic base, creating a homogeneous reaction environment that maximizes collision frequency and reaction rates.
From an impurity control perspective, this mechanism is exceptionally clean. The absence of transition metals removes the risk of metal-mediated decomposition or oligomerization, which are common pitfalls in traditional heterocycle synthesis. The primary byproduct, sodium bromide, precipitates out or remains in the aqueous phase during workup, ensuring that the final organic product is obtained with high purity. This inherent cleanliness is crucial for R&D teams focusing on structure-activity relationship (SAR) studies, where trace impurities can confound biological data. Moreover, the robustness of the base-catalyzed system allows for a broad tolerance of functional groups, enabling the synthesis of diverse derivatives without the need for extensive protecting group strategies, thereby streamlining the overall synthetic design.
How to Synthesize Benzofuran β-Dehydrogenated Unnatural Amino Acids Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and reaction parameters to ensure optimal yields. The patent outlines a straightforward protocol where the phenoxyamide substrate and sodium methoxide are dissolved in ethyl acetate, followed by the addition of the bromopropiolate. The reaction mixture is then stirred at ambient temperature for a duration of 6 to 12 hours. This simplicity belies the sophistication of the chemical transformation, which effectively constructs two new bonds and a heterocyclic ring in one pot. For detailed operational procedures and safety considerations, please refer to the standardized synthesis guide below.
- Dissolve 1 mmol of phenoxyamide substrate and 2 mmol of sodium methoxide in 10 ml of ethyl acetate solvent under standard conditions.
- Add 2 mmol of bromopropiolate (such as ethyl 3-bromopropynoate) to the reaction mixture while maintaining room temperature.
- Stir the reaction mixture at room temperature for 6 to 12 hours to complete the cyclization, followed by simple workup to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend beyond mere chemical curiosity. The elimination of expensive transition metal catalysts represents a direct reduction in raw material costs, while the use of commodity solvents like ethyl acetate ensures stable pricing and easy sourcing. The simplified workup procedure reduces the demand for specialized purification resins or extensive chromatography, lowering both consumable costs and labor hours. Furthermore, the mild reaction conditions enhance process safety, reducing the risk of thermal runaways and allowing for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys resistant to corrosive metal salts.
- Cost Reduction in Manufacturing: The economic impact of switching to this base-catalyzed route is profound. By removing the dependency on palladium or copper catalysts, manufacturers avoid the volatility associated with precious metal markets. Additionally, the high atom economy means that a larger proportion of the starting mass ends up in the final product, minimizing waste disposal fees. The ability to run the reaction at room temperature significantly cuts energy costs associated with heating and cooling cycles, contributing to a leaner and more competitive cost structure for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Phenoxyamides and bromopropiolates are commodity chemicals that can be sourced from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility quality or environmental factors, ensuring consistent batch-to-batch quality. This reliability is essential for maintaining continuous production schedules and meeting the tight delivery windows demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is straightforward due to the absence of exothermic hazards and the use of green solvents. The generation of sodium bromide as the primary waste stream simplifies effluent treatment, aligning with increasingly strict environmental regulations. This eco-friendly profile not only reduces compliance costs but also enhances the corporate sustainability image, a factor that is becoming increasingly important in vendor selection processes for major multinational corporations seeking green supply chains.
Frequently Asked Questions (FAQ)
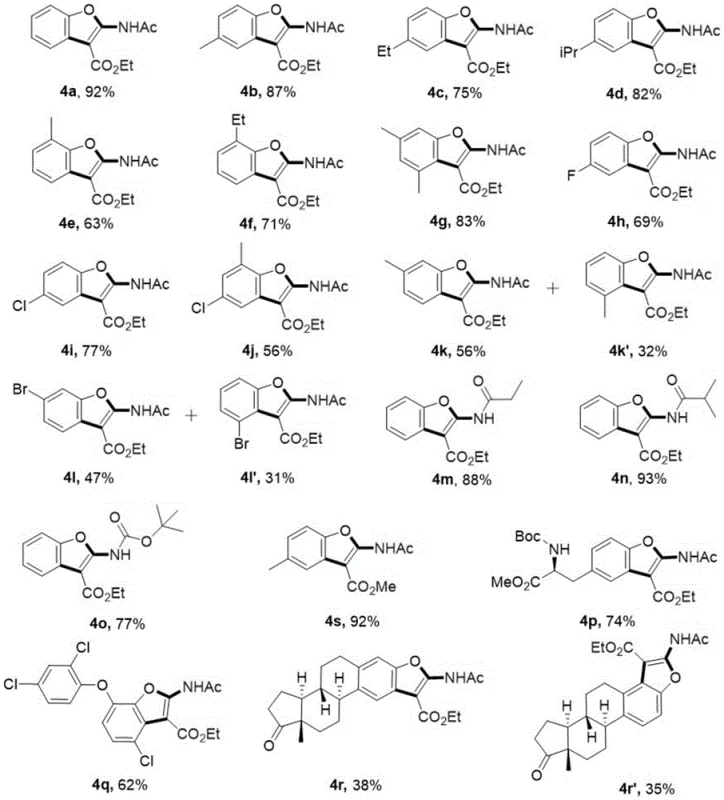
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, unlike conventional methods that rely on palladium or copper catalysts, this patented process utilizes sodium methoxide, a cheap and readily available base, eliminating the need for costly metal removal steps.
Q: What is the substrate scope for this benzofuran synthesis?
A: The method demonstrates excellent adaptability, successfully synthesizing 22 different derivatives with various substituents including methyl, ethyl, halogen (Cl, Br, F), and bulky groups like tert-butoxy, with yields ranging from 31% to 93%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction proceeds at room temperature and atmospheric pressure using ethyl acetate as a green solvent, producing minimal waste (primarily sodium bromide), which makes it highly scalable and environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced intermediates like benzofuran β-dehydrogenated unnatural amino acids play in the next generation of therapeutics. Our team of expert chemists has thoroughly analyzed the pathway described in CN108047177B and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are designed to handle complex heterocyclic chemistry with stringent purity specifications, supported by rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your drug discovery programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can accelerate your project timelines and optimize your overall development budget.
