Scalable Synthesis of High-Potency Diosgenin Quaternary Ammonium Salt Derivatives for Oncology
Scalable Synthesis of High-Potency Diosgenin Quaternary Ammonium Salt Derivatives for Oncology
The pharmaceutical industry is constantly seeking novel scaffolds that can overcome the limitations of natural products while retaining their inherent biocompatibility. Patent CN110452283B introduces a significant advancement in this domain by disclosing a series of diosgenin quaternary ammonium salt derivatives designed specifically for antitumor applications. This technology addresses the critical challenge of low bioactivity often associated with native steroidal sapogenins by chemically modifying the C3-hydroxyl group. The disclosed methodology provides a robust pathway for generating high-value pharmaceutical intermediates that demonstrate superior inhibitory effects against prominent cancer cell lines such as A549 lung cancer and H1975 lung adenocarcinoma. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural novelty of these compounds is essential for integrating them into next-generation oncology pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct utilization of diosgenin as an anticancer agent has been hampered by its relatively modest potency and poor water solubility, which restricts its therapeutic window and bioavailability. Conventional modification strategies often focus on the A-ring or F-ring but frequently fail to introduce sufficient polarity or cationic character required for enhanced cellular uptake and membrane interaction. Furthermore, traditional esterification methods using harsh acidic conditions can lead to the degradation of the sensitive steroid backbone or the spiroketal side chain, resulting in complex impurity profiles that are difficult to separate. These synthetic bottlenecks not only increase the cost of goods but also delay the timeline for preclinical evaluation, making the development of efficient, mild, and high-yielding protocols a priority for modern medicinal chemistry teams.
The Novel Approach
The innovative strategy outlined in the patent employs a two-step sequence that elegantly functionalizes the diosgenin core without compromising its structural integrity. By first converting the C3-hydroxyl group into a reactive halo-ester intermediate using mild coupling agents, the process sets the stage for a clean nucleophilic substitution. This intermediate then reacts with a diverse array of tertiary amines to form stable quaternary ammonium salts, introducing a permanent positive charge that significantly enhances water solubility and biological interaction. 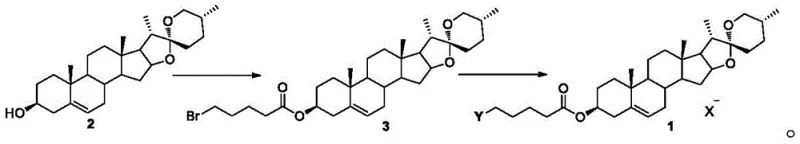 This approach allows for rapid diversification of the side chain, enabling the fine-tuning of physicochemical properties to optimize antitumor efficacy. The result is a library of derivatives that consistently outperform the parent diosgenin in cytotoxicity assays, offering a compelling value proposition for reliable pharmaceutical intermediate supplier partnerships.
This approach allows for rapid diversification of the side chain, enabling the fine-tuning of physicochemical properties to optimize antitumor efficacy. The result is a library of derivatives that consistently outperform the parent diosgenin in cytotoxicity assays, offering a compelling value proposition for reliable pharmaceutical intermediate supplier partnerships.
Mechanistic Insights into Esterification and Quaternization
The core of this synthetic route relies on a classic carbodiimide-mediated esterification followed by an SN2 quaternization mechanism. In the first step, 5-halovaleric acid is activated by EDC·HCl in the presence of a catalytic amount of DMAP, forming an O-acylisourea intermediate that is highly susceptible to nucleophilic attack by the C3-hydroxyl of diosgenin. This reaction proceeds efficiently at room temperature (25°C) in dichloromethane, minimizing thermal stress on the steroid skeleton. The formation of the ester linkage is crucial as it serves as a spacer that positions the reactive halogen atom at an optimal distance for the subsequent displacement reaction. Careful control of stoichiometry and reaction time ensures high conversion to the bromo-ester intermediate, which is isolated as a white solid with high purity after simple aqueous workup and recrystallization.
In the second mechanistic phase, the bromo-ester intermediate undergoes quaternization upon heating with excess tertiary amine in acetonitrile at 80°C. The nitrogen lone pair of the amine attacks the terminal carbon bearing the bromine atom, displacing the bromide ion and forming a stable quaternary ammonium cation. This transformation is driven by the high nucleophilicity of the amine and the good leaving group ability of the bromide. The resulting cationic head group not only improves the aqueous solubility of the molecule but also facilitates electrostatic interactions with negatively charged cell membranes, potentially enhancing cellular internalization. The versatility of this mechanism allows for the incorporation of various amine structures, from simple alkyl amines to complex heterocycles, providing a powerful tool for structure-activity relationship studies in drug discovery.
How to Synthesize Diosgenin Quaternary Ammonium Salts Efficiently
The practical execution of this synthesis is designed for scalability and reproducibility, making it suitable for both laboratory research and commercial production environments. The process begins with the activation of the carboxylic acid linker, followed by conjugation to the steroid core, and concludes with the quaternization step which locks in the desired pharmacological features. Operators should pay close attention to the purification methods described, such as silica gel chromatography or specific solvent recrystallization protocols, to ensure the removal of unreacted amines and urea byproducts. 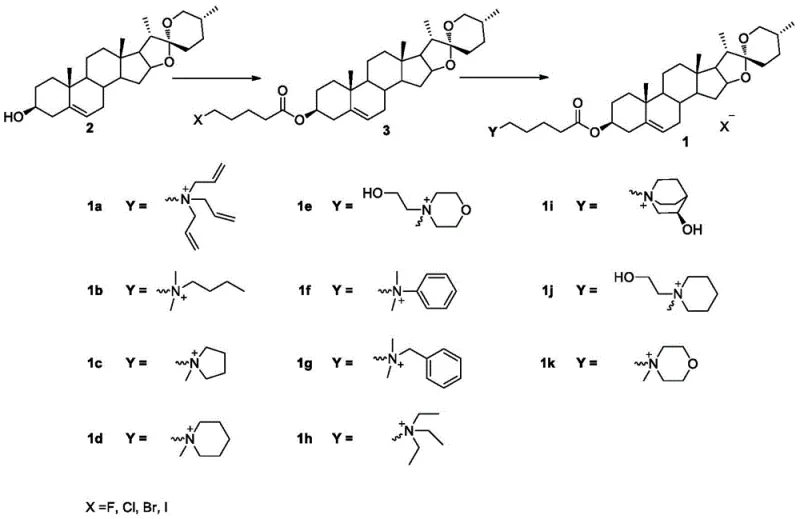
- Dissolve diosgenin in dichloromethane and react with 5-halovaleric acid using EDC·HCl and DMAP at 25°C to form the bromo-ester intermediate.
- Dissolve the intermediate in acetonitrile, add the specific tertiary amine, and heat to 80°C until reaction completion monitored by TLC.
- Concentrate the reaction mixture under reduced pressure and purify the final quaternary ammonium salt via silica gel chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this synthetic route offers distinct advantages regarding raw material availability and process safety. The starting material, diosgenin, is a naturally abundant steroidal sapogenin derived from plant sources, ensuring a stable and sustainable supply chain that is less vulnerable to the fluctuations often seen with fully synthetic petrochemical feedstocks. Additionally, the reagents used, such as EDC·HCl, DMAP, and common tertiary amines, are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency. The reaction conditions are relatively mild, avoiding the need for cryogenic temperatures or high-pressure equipment, which simplifies the engineering requirements for scale-up and lowers the capital expenditure associated with manufacturing infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of standard organic solvents like dichloromethane and acetonitrile contribute to a streamlined cost structure. The high yields reported for the intermediate and final products minimize material waste and reduce the overall cost per kilogram of the active pharmaceutical ingredient. Furthermore, the straightforward workup procedures, which often involve simple filtration or crystallization rather than complex distillation, lower energy consumption and labor costs significantly.
- Enhanced Supply Chain Reliability: The robustness of the two-step synthesis ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and meeting delivery schedules. The ability to synthesize a wide range of derivatives from a common intermediate allows manufacturers to respond quickly to changing market demands or specific client requirements without retooling the entire production line. This flexibility acts as a buffer against supply disruptions and enables just-in-time manufacturing strategies for downstream partners.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods that might utilize heavy metals or toxic reagents. The byproducts, primarily urea derivatives and inorganic salts, are easier to treat and dispose of in accordance with environmental regulations. The scalability of the reaction from gram to kilogram scales has been demonstrated in the patent examples, confirming that the chemistry translates well to industrial reactors without significant loss of efficiency or selectivity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these novel diosgenin derivatives. Understanding these details helps stakeholders evaluate the feasibility of incorporating these intermediates into their own development programs. The answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of these diosgenin derivatives over the parent compound?
A: The quaternary ammonium salt derivatives exhibit significantly enhanced antitumor activity against various cancer cell lines including A549, H1975, and Aspc-1 compared to unmodified diosgenin.
Q: What are the critical reaction conditions for the quaternization step?
A: The quaternization requires heating the bromo-ester intermediate with excess tertiary amine in acetonitrile at 80°C to ensure complete conversion to the quaternary salt.
Q: How is the purity of the final intermediate ensured?
A: High purity is achieved through rigorous workup procedures including washing with acid/base solutions and final purification via silica gel column chromatography or recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diosgenin Derivatives Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our team is equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications for every batch of diosgenin quaternary ammonium salts we produce. We understand the critical nature of oncology intermediates and are committed to delivering materials that meet the highest standards of quality and consistency required for clinical trials and commercial drug launch.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to optimize your supply chain and reduce overall production expenses. Let us help you accelerate your drug development timeline with our proven expertise in steroidal chemistry and process optimization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
